Abstract
Purpose. To determine the extent of amino group crosslinking in gelatin matrices by chemical assay, and to compare these results to crosslinking evaluations from swelling measurements.
Methods. Matrices crosslinked with a water soluble carbodiimide (EDC/G), glutaraldehyde (GTA/G), as well as a GTA crosslinked matrix prepared from gelatin modified to contain 230% greater crosslinking sites (GTA/Mod) were evaluated. Crosslinking extent, X c, was determined by a UV assay of uncrosslinked amino groups before and after crosslinking, and was used to obtain crosslinking densities. Equilibrium swelling ratios, Q m at 37°C in isotonic pH 7.4 were used to calculate crosslinking degree from the Flory equation for swelling of ionic polymers for comparison to the chemically determined crosslinking densities.
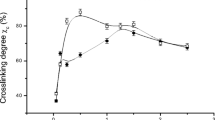
Results. Of the original 33 × 10−5 moles ε-amino groups/g gelatin, 91 to 95% were crosslinked in EDC/G and GTA/G. GTA/Mod lost 95% of the original 108 × 10−5 moles amino groups/g gelatin. Crosslinking densities were 4.1 × 10−4 and 4.2 × 10−4 moles/mL for EDC/G and GTA/G, respectively. The value for GTA/Mod increased to 14.2 × 10−4 moles/mL. Values of Q m followed the same trend. The Flory crosslinking degrees for both gelatin matrices were 12 × 10−4 and 13 × 10−4 moles/mL, respectively. The value for the more extensively crosslinked GTA/Mod was 280 × 10−4moles/mL.
Conclusions. The swelling and chemical evaluations of crosslinking are in general agreement for matrices with the lower of two crosslinking levels. The chemical determination appears suitable for evaluating amino group crosslinking in gelatin and it may be suitable for other proteinaceous materials.
Similar content being viewed by others
REFERENCES
O. P. Rubino, R. Kowalsky, and J. Swarbrick. Pharm. Res. 10:1059–1065 (1993).
D. L. Gilbert, T. Okano, T. Miyata, and S. W. Kim. Int. J. Pharm. 47:79–88 (1988).
M. M. Welz and C. M. Ofner III. J. Pharm. Sci. 81:85–90 (1992).
D. L. Gilbert and S. W. Kim. J. Biomed. Mat. Res. 24:1221–1239 (1990).
A. F. Habeeb. Anal. Biochem. 14:328–336 (1966).
R. Fields. In C. H. W. Hirs and S. N. Timasheff (eds.), Methods of Enzymology, Academic Press, New York, 1972, Vol. 25, pp. 464–468.
A. K. Hazra, S. P. Chock, and R. W. Albers. Anal. Biochem. 137:437–443 (1984).
W. A. Bubnis and C. M. Ofner III. Anal. Biochem. 207:129–133 (1992).
P. J. Flory and J. Rehner, Jr. J. Chem. Phys. 11:521–526 (1943).
P. J. Flory. Principles of Polymer Chemistry, Cornell University Press, Ithaca, p. 591 (1953).
J. C. Bray and E. W. Merrill. J. Appl. Polym. Sci. 17:3781–3796 (1973).
L. Brannon-Peppas and N. A. Peppas. J. Contr. Rel. 16:319–330 (1991).
W. Oppermann. In R. S. Harland and R. K. Prud'homme (eds.), Polyelectrolyte Gels-Properties, Preparation, and Applications, American Chemical Society, Washington DC, pp. 159–170 (1992).
A. Veis. The Macromolecular Chemistry of Gelatin, Academic Press, New York, 1964, chapter 1.
J. Boratynski and T. Zal. Anal. Biochem. 184:259–262 (1990).
D. G. Hoare and D. E. Koshland. J. Biol. Chem. 242:2447–2453 (1967).
M. R. Mejillano and R. H. Himes. J. Biol. Chem. 266:657–664 (1991).
W. A. Bubnis. Ph.D. Dissertation, Philadelphia College of Pharmacy and Science, 1994.
D. T. Cheung and M. E. Nimni. Conn. Tiss. Res. 10:187–199 (1982).
P. C. Hiemenz. Principles of Colloid and Surface Chemistry, Marcel Dekker, New York, p. 154 (1986).
C. M. Ofner III and H. Schott. J. Pharm. Sci. 74: 1317–1321 (1985).
I. D. Robinson. Phot. Sci. Eng. 8:220–224 (1964).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ofner, III, C.M., Bubnis, W.A. Chemical and Swelling Evaluations of Amino Group Crosslinking in Gelatin and Modified Gelatin Matrices. Pharm Res 13, 1821–1827 (1996). https://doi.org/10.1023/A:1016029023910
Issue Date:
DOI: https://doi.org/10.1023/A:1016029023910