Abstract
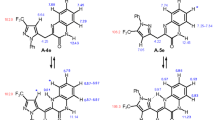
Condensation of 5-arylethynyl-3-diethylaminonaphthoquinones with NH2NH2 afford 3-benzyl-9-diethylaminobenzo[de]cinnolin-7-ones. The substituents in the phenyl ring have a pronounced effect on the reaction time and the yields of benzocinnolinones and by-products. The replacement of the arylethynyl substituent in the starting naphthoquinone by the 3-hydroxyalk-1-ynyl group leads to a change in the direction of cyclization resulting in substituted naphtho[1,8-cd]-1,2-diazepin-8-ones as condensation products.
Similar content being viewed by others
References
M. S. Shvartsberg, I. D. Ivanchikova, and S. F. Vasilevsky, Tetrahedron Lett., 1994, 35, 2077.
M. S. Shvartsberg, I. D. Ivanchikova, and S. F. Vasilevsky, Izv. Akad. Nauk, Ser. Khim., 1998, 2027 [Russ. Chem. Bull., 1998, 47, 1971 (Engl. Transl.)].
I. D. Ivanchikova, R. N. Myasnikova, and M. S. Shvartsberg, Izv. Akad. Nauk, Ser. Khim., 1998, 2031 [Russ. Chem. Bull., 1998, 46, 1975 (Engl. Transl.)].
M. S. Shvartsberg and I. D. Ivanchikova, Tetrahedron Lett., 2000, 41, 771.
I. I. Barabanov, I. D. Ivanchikova, and M. S. Shvartsberg, Mendeleev Commun., 2000, 188.
A. V. Piskunov, A. A. Moroz, and M. S. Shvartsberg, Izv. Akad. Nauk SSSR, Ser. Khim., 1987, 828 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1987, 36, 755 (Engl. Transl.)].
M. S. Shvartsberg, A. A. Moroz, N. V. Ivashkina, and S. B. Cherepanov, Izv. Akad. Nauk SSSR, Ser. Khim., 1986, 2485 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1986, 35, 2273 (Engl. Transl.)].
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ivanchikova, I.D., Myasnikova, R.N. & Shvartsberg, M.S. Cyclocondensation of 5-ethynyl-1,4-naphthoquinone derivatives with hydrazine. Russian Chemical Bulletin 50, 1668–1672 (2001). https://doi.org/10.1023/A:1013007223337
Issue Date:
DOI: https://doi.org/10.1023/A:1013007223337