Abstract
Purpose. To compare the enthalpy relaxation of amorphous sucrose and co-lyophilized sucrose-additive mixtures near the calorimetric glass transition temperature, so as to measure the effects of additives on the molecular mobility of sucrose.
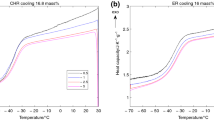
Methods. Amorphous sucrose and sucrose-additive mixtures, containing poly(vinylpyrrolidone) (PVP), poly(vinylpyrrolidone-co-vinyl-acetate) (PVP/VA) dextran or trehalose, were prepared by lyophilization. Differential scanning calorimetry (DSC) was used to determine the area of the enthalpy recovery endotherm following aging times of up to 750 hours for the various systems. This technique was also used to compare the enthalpy relaxation of a physical mixture of amorphous sucrose and PVP.
Results. Relative to sucrose alone, the enthalpy relaxation of co-lyophilized sucrose-additive mixtures was reduced when aged for the same length of time at a comparable degree of undercooling in the order: dextran ≈ PVP > PVP/VA > trehalose. Calculated estimates of the total enthalpy change required for sucrose and the mixtures to relax to an equilibrium supercooled liquid state (ΔH∞) were essentially the same and were in agreement with enthalpy changes measured at longer aging times (750 hours).
Conclusions. The observed decrease in the enthalpy relaxation of the mixtures relative to sucrose alone indicates that the mobility of sucrose is reduced by the presence of additives having a Tg that is greater than that of sucrose. Comparison with a physically mixed amorphous system revealed no such effects on sucrose. The formation of a molecular dispersion of sucrose with a second component, present at a level as low as 10%, thus reduces the mobility of sucrose below Tg, most likely due to the coupling of the molecular motions of sucrose to those of the additive through molecular interactions.
Similar content being viewed by others
REFERENCES
B. C. Hancock and G. Zografi. Characteristics and significance of the amorphous state in pharmaceutical systems. J. Pharm. Sci. 86:1–12 (1997).
A. Saleki-Gerhardt and G. Zografi. Non-isothermal and isothermal crystallization of sucrose from the amorphous state. Pharm. Res. 11:1166–1173 (1994).
S. L. Shamblin, E. Y. Huang, and G. Zografi. The effects of colyophilized polymeric additives on the glass transition temperature and crystallization of amorphous sucrose. J. Therm. Anal. 47:1567–1579 (1996).
J. Cowie and R. Ferguson. Physical aging studies in poly(vinyl methyl ether). 1. Enthalpy relaxation as a function of aging temperature. Macromol. 22:2307–2312 (1989).
B. C. Hancock, S. L. Shamblin, and G. Zografi. The molecular mobility of amorphous pharmaceutical solids below their glass transition temperatures. Pharm. Res. 12:799–806 (1995).
S. L. Shamblin. The Characteristics of Sucrose-Polymer Mixtures in the Amorphous State. Ph.D. Thesis, The University of Wisconsin-Madison, Madison, 1997.
S. P. Duddu, G. Zhang, and P. R. Dal Monte. The relationship between protein aggregation and molecular mobility below the glass transition temperature of lyophilized formulations containing a monoclonal antibody. Pharm. Res. 14:596–599 (1997).
G. Blond. Mechanical properties of frozen model solutions. J. Food Eng. 22:253–269 (1994).
V. Buhler, Kollidone, Second ed., BASF Aktiengesellschaft, Ludwigshafen, 1993.
S. L. Shamblin, L. S. Taylor, and G. Zografi. The mixing behavior of binary amorphous systems. J. Pharm. Sci. 87:694–701 (1998).
I. M. Hodge. Enthalpy relaxation and recovery in amorphous materials. J. Non-Cryst. Solids 169:211–266 (1994).
L. C. E. Struik. Physical Aging in Amorphous Polymers and Other Materials, Elsevier Scientific Publishing Company, New York, 1978.
A. Brunacci, J. Cowie, R. Ferguson, and I. McEwen. Enthalpy relaxation in glassy polystyrenes: 1. Polymer 38:865–870 (1997).
M. D. Ediger, C. A. Angell, and S. R. Nagel. Supercooled liquids and glasses. J. Phys. Chem. 100:13200–13212 (1996).
K. Karatasos, S. H. Anastasiadis, A. J. Semenov, G. Fytas, M. Pitsikalis, and N. Hadijichristidis. Composition fluctuation effects on dielectric normal-mode relaxation in diblock copolymers I. Weak segregation regime. Macromol. 27:3543–3552 (1994).
A. A. C. M. Oudhuis and G. ten Brinke. Enthalpy relaxation and concentration fluctuations in blends of polystyrene and poly(oxy-2,6-dimethyl-1,4-phenylene). Macromol. 25:698–702 (1992).
J. R. Fried, F. E. Karasz, and W. J. MacKnight. Compatibility of poly(2,6-dimethyl-1,4-phenylene oxide) (ppo)/poly(styrene-co-4-chlorostyrene) blends. I. Differential scanning calorimetry and density studies. Macromol. 11:150–158 (1978).
K. Assman and H. A. Schneider. The thermal analysis of polymer blends of poly(ethylene oxide)/poly(methyl methacrylate). J. Therm. Anal. 35:459–468 (1989).
J. A. Pomposo, I. Eguiazabal, E. Calahorra, and M. Cortazar. Glass transition behaviour and interactions in poly(p-vinyl phenol)/polymethacrylate blends. Polymer 34:95–102 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shamblin, S.L., Zografi, G. Enthalpy Relaxation in Binary Amorphous Mixtures Containing Sucrose. Pharm Res 15, 1828–1834 (1998). https://doi.org/10.1023/A:1011997721086
Issue Date:
DOI: https://doi.org/10.1023/A:1011997721086