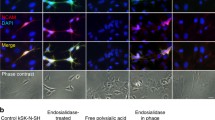
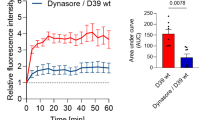
Abstract
There is a molecular mimicry between the polysialic acid polysaccharide of bacterial pathogens causing sepsis and meningitis, and the carbohydrate units of the neural cell adhesion molecule NCAM. We investigated whether bacteriophage mutants with catalytically disabled endosialidase, which bind but do not cleave polysialic acid, could recognise and bind to bacterial and eukaryotic polysialic acid. In nitrocellulose dot blot assay the mutant bacteriophages, but not the wild-type phages, remained specifically bound to polysialic acid–containing bacteria including Escherichia coli K1 and K92, group B meningococci, Mannheimia (Pasteurella) haemolytica A2, and Moraxella nonliquefaciens. A minimum binding requirement was determined to be 10 sialyl residues in the polysialic acid chain. In Western blots the mutant phages specifically bound to the embryonic polysialylated form of NCAM, but not to the adult less sialylated form of the molecule. The mutant phages together with secondary anti-phage antibodies were subsequently successfully used in fluorescence microscopy of cultured cells and light microscopy of paraffin-embedded tissue sections as a probe for the eukaryotic polysialic acid. Thus, mutant bacteriophages of meningitis causing bacteria bind to and detect the molecularly mimicked polysialic acid of the neural cell adhesion molecule in host tissues.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Jennings HJ, Katzenellenbogen E, Lugowski C, Michon F, Roy R, Kasper DL, Structure, conformation, and immunology of sialic acid-containing polysaccharides of human pathogenic bacteria, Pure Appl Chem 56, 893–905 (1984).
Adlam C, Knights JM, Mugridge A, Williams JM, Lindon JC, Production of colominic acid by Pasteurella haemolytica serotype A2 organism, FEMS Microbiol Lett 42, 23–5 (1987).
Devi SJN, Schneerson R, Egan W, Vann WF, Robbins JB, Shiloach J, Identity between polysaccharide antigens of Moraxella nonliquefaciens, group B Neisseria meningitidis, and Escherichia coli K1 (non-O acetylated), Infect Immun 59, 732–6 (1991).
Finne J, Occurrence of unique polysialosyl carbohydrate units in glycoproteins of developing brain, J Biol Chem 257, 11966–70 (1982).
Finne J, Finne U, Deagostini-Bazin H, Goridis C, Occurrence of α2–8 linked polysialosyl units in a neural cell adhesion molecule, Biochem Biophys Res Commun 112, 482–7 (1983).
Rutishauser U, Polysialic acid and the regulation of cell interactions, Curr Opin Cell Biol 8, 679–84 (1996).
Kiss JZ, Rougon G, Cell biology of polysialic acid, Curr Opin Neurobiol 7, 640–6 (1997).
Troy FA,II, Polysialylation: From bacteria to brains, Glycobiology 2, 5–23 (1992).
Rutishauser U, Polysialic acid at the cell surface: Biophysics in service of cell interactions and tissue plasticity, J Cell Biochem 70, 304–12 (1998).
Finne J, Leinonen M, Mäkelä PH, Antigenic similarities between brain components and bacteria causing meningitis. Implications for vaccine development and pathogenesis, Lancet 2, 355–8 (1983).
Robbins JB, Vaccines for the prevention of encapsulated bacterial diseases: Current status, problems and prospects for the future, Immunochemistry 15, 839–54 (1978).
Frosch M, Görgen I, Boulnois GJ, Timmis KN, Bitter-Suermann D, NZB mouse system for production of monoclonal antibodies to weak bacterial antigens: Isolation of an IgG antibody to the polysaccharide of Escherichia coli K1 and group B meningococci, Proc Natl Acad Sci USA 82, 1194–8 (1985).
Dubois C, Okandze A, Figarella-Branger D, Rampini C, Rougon G, A monoclonal antibody against Meningococcus group B polysaccharides used to immunocapture and quantify polysialylated NCAM in tissues and biological fluids, J Immunol Methods 181, 125–35 (1995).
Sato C, Kitajima K, Inoue S, Seki T, Troy FA,II, Inoue Y, Characterization of the antigenic specificity of four different anti-(α2–>8-linked polysialic acid) antibodies using lipid-conjugated oligo/polysialic acids, J Biol Chem 270, 18923–8 (1995).
Gross RJ, Cheasty T, Rowe B, Isolation of bacteriophages specific for the K1 polysaccharide antigen of Escherichia coli, J Clin Microbiol 6, 548–50 (1977).
