Abstract
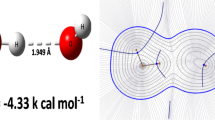
In order to design new ligands for protein-binding sites of unknownstructure, it would be useful to predict the likely sites of hydrogenbonding of an unknown protein fragment to a known molecule. The positions ofmaxima and minima in the electrostatic potential at appropriate distancesfrom the van der Waals surface were calculated for various small molecules,nucleic acid bases, peptide units and amino acid side chains containinggroups which can form the biologically important N-H...O=C andN-H...N hydrogen bonds. Their ability to predict the positions of H andO/N in hydrogen bonded complexes, as predicted by optimising theelectrostatic interactions of pairs of such molecules constrained by themolecular shapes, was assessed. It is shown that extrema in theelectrostatic potential around the isolated molecules give worthwhilepredictions for the locations of hydrogen bonding partners. For moleculesbound by a single N-H...O=C hydrogen bond, the electrostatic maximumassociated with the H is usually less than 1 Å from an acceptor atom,while a C=O electrostatic minimum is generally less than 1.5 Å fromthe hydrogen bond proton. However, a significant number of hydrogen bondsform to the opposite lone pair from the electrostatic minimum, in which casethe separation is up to 3.3 Å. This reflects the broad electrostaticpotential well around a carbonyl oxygen between the lone pair directions.The model predicts when neighbouring atoms drastically change the hydrogenbonding characteristics of an N-H or C=O group. Although the geometries ofhydrogen bonded complexes are influenced by the other van der Waals contactsbetween the molecules, particularly multiple hydrogen bonds, theseinfluences are constant when considering hydrogen bonding to a specificuncharacterised binding site. Hence, the consideration of stericallyaccessible electrostatic extrema will be useful in the design of newligands.
Similar content being viewed by others
References
Dean, P.M., Molecular Foundations of Drug–Receptor Interaction, Cambridge University Press, Cambridge, U.K., 1987.
Jeffrey, G.A. and Saenger, W., Hydrogen Bonding in Biological Structures, Springer, Berlin, Germany, 1991.
Mills, J.E.J. and Dean, P.M., J. Comput.-Aided Mol. Design, 10 (1996) 607.
Taylor, R., Kennard, O. and Versichel, W., J. Am. Chem. Soc., 105 (1983) 5761.
Baker, E.N. and Hubbard, R.E., Prog. Biophys. Mol. Biol., 44 (1983) 5761.
Ippolito, J.A., Alexander, R.S. and Christianson, D.W., J. Mol. Biol., 215 (1990) 457.
Náray-Szabó, G. and Ferenczy, G.G., Chem. Rev., 95 (1995) 829.
Murray, J.S., Lane, P. and Politzer, P., J. Mol. Struct. (THEOCHEM), 209 (1990) 163.
Murray, J.S., Ranganathan, S. and Politzer, P., J. Org. Chem., 56 (1991) 3734.
Murray, J.S. and Politzer, P., J. Org. Chem., 56 (1991) 6715.
Kenny, P.W., J. Chem. Soc. Perkin Trans., 2 (1994) 199.
Pauling, L., Proc. Natl. Acad. Sci. USA, 14 (1928) 359.
Stone, A.J., Chem. Phys. Lett., 83 (1981) 233.
Stone, A.J. and Alderton, M., Mol. Phys., 56 (1985) 1047.
Buckingham, A.D. and Fowler, P.W., J. Chem. Phys., 79 (1983) 6426.
Buckingham, A.D. and Fowler, P.W., Can. J. Chem., 63 (1985) 2018.
Mitchell, J.B.O. and Price, S.L., Chem. Phys. Lett., 154 (1989) 267.
Mitchell, J.B.O., Nandi, C.L., Thornton, J.M., Price, S.L., Singh, J. and Snarey, M., J. Chem. Soc. Faraday Trans., 89 (1993) 2619.
Mitchell, J.B.O., Nandi, C.L., McDonald, I.K., Thornton, J.M. and Price, S.L., J. Mol. Biol., 239 (1994) 315.
Flanagan, K., Walshaw, J., Price, S.L. and Goodfellow, J.M., Protein Eng., 8 (1995) 109.
Price, S.L., Lo Celso, F., Treichel, J.A., Goodfellow, J.M. and Umrania, Y., J. Chem. Soc. Faraday Trans., 89 (1993) 3407.
Hobza, P. and Sandorfy, C., J. Am. Chem. Soc., 109 (1987) 1302.
Hurst, G.J.B., Fowler, P.W., Stone, A.J. and Buckingham, A.D., Int. J. Quantum Chem., 29 (1986) 1223.
Apaya, R.P., Lucchese, B., Price, S.L. and Vinter, J.G., J. Comput.-Aided Mol. Design, 9 (1995) 33.
Van der Wenden, E.M., Price, S.L., Apaya, R.P., IJzerman, A.P. and Soudijn, W., J. Comput.-Aided Mol. Design, 9 (1995) 44.
Frau, J. and Price, S.L., J. Comput.-Aided Mol. Design, 10 (1996) 107.
Faerman, C.H. and Price, S.L., J. Am. Chem. Soc., 112 (1990) 4915.
INSIGHT II, v. 2.3.0, Biosym Technologies, San Diego, CA, U.S.A., 1993.
Dauber-Osguthorpe, P., Roberts, V.A., Osguthorpe, D.J., Wolff, J., Genest, M. and Hagler, A.T., Proteins Struct. Funct. Genet., 4 (1988) 31.
CADPAC5: The Cambridge Analytical Derivatives Package, Issue 5.0, 1992. A suite of quantum chemistry programs developed by R.D. Amos with contributions from I.L. Alberts, J.S. Andrews, S.M. Colwell, N.C. Handy, D. Jayatilaka, P.J. Knowles, R. Kobayashi, N. Koga, K.E. Laidig, P.E. Malsen, C.W. Murray, J.E. Rice, J. Sanz, D. Simandiras, A.J. Stone and M.-D. Su.
Binkley, J.S., Pople, J.A. and Hehre, W.J., J. Am. Chem. Soc., 102 (1980) 939.
Stone, A.J., Popelier, P.L.A. and Wales, D.J., ORIENT: A program for calculating the electrostatic interactions between molecules, v. 3.0, University of Cambridge, Cambridge, U.K., 1994.
Spackman, M.A., J. Chem. Phys., 85 (1986) 6587.
Price, S.L. and Stone, A.J., J. Chem. Soc. Faraday. Trans., 88 (1992) 1755.
Taylor, R. and Kennard, O., J. Am. Chem. Soc., 104 (1982) 5063.
Desiraju, G.R., Acc. Chem. Res., 24 (1991) 290.
Mitchell, J.B.O. and Price, S.L., J. Comput. Chem., 11 (1990) 1217.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Apaya, R.P., Bondí, M. & Price, S.L. The orientation of N-H...O=C and N-H...N hydrogen bonds in biological systems: How good is a point charge as a model for a hydrogen bonding atom?. J Comput Aided Mol Des 11, 479–490 (1997). https://doi.org/10.1023/A:1007923124523
Issue Date:
DOI: https://doi.org/10.1023/A:1007923124523