Abstract
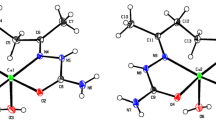
Reaction of Cu(II) triflate with an excess of 2-aminopyrimidine (ampym) in ethanol followed by slow crystallization results in a most unusual crystal lattice, which can be considered as consisting of two interpenetrating sublattices. The compound analyzes as [Cu(ampym)2(H2O)2(CF3SO3)2(ampym)4]. Crystal data: Triclinic, P\(\overline 1\), a = 7.6179(5), b = 11.4311(14), c = 11.8373(13) Å, α = 84.098(9), β = 79.998(7), γ = 84.253(8)°, Vol = 1010.151(8) Å, Z = 1, Dcalc = 1.592 g/cm3. One part of the lattice consists of the unprecendented linear chain of neutral ampym molecules; the chain is built up by Watson–Crick type bis-hydrogen bonds between imine N atoms and N-H groups of the NH2. This ampym chain is apparently stabilized by the other part in the crystal lattice, which can best be described by starting from the centrosymmetric trans-CuII(ampym)2(H2O)2 ion (Cu-N = 2.01 Å; Cu-O = 1.94 Å). This Cu ion is coordinated by two monodentate ampym ligands, two water molecules and two semi-coordinating triflate oxygen atoms. The Cu chromophore is held in position by hydrogen bonding towards a triflate ion (two times for symmetry reasons), and (again two times) a free ampym ligand. The triflate ion bridges a water hydrogen to a next-neighboring free ampym ligand. The noncoordinating ampym molecule has four bonding sites, of which two are used in H-bonding with the coordinated ligand, one with the triflate and the last one with the water hydrogen atom. The noncoordination ampym molecules in this sublattice finally form aromatic stacks with coordinated ampym and with itself in pairs (ring-ring distance 3.77-3.80 Å).
Similar content being viewed by others
References
Batten, S.R.; Robson, R. Angew.Chem. Int. Ed. 1998, 37, 1460.
Ouali, M.; Letellier, R.; Taillandier, E. J. Mol. Struc. 1996, 377, 57.
Marfurt, J.; Leumann, C. Angew. Chem. Int. Ed. 1998, 37, 175.
Sijbesma, R.P.; Beijer, F.H.; Brunsveld, L.; Folmer, B.J.B.; Hirschberg, J.H.K.K.; Lange, R.F.M.; Lowe, J.K.L.; Meijer, E.W. Science 1997, 278, 1601.
Russell, K.C.; Lehn, J.-M.; Kyritsakas, N.; DeCian, A.; Fischer, J. New J. Chem. 1998, 123.
Lynch, D.E.; Latif, T.; Smith, G.; Byriel, K.A.; Kennard, C.H.L. J. Chem. Crystallogr. 1997, 27, 567.
Lynch, D.E.; Latif, T.; Smith, G.; Byriel, K.A.; Kennard, C.H.L.; Parsons, S. Aust. J. Chem. 1998, 51, 403.
a.Lumme, P.; Kekarainen, P.; Knuuttila, H.; Kurkirinne, T.; Latvala, M.; Ronkonharju, L.; Salonen, S. Finn. Chem. Lett. 1981, 25, 1981. b.Lumme, P.O.; Knuuttila H.; Lindell, E. Acta Crystallogr. 1996, C52, 51.
O'Reilly, E.J.; Smith, G.; Kennard, C.H.L.; White, A.H. Aust. J. Chem. 1983, 36, 183.
O'Reilly, E.J.; Smith, G.; Kennard, C.H.L. Inorg. Chim. Acta 1984, 90, 63.
Kennard, C.H.L.; Stewart, S.W.; O'Reilly, E.J.; Smith, G.; White, A.H. Polyhedron 1985, 4, 697.
Mak, T.C.W.; Kennard, C.H.L.; Smith, G.; O'Reilly, E.J.; Sagatys, D.S.; Fulwood, J.C. Polyhedron 1987, 6, 855.
