Abstract
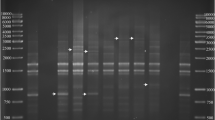
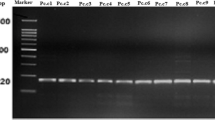
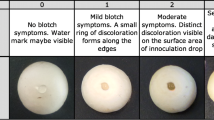
Black rot, caused by Xanthomonas campestris pv. campestris (Xcc), is a major disease constraint to cabbage production by smallholder farmers in Africa. Variability exists within the pathogen, and yet differentiation of Xcc strains from other closely-related xanthomonads attacking crucifers is often difficult. The Biolog system, fatty acid methyl ester analysis using microbial identification system (MIS), rep-PCR and pathogenicity tests were used to identify and characterise Xcc strains from Tanzania. Great diversity was observed among Xcc strains in their Biolog and rep-PCR profiles. Specific rep-PCR genomic fingerprints were linked to some geographical areas in the country. Most of the Xcc strains were clustered in two groups based on their fatty acid profiles and symptom expression in cabbage although some deviant strains were found. Each of the methods allowed a degree of identification from species, pathovar to the strain level. Biolog and MIS identified all Xcc strains at least to the genus level. Additionally, Biolog identified 47% of Xcc strains to the pathovar and 43% to strain level, whereas MIS identified 43% of the strains to pathovar level. In the absence of a database, the utility of rep-PCR for routine diagnosis of strains was limited, although the procedure was good for delineation of Xcc to the strain level. These findings indicate the existence of Xcc strains in Tanzania that are distinct from those included in Biolog and MIS databases. The limitations noticed warrant continued improvement of databases and inclusion of pathogenicity testing, using universally susceptible cultivars, as an integral part of strain identification.
Similar content being viewed by others
References
Alvarez A (2000) Black rot of crucifers. In: Slusarenko A, Fraser RSS and van Loon LC (eds) Mechanisms of Resistance to Plant Diseases (pp 21-52) Kluwer Academic Publishers, Dordrecht
Alvarez A, Benedict AA, Mizumoto CY, Hunter JE and Gabriel DW (1994) Serological, pathological, and genetic diversity among strains of Xanthomonas campestris infecting crucifers. Phytopathology 81: 1449-1457
Anonymous (1985) Black rot. Xanthomonas campestris pv. campestris (Pam) Dow. (Xcc) Crucifer Genetics Cooperatives (pp 1-7) University of Wisconsin, Madison, United States of America
Black LL and Machmud M (1983) Xanthomonas leaf spot of crucifers. In: 4th International Congress of Plant Pathology (p 126) Melbourne, Australia
Black R, Abubakar ZM and Seal S (2000) Opportunities and constraints in the adaptation of technology for the diagnosis of bacterial plant diseases-experience from Tanzania. EPPO Bulletin 30: 367-374
Chen J, Roberts PD and Gabriel DW (1994) Effects of a virulence locus from Xanthomonas campestris 528T on pathovar status and ability to elicit blight symptoms on crucifers. Phytopathology 84: 1458-1464
Cook AA, Walker JC and Larson RH (1952) Studies on the disease cycle of black rot of crucifers. Phytopathology 42: 162-167
Day LA, Feldman S, Minja R and Wien C (1992) Vegetable production in Tanzania, a Four case study in Arumeru District. Special Survey Report (pp 1-46) Cornell University, United States of America and Sokoine University of Agriculture, Tanzania
de Bruijn FJ, Rademaker J, Schneider M, Rossbach U and Louws FJ (1996) Rep-PCR Genomic fingerprinting of plant-associates bacteria and computer-assisted phylogenetic analyses. In: Stacey G, Mullin B and Greshoff P (eds) Biology of Plant-Microbe Interaction: Proceedings of the 8th International Congress of Molecular Plant-Microbe Interactions (pp 497-502) American Phytopathological Society Press
Felsenstein J (1993) PHYLIP (Phylogeny Inference Package) version 3.5c. Distributed by author. Department of Genetics, University of Washington, Seattle, United States of America
Franken AAJM (1992) Immunofluorescence microscopy and dilution-plating for the detection of Xanthomonas campestris pv. campestris in crucifer seeds-methods to determine seed health and seed infection. Ph.D. Thesis. Wageningen Agricultural University, 164 pp
Ignatov A, Kuginuki Y and Hida K (1998) Race-specific reaction of resistance to black rot in Brassica oleracea. European Journal of Plant Pathology 104: 821-827
Jones JB, Chase AR and Harris GK (1993) Evaluation of the Biolog GN MicroPlate system for identification of some plantpathogenic bacteria. Plant Disease 77: 553-558
Leach JE, Rhoads ML, Vera Cruz CM, White FF, Mew TW and Leung H (1992) Assessment of genetic diversity and population structure of Xanthomonas oryzae pv. oryzae with repetitive DNA element. Applied and Environmental Microbiology 58: 2188-2195
Lelliot RA and Stead DE (1987) Methods for the Diagnosis of Bacterial Diseases of Plants. Blackwell Scientific Publications, 216 pp.
