Abstract
The small keratan sulphate proteoglycan, fibromodulin, has been isolated from pooled human articular cartilage. The main chain repeat region and the chain caps from the attached N-linked keratan sulphate chains have been fragmented by keratanase II digestion, and the oligosaccharides generated have been reduced and isolated. Their structures and abundance have been determined by high pH anion-exchange chromatography.
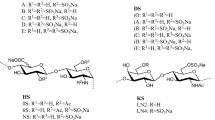
These regions of the keratan sulphate from human articular cartilage fibromodulin have been found to have the following general structure:
Significantly, both α(2-6)- and α(2-3)-linked N-acetyl-neuraminic acid have been found in the capping oligosaccharides. Fucose, which is α(1-3)-linked as a branch to N-acetylglucosamine, has also been found along the length of the repeat region and in the capping region. The chains, which have been found to be very highly sulphated, are short; the length of the repeat region and chain caps is ca. nine disaccharides.
These data demonstrate that the structure of the N-linked keratan sulphate chains of human articular cartilage fibromodulin is similar, in general, to articular cartilage derived O-linked keratan sulphate chains. Further, the general structure of the keratan sulphate chains attached to human articular cartilage fibromodulin has been found to be generally similar to that of both bovine and equine articular cartilage fibromodulin.
Abbreviations: KS, keratan sulphate; IEC, ion-exchange chromatography; ELISA, enzyme linked immunosorbent assay; Gal, β-D-galactose; Fuc, α-L-Fucose; GlcNAc, N-acetylglucosamine (2-acetamido-β-D-glucose); GlcNAc-ol, N-acetylglucosaminitol (2-acetamido-D-glucitol); NeuAc, N-acetyl-neuraminic acid; 6S/(6S), O-ester sulphate group on C6 present/sometimes present; NMR -nuclear magnetic resonance; HPAE, high pH anion-exchange; PED, pulsed electrochemical detection; HPLC, high performance liquid chromatography
Similar content being viewed by others
References
Sztrolovics R, Chen XN, Grover J, Roughley PJ, Korenberg JR (1994) Genomics 23: 715–17.
Heinegard D, Larsson T, Sommarin Y, Franzen A, Paulsson M, Hedbom E (1986) J Biol Chem 261: 13866–72.
Oldberg A, Antonsson P, Lindblom K, Heinegard D (1989) EMBO J 8: 2601–4.
Garg HG, Lyon NB (1991) Adv Carbohydr Chem Biochem 49: 239–61.
Antonsson P, Heinegard D, Oldberg A (1993) Biochim Biophys Acta 1174: 204–6.
Antonsson P, Heinegard D, Oldberg A (1991) J Biol Chem 266: 16859–61.
Plaas AHK, Neame PJ, Nivens CM, Reiss L (1990) J Biol Chem 265: 20634–40.
Lauder RM, Huckerby TN, Nieduszynski IA (1994) Biochem J 302: 417–23.
Lauder RM, Huckerby TN, Nieduszynski IA (1995) Glycoconjugate J 12: 651–59.
Lauder RM, Huckerby TN, Nieduszynski IA (1996) Eur J Biochem 242: 402–9.
Vogel KG, Paulsson M, Heinegard D (1984) Biochem J 223: 587–97.
Hedbom E, Heinegard D (1989) J Biol Chem 264: 6898–905.
Noyori K, Jasin HE (1994) Arthr Rheum 37: 1656–63.
Hedlund H, Mengarelliwidholm S, Heinegard D, Reinholt FP, Svensson O (1994) Matrix Biol 14: 227–32.
Bray BA, Lieberman R, Meyer K (1967) J Biol Chem 242: 3373–80.
Krusius T, Finne J, Margolis RK, Margolis RU (1986) J Biol Chem 261: 8237–42.
Bhavanandan VP, Meyer K (1968) J Biol Chem 243: 1052–59.
Nieduszynski IA, Huckerby TN, Dickenson JM, Brown GM, Tai GH, Morris HG, Eady S (1990) Biochem J 271: 243–45.
Nakazawa K, Ito M, Yamagata T, Suzuki S (1989) In Keratan Sulphate: Chemistry, Biology and Chemical Pathology (Greiling H, Scott J eds) 99–110. London. The Biochemical Society.
Brown GM, Nieduszynski IA, Morris HG, Abram BL, Huckerby TN, Block JA (1995) Glycobiology 5: 311–7.
Farndale RW, Sayers CA, Barrett AJ (1982) Connect Tiss Res 9: 247–48
Dickenson JM, Huckerby TN, Nieduszynski IA (1992) Biochem J 282: 267–71.
Vliegenthart JFG, Dorland L, Vanhalbeek H (1983) Adv Carbohydr Chem Biochemi 41: 209–374.
Huckerby TN, Nieduszynski IA, Brown GM, Cockin GH (1991) Glycoconjugate J 8: 39–44.
Dickenson JM, Huckerby TN, Nieduszynski IA (1991) Biochem J 278: 779–85.
Tai GH, Morris HG, Brown GM, Huckerby TN, Nieduszynski IA (1992) Biochem J 286: 231–34.
Macher BA, Buehler J, Scudder P, Knapp W, Feizi T (1988) J Biol Chem 263: 10186–91.
Brown GM, Huckerby TN, Nieduszynski IA (1996) Biochem J 319: 137–41.
Brown GM, Huckerby TN, Morris HG, Abram BL, Nieduszynski IA (1994) Biochemistry 33: 4836–46.
Tai GH, Huckerby TN, Nieduszynski IA (1994) Carbohydr Res 255: 303–9.
Brown GM, Huckerby TN, Morris HG, Nieduszynski IA (1992) Biochem J 286: 235–41.
Feizi T. (1991) Trends Biochem Sci 16: 84–86
Hounsell EF. (1987) Chem Soc Rev 16: 161–85.
Oeben M, Keller R, Stuhlsatz HW, Greiling H (1987) Biochem J 248: 85–93.
Stuhlstatz HW, Keller R, Becker G, Oeben M, Lennartz L, Fisher DC, Greiling H (1989) In Keratan Sulphate: Chemistry, Biology and Chemical Pathology. (Greiling H, Scott J eds) 1–11. London. The Biochemical Society.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lauder, R.M., Huckerby, T.N. & Nieduszynski, I.A. The structure of the keratan sulphate chains attached to fibromodulin from human articular cartilage. Glycoconj J 14, 651–660 (1997). https://doi.org/10.1023/A:1018552913584
Issue Date:
DOI: https://doi.org/10.1023/A:1018552913584