Abstract
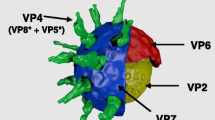
Murine rotavirus gene six encoding the 41 kDa group specific capsid structural protein VP6 was stably inserted into the Solanum tuberosum genome by Agrobacterium tumefaciens mediated transformation. The molecular mass of plant synthesized VP6 capsid protein determined by immunoblot was similar to the size of both purified virus VP6 monomeric peptides and partially assembled virus-like particles. The amount of VP6 protein synthesized in transgenic potato leaf and tuber was determined by enzyme-linked immunosorbent assay to be approximately 0.01% of total soluble protein. Oral immunization of CD-1 mice with transformed potato tuber tissues containing VP6 capsid protein generated measurable titers of both anti-VP6 serum IgG and intestinal IgA antibodies. The presence of detectable humoral and intestinal antibody responses against the rotavirus capsid protein following mucosal immunization provides an optimistic basis for the development of edible plant vaccines against enteric viral pathogens.
Similar content being viewed by others
References
Arakawa T, Chong DK, Merritt JL and Langridge WH (1997) Expression of cholera toxin B subunit oligomers in transgenic potato plants. Transgenic Res 6: 403–413.
Arakawa T, Yu J, Chong DKX, Hough J, Engen PC and Langridge WH (1998) A plant-based cholera toxin B subunit–insulin fusion protein protects against the development of autoimmune diabetes. Nat Biotechnol 16: 934–938.
Bajolet O and Chippaux-Hyppolite C (1998) Rotavirus and other viruses of diarrhea. Bull Soc Pathol Exot 91: 432–437.
Ball JM, Tian P, Zeng CQY, Morris AP and Estes MK (1996) Age-dependent diarrhea induced by a rotavirus nonstructural glycoprotein. Science 272: 101–104.
Bishop RF, Davidson GP, Homes IH and Ruck BJ (1973) Virus particles in epithelial cells of duodenal mucosa from children with viral gastroenteritis. Lancet 2: 1281–1283.
Bishop RF, Barnes GL, Cipriani E and Lund JS (1983) Clinical immunity after neonatal rotavirus infection. A prospective longitudinal study in young children. N Engl J Med 309: 72–76.
Burns JW, Krishnaney AA, Phuoct VO, Rouse RV, Anderson LJ and Greenberg HB (1995) Analyses of homologous rotavirus infection in the mouse model. Virology 207: 143–153.
Burns JW, Pajouh MS, Krishnaney AA and Greenberg HB (1996) Protective effect of rotavirus VP6-specific IgA monoclonal antibodies that lack neutralizing activity. Science 272: 104–107.
Chen SC, Fynan EF, Robinson HL, Lu S, Greenberg HB, Santoro JC et al. (1997) Protective immunity induced by rotavirus DNA vaccines. Vaccine 8: 899–902.
Conner ME, Zarley CD, Parsons BHU, Drabinski D, Greiner S, Smith R et al. (1996) Virus-like particles as a rotavirus subunit vaccine. J Infect Dis 174: 88–92.
Crawford SE, Labbe M, Cohen J, Burroughs MH, Zhou YJ and Estes MK (1994) Characterization of virus-like particles produced by the expression of rotavirus capsid proteins in insect cells. J Virol 68: 5945–5952.
Estes MK and Cohen J (1989) Rotavirus gene structure and function. Microbiol Rev 53: 410–449.
Eydelloth RS, Vonderfecht SL, Sheridan JF, Enders LD and Yollen RH (1984) Kinetics of viral replication and local and systemic immune responses in experimental rotavirus infection. J Virol 50: 947–950.
Haq TA, Mason HS, Clements JD and Arntzen CJ (1995) Oral immunization with a recombinant bacterial antigen produced in transgenic plants. Science 268: 714–716.
Hoshino Y and Kapikian AZ (1994a) Rotavirus vaccine development for the prevention of severe diarrhea in infants and young children. Trends Microbiol 2: 242–249.
Hoshino Y and Kapikian AZ (1994b) Rotavirus antigens. Curr Top Microbiol Immunol 185: 179–227.
Hsu GG, Bellamy AR and Yeager M (1997) Projection structure of VP6, the rotavirus inner capsid protein, and comparison with bluetongue VP7. J Mol Biol 272: 362–368.
Ishida S, Feng N, Tang B, Gilbert JM and Greenberg HB (1996) Quantification of systemic and local immune responses to individual rotavirus proteins during rotavirus infection in mice. J Clin Microbiol 34: 1694–1700.
Ishida SI, Feng N, Gibert JM, Tang B and Greenberg HB (1997) Immune responses to individual rotavirus proteins following heterologous and homologous rotavirus infection in mice. J Infect Dis 175: 1317–1323.
Langridge WHR, Fitzgerald KJ, Koncz C, Schell J and Szalay AA (1989) The dual promoter of A. tumefaciens mannopine synthase genes is regulated by plant growth hormones. Proc Natl Acad Sci USA 86: 3219–3223.
Mason HS and Arntzen CJ (1995) Transgenic plants as vaccine production systems. Trends Biotechnol 13: 388–392.
Mason HS, Ball JM, Shi JJ, Jiang X, Estes MK and Arnten CJ (1996) Expression of Norwalk virus capsid protein in transgenic tobacco and potato and its oral immunogenicity in mice. Proc Natl Acad Sci 93: 5335–5340.
O'Brien GJ, Bryant CJ, Voogd C, Greenberg HB, Gardner RC and Bellamy AR (2000) Rotavirus VP6 expressed by PVX vectors in Nicotiana benthamiana coats PVX rods and also assembles into virus like particles. Virology 270(2): 444–453.
Offit PA and Dudzik KI (1990) Rotavirus-specific cytotoxic T lymphocytes passively protect against gastroenteritis in suckling mice. J Virol 64: 6325–6328.
O'Neal CM, Crawford SE, Estes MK and Conner ME (1997) Rotavirus virus-like particles administered mucosally induce protective immunity. J Virol 71: 8707–8717.
Sabara M, Parker M, Aha P, Cosco C, Gibbons E, Parsons S et al. (1991) Assembly of double-shelled rotavirus-like particles by simultaneous expression of recombinant VP6 and VP7 proteins. J Virol 65: 6994–6997.
Velten J, Velten L, Hain R and Schell J (1984) Isolation of a dual plant promoter fragment from the Ti Plasmid of Agrobacterium tumefaciens. EMBO J 12: 2723–2730.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, J., Langridge, W. Expression of Rotavirus Capsid Protein VP6 in Transgenic Potato and Its Oral Immunogenicity in Mice. Transgenic Res 12, 163–169 (2003). https://doi.org/10.1023/A:1022912130286
Issue Date:
DOI: https://doi.org/10.1023/A:1022912130286