Pon, G.; Willett, R.D.; Prince, B.A.; Robinson, W.T.; Turnbull, M.M. Inorg. Chim. Acta 1997, 255, 325.
Albada, G.A. van; Quiroz-Castro, M.E.; Mutikainen, I.; Turpeinen, U.; Reedijk, J. Inorg. Chim. Acta 2000, 298, 74.
Smith, G.; O'Reilly, E.J.; Kennard, C.H.L.; White, A.H. J. Chem. Soc. Dalton Trans. 1985, 243.
Smith, G.; Kennard, C.H.L.; Byriel, K.A. Polyhedron 1991, 10, 873.
Smith, G.; O'Reilly, E.J.; Carrell, H.L.; Carrell, C.J.; Kennard, C.H.L. Polyhedron 1996, 15, 1995.
Zanchini, C.; Willett, R.D. Inorg. Chem. 1990, 29, 3027.
Manfredini, T.; Pellacani, G.C.; Bonamartini-Corradi, A.; Battaglia, L.P.; Guarini, G.G.T.; Giusti, J.G.; Pon, G.; Willett, R.D.; West, D.X. Inorg. Chem. 1990, 29, 2221.
Albada, G.A. van; Mutikainen, I.; Turpeinen, U.; Reedijk, J. Eur. J. Inorg. Chem. 1998, 547.
Albada, G.A. van; Koningsbruggen, P.J. van; Mutikainen, I.; Turpeinen, U.; Reedijk, J. Eur. J. Inorg. Chem. 1999, 2269.
Spek, A.L. J. Appl. Cryst. 1988, 21, 578.
Beurskens, P.Y.; Admiraal, G.; Beurskens, G.; Bosman, W.P.; Garcia-Granda, S.; Gould, R.O.; Smits, J.M.M.; Smykalla, C. TheDIRDIF program system. Technical report of the Crystallography Laboratory. University of Nijmegen, The Netherlands, 1996.
Sheldrick, G.M., SHELXL96, Program for Crystal Structure Refinement. University of Gö ttingen: Germany, 1996.
Hathaway, B.J. In Comprehensive Coordination Chemistry Vol. 5; Wilkinson, G.; Gill, R.D.; McCleverty, J.A., Eds.; Pergamon Press: Oxford 1987.
West D.X.; Palaniandavar, M. Inorg. Chim.Acta 1983, 77, L97.
McWhinnie, W. J. Chem. Soc. Part III 1964, 2959.
Yamashita, M.; Ito, H.; Ito, T. Inorg. Chem. 1983, 22, 2102.
Kida, S.; Nishida, Y.; Sakamoto, M. Bull. Chem. Soc. of Japan 1973, 46, 2428.
Albada, G.A. van; Smeets, W.J.J.; Spek, A.L.; Reedijk, J. Inorg. Chim. Acta 1997, 260, 151.
Albada, G.A. van; Smeets, W.J.J.; Spek, A.L.; Reedijk, J. J. Chem. Cryst. 1998, 28, 427.
Lawrance, G.A.; Skelton, B.W.; White, A.H.; Comba, P. Aust. J. Chem. 1986, 39, 1101.
Comba, P.; Curtis, N.F.; Lawrance, G.A.; Sargeson, A.M.; Skelton, B.W.; White, A.H. Inorg. Chem. 1986, 25, 4260.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
van Albada, G.A., Smeets, W.J., Spek, A.L. et al. Crystal structure and spectroscopy of a most unusual Cu(II) coordination compound containing an infinite chain of lattice ligand interwoven with stacked sheets of neutral bis(2-aminopyrimidine-bridged) copper ions: X-ray structure of [Cu(ampym)2(H2O)2(CF3SO3)2](ampym)2(ampym)2. Journal of Chemical Crystallography 30, 11–16 (2000). https://doi.org/10.1023/A:1009533729785
Issue Date:
DOI: https://doi.org/10.1023/A:1009533729785