Louws FJ, Fulbright DW, Stevens CT and de Bruijn FJ (1994) Specific genomic fingerprints of phytogenic Xanthomonas and Pseudomonas pathovars and strains generated with repetitive sequences and PCR. Applied Environmental Microbiology 60: 2286-2295
Louws FJ, Fulbright DW, Stevens CT and de Bruijn FJ (1995) Differentiation of genomic structure by rep-PCR fingerprinting to rapidly classify Xanthomonas campestris pv. vesicatoria. Phytopathology 85: 528-536
Massomo SMS (2002) Black rot of cabbage in Tanzania: Characterisation of Xanthomonas campestris pv. campestris and disease management strategies. Ph.D. Thesis. The Royal Veterinary and Agricultural University, Copenhagen, Denmark, 215pp
Mguni CM (1996) Bacterial black rot (Xanthomonas campestris pv. campestris) of vegetable brassicas in Zimbabwe. Ph.D. Thesis. The Royal Veterinary and Agricultural University, Copenhagen, Denmark, 144pp
Mguni CM, Mortensen CN, Keswani CL and Hockenhull J (1999) Detection of the black rot pathogen (Xanthomonas campestris pv. campestris) and other xanthomonads in Zimbabwean and imported Brassica seed. Seed Science and Technology 27: 447-454
Minsavage GV and Schaad NW (1983) Characterisation of membrane proteins of Xanthomonas campestris pv. campestris. Phytopathology 73: 747-754
Onsando JM (1992) Black rot of Crucifers. In: Chaube HS, Singh US, Mukhopadyay AN and Kumar J (eds) Plant Diseases of International Importance. Diseases of Vegetables and Oil Seed Crops (pp 243-252) Prentice Hall, Englewood Cliffs, New Jersey, United States of America
Pierce L, Schroth MN and McCain AH (1990) Viscosity test for preliminary identification of strains of Xanthomonas campestris. Plant Disease 74: 646-647
Sahin F and Miller SA (1997) A new pathotype of Xanthomonas campestris pv. armoraciae that causes bacterial leaf spot of radish. Plant Disease 81: 1334
Sasser M (1990) Identification of bacteria through fatty acid analysis. In: Klement Z, Rudolph K and Sands DC (eds) Methods in Phytobacteriology (pp 199-204) Akademiai Kiadó, Budapest
Schaad NW (1989) Detection of Xanthomonas campestris pv. campestris in crucifers. In: Saettler AW, Schaad NW and Roth DA (eds) Detection of Bacteria in Seed and Other Planting Material (pp 68-75) The American Phytopathological Society, St Paul, United States of America
Schaad NW and Kendrick R (1975) A qualitative method of detecting Xanthomonas campestris pv. campestris in crucifer seed. Phytopathology 65: 1034-1036
Schaad NW and Alvarez A (1983) Xanthomonas campestris pv. campestris: Cause of black rot of crucifers. In: Swing JG and Civerolo EL (eds) Xanthomonas (pp 51-56) Chapman and Hall, London, United Kingdom
Schaad NW and Franken AAJM (1990) Xanthomonas campestris pv. campestris. International Seed Testing Association Handbook on Seed Health Testing. Working sheet number 50, 2nd edn. International Seed Testing Association, Zurich
Schaad NW, Sitterley WR and Mumaydan H (1980) Relation of incidence of seedborne Xanthomonas campestris to black rot of crucifers. Plant Disease 64: 91-92
Scortichini M, Marchesi U and Prospero PD (2001) Genetic diversity of Xanthomonas arboricola pv. juglandis (synonyms: X. campestris pv. juglandis; X. juglandis pv. juglandis) strains from different geographical areas shown by repetitive polymerase chain reaction genomic fingerprinting. Journal of Phytopathology 149: 325-332