Lindberg AA, Bacterial surface carbohydrates and bacteriophage adsorption. In Surface carbohydrates of the prokaryotic cell, edited by Sutherland I (Academic Press, New York, 1977), pp. 289–356.
Pelkonen S, Aalto J, Finne J, Differential activities of bacteriophage depolymerase on bacterial polysaccharide: Binding is essential but degradation is inhibitory in phage infection of K1-defective Escherichia coli, J Bacteriol 174, 7757–61 (1992).
Korhonen TK, Valtonen MV, Parkkinen J, Väisänen-Rhen V, Finne J, Ørskov F, Ørskov I, Svenson SB, Mäkelä PH, Serotypes, hemolysin production, and receptor recognition of Escherichia coli strains associated with neonatal sepsis and meningitis, Infect Immun 48, 486–91 (1985).
Pelkonen S, Finne J, A rapid turbidimetric assay for the study of serum sensitivity of Escherichia coli, FEMS Microbiol Lett 42, 53–7 (1987).
Gupta DS, Jann B, Schmidt G, Golecki JR, Ørskov I, Ørskov F, Jann K, Coliphage K5, specific for E. coli exhibiting the capsular K5 antigen, FEMS Microbiol Lett 14, 75–8 (1982).
Finne J, Mäkelä PH, Cleavage of the polysialosyl units of brain glycoproteins by a bacteriophage endosialidase. Involvement of a long oligosaccharide segment in molecular interactions of polysialic acid, J Biol Chem 260, 1265–70 (1985).
Pelkonen S, Häyrinen J, Finne J, Polyacrylamide gel electrophoresis of the capsular polysaccharides of Escherichia coliK1and other bacteria, J Bacteriol 170, 2646–53 (1988).
Pelkonen S, Finne J, Polyacrylamide gel electrophoresis of capsular polysaccharides of bacteria, Methods Enzymol 179, 104–10 (1989).
Wedege E, Svenneby G, Effects of the blocking agents bovine serum albumin and Tween 20 in different buffers on immunoblotting of brain proteins and marker proteins, J Immunol Methods 88, 233–7 (1986).
Laemmli UK, Cleavage of the structural proteins during the assembly of the head of bacteriophage T4, Nature 227, 680–5 (1970).
Finne J, Bitter-Suermann D, Goridis C, Finne U, An IgG monoclonal antibody to group B meningococci crossreacts with developmentally regulated polysialic acid units of glycoproteins in neural and extraneural tissues, J Immunol 138, 4402–7 (1987).
Kyhse-Andersen J, Electroblotting of multiple gels: A simple apparatus without buffer tank for rapid transfer of proteins from polyacrylamide to nitrocellulose, J Biochem Biophys Methods 10, 203–9 (1984).
Sternberger LA, The unlabeled antibody method. Hormone receptor, Golgi-like and dual color immunocytochemistry, J Histochem Cytochem 27, 1658–9 (1979).
Kwiatkowski B, Boschek B, Thiele H, Stirm S, Endo-N-acetylneuraminidase associated with bacteriophage particles, J Virol 43, 697–704 (1982).
Cremer H, Lange R, Christoph A, Plomann M, Vopper G, Roes J, Brown R, Baldwin S, Kraemer P, Scheff S, Barthels D, Rajewsky K, Wille W, Inactivation of the N-CAM gene in mice results in size reduction of the olfactory bulb and deficits in spatial learning, Nature 367, 455–9 (1994).
Pelkonen S, Pelkonen J, Finne J, Common cleavage pattern of polysialic acid by bacteriophage endosialidases of different properties and origins, J Virol 63, 4409–16 (1989).
Jennings HJ, Roy R, Michon F, Determinant specificities of the groups B and C polysaccharides of Neisseria meningitidis, J Immunol 134, 2651–7 (1985).
Häyrinen J, Bitter-Suermann D, Finne J, Interaction of meningococcal group B monoclonal antibody and its Fab fragment with α2–8-linked sialic acid polymers: Requirement of a long oligosaccharide segment for binding, Mol Immunol 26, 523–9 (1989).
Häyrinen J, Jennings H, Raff HV, Rougon G, Hanai N, Gerardy-Schahn R, Finne J, Antibodies to polysialic acid and its N-propyl derivative: Binding properties and interaction with human embryonal brain glycopeptides, J Infect Dis 171, 1481–90 (1995).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aalto, J., Pelkonen, S., Kalimo, H. et al. Mutant bacteriophage with non-catalytic endosialidase binds to both bacterial and eukaryotic polysialic acid and can be used as probe for its detection. Glycoconj J 18, 751–758 (2001). https://doi.org/10.1023/A:1021147316647
Issue Date:
DOI: https://doi.org/10.1023/A:1021147316647