Sharples GJ and Lloyd RG (1990) A novel repeated DNA sequence located in the intergenic regions of bacterial chromosomes. Nucleic Acids Research 18: 6503-6508
Stead DE, Sellwood JE, Wilson J and Viney I (1992) Evaluation of a commercial microbial identification system based on fatty acid profiles for rapid, accurate identification of plant pathogenic bacteria. Journal of Applied Bacteriology 72: 315-321
Tamura K, Takikawa Y, Tsuyumu S and Goto M (1994) Bacterial spot of crucifers caused by Xanthomonas campestris pv. raphani. Annals of Phytopathological Society of Japan 60: 281-287
Taylor JD, Conway J, Roberts SJ, Astley D and Vicente JG (2002) Sources and origin of resistance to Xanthomonas campestris pv. campestris in Brassica genomes. Phytopathology 92: 105-111
Thaveechai N and Schaad NW (1986) Serological and electrophoretic analysis of membrane protein extract of Xanthomonas campestris pv. campestris from Thailand. Phytopathology 76: 139-147
Vauterin L, Hoste B, Kersters K and Swings J (1995) Reclassi-fication of Xanthomonas. International Journal of Systematic Bacteriology 45: 472-489
Verdier V, Boher B, Maraite H and Geiger J (1994) Pathological and molecular characterization of Xanthomonas campestris strains causing diseases of cassava. Applied Environmental Microbiology 60: 4478-4486
Versalovic J, Koeuth T and Lupski JR (1991) Distribution of repetitive DNA sequences in eubacteria and application to fingerprinting of bacterial genomes. Nucleic Acids Research 19: 6823-6831
Versalovic J, Schneider M, de Bruijn FJ and Lupski JR (1994) Genomic fingerprinting of bacteria using repetitive sequencebased polymerase chain reaction. Methods in Molecular and Cellular Biology 5: 25-40
Vicente JG, Conway J, Roberts SJ and Taylor JD (1998) Identification of Xanthomonas campestris pv. campestris races and related pathovars. (Poster) Horticulture Research International, Wellesbourne, Warwick CV35, United Kingdom
Vicente JG, Conway J, Roberts SJ and Taylor JD (2001) Identification and origin of Xanthomonas campestris pv. campestris races and related pathovars. Phytopathology 91: 492-499
Weller SA, Aspin A and Stead DE (2000) Classification and identification of plant-associated bacteria by fatty acid profiling. EPPO Bulletin 30: 375-380
William PH, Staub T and Sutton JC (1972) Inheritance of resistance to black rot. Phytopathology 62: 247-252
Williams PH (1980) Black rot: A continuing threat to world crucifers. Plant Disease 64: 736-745
Wulff EG (2000) The use of antagonistic, endophytic bacteria for controlling Xanthomonas campestris pv. campestris, in Zimbabwe. Ph.D. Thesis. The Royal Veterinary and Agricultural University, Copenhagen, Denmark. p134
Yang P, Vauterin L, Vancanneyt M, Swings JS and Kersters K (1993) Application of fatty acid methyl esters for the taxonomic analysis of the genus Xanthomonas. Systematic Applied Microbiology 16: 47-71
Zhao Y, Damicone JP, Demezas DH and Bender CL (2000) Bacterial leaf spot of leafy Crucifers in Oklahoma caused by pathovars of Xanthomonas campestris. Plant Disease 84: 1008-1014
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Massomo, S.M., Nielsen, H., Mabagala, R.B. et al. Identification and Characterisation of Xanthomonas campestris pv. campestris Strains from Tanzania by Pathogenicity Tests, Biolog, rep-PCR and Fatty Acid Methyl Ester Analysis. European Journal of Plant Pathology 109, 775–789 (2003). https://doi.org/10.1023/A:1026194402701
Issue Date:
DOI: https://doi.org/10.1023/A:1026194402701