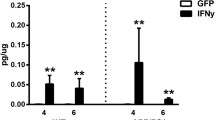
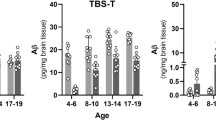
Abstract
Human C1q when injected directly into hippocampus and cortex of doubly transgenic APP+PS1 mice results in the increase of Congo red-positive fibrillar deposits. Although there was no significant change in overall area stained for Aβ total, qualitatively it appeared that there was less diffuse Aβ in C1q-treated mice versus vehicle. There was no apparent change in astroglial or microglial activation caused by injection of C1q with respect to vehicle injections. These effects of C1q were only found in 50% BUB/BnJ mice, a strain with higher serum complement activity than other mouse lines. These in vivo data were consistent with the effects of C1q to increase fibrillogenesis of Aβ in vitro. In conclusion, complement protein C1q, believed to be involved in the pathogenesis of Alzheimer's disease in humans, can cause increased fibrillogenesis in the APP+PS1 mouse model of amyloid deposition.
Similar content being viewed by others
REFERENCES
Eikelenboom, P. and Stam, F. C. 1982. Immunoglobulins and complement factors in senile plaques: An immunoperoxidase study. Acta Neuropathol. (Berl). 57:239–242.
Webster, S. D., Yang, A. J., Margol, L., Garzon-Rodriguez, W., Glabe, C. G., and Tenner, A. J. 2000. Complement component C1q modulates the phagocytosis of Abeta by microglia. Exp Neurol. 161:127–138.
Webster, S., Bonnell, B., and Rogers, J. 1997. Charge-based binding of complement component C1q to the Alzheimer amyloid beta-peptide. Am. J. Pathol. 150:1531–1536.
Rogers, J., Cooper, N. R., Webster, S., Schultz, J., McGeer, P. L., Styren, S. D., Civin, W. H., Brachova, L., Bradt, B., and Ward, P. 1992. Complement activation by beta-amyloid in Alzheimer disease. Proc. Natl. Acad. Sci. U.S.A. 89:10016–10020.
Jiang, H., Burdick, D., Glabe, C. G., Cotman, C. W., and Tenner, A. J. 1994. Beta-amyloid activates complement by binding to a specific region of the collagen-like domain of the C1q A chain. J. Immunol. 152:5050–5059.
Watson, M. D., Roher, A. E., Kim, K. S., Spiegel, K., and Emmerling, M. R. 1997. Complement interactions with amyloid-beta1-42: A nidus for inflammation in AD brains. Amyloid: Int. J. Exp. Clin. Invest. 4:147–156.
Selkoe, D. J. 2001. Alzheimer's disease: Genes, proteins, and therapy. Physiol. Rev. 81:741–766.
Holcomb, L., Gordon, M. N., McGowan, E., Yu, X., Benkovic, S., Jantzen, P., Wright, K., Saad, I., Mueller, R., Morgan, D., Sanders, S., Zehr, C., O'Campo, K., Hardy, J., Prada, C. M., Eckman, C., Younkin, S., Hsiao, K., and Duff, K. 1998. Accelerated Alzheimer-type phenotype in transgenic mice carrying both mutant amyloid precursor protein and presenilin 1 transgenes. Nat. Med. 4:97–100.
Gordon, M. N., King, D. L., Diamond, D. M., Jantzen, P. T., Boyett, K. V., Hope, C. E., Hatcher, J. M., DiCarlo, G., Gottschall, W. P., Morgan, D., and Arendash, G. W. 2001. Correlation between cognitive deficits and Abeta deposits in transgenic APP+PS1 mice. Neurobiol. Aging 22:377–385.
Matsuoka, Y., Picciano, M., Malester, B., LaFrancois, J., Zehr, C., Daeschner, J. M., Olschowka, J. A., Fonseca, M. I., O'Banion, M. K., Tenner, A. J., Lemere, C. A., and Duff, K. 2001. Inflammatory responses to amyloidosis in a transgenic mouse model of Alzheimer's disease. Am. J. Pathol. 158:1345–1354.
Games, D., Adams, D., Alessandrini, R., Barbour, R., Berthelette, P., Blackwell, C., Carr, T., Clemens, J., Donaldson, T., and Gillespie, F. 1995. Alzheimer-type neuropathology in transgenic mice overexpressing V717F beta-amyloid precursor protein. Nature 373:523–527.
Irizarry, M. C., McNamara, M., Fedorchak, K., Hsiao, K., and Hyman, B. T. 1997. APPSw transgenic mice develop age-related A beta deposits and neuropil abnormalities, but no neuronal loss in CA1. J. Neurophathol. Exp. Neurol. 56:965–973.
Irizarry, M. C., Soriano, F., McNamara, M., Page, K. J., Schenk, D., Games, D., and Hyman, B. T. 1997. Abeta deposition is associated with neuropil changes, but not with overt neuronal loss in the human amyloid precursor protein V717F (PDAPP) transgenic mouse. J. Neurosci. 17:7053–7059.
Lorenzo, A. and Yankner, B. A. 1994. Beta-amyloid neurotoxicity requires fibril formation and is inhibited by Congo Red. Proc. Natl. Acad. Sci. U.S.A. 91:12243–12247.
Howlett, D. R., Jennings, K. H., Lee, D. C., Clark, M. S., Brown, F., Wetzel, R., Wood, S. J., Camilleri, P., and Roberts, G. W. 1995. Aggregation state and neurotoxic properties of Alzheimer beta-amyloid peptide. Neurodegeneration. 4:23–32.
Simmons, L. K., May, P. C., Tomaselli, K. J., Rydel, R. E., Fuson, K. S., Brigham, E. F., Wright, S., Lieberburg, I., Becker, G. W., and Brems, D. N. 1994. Secondary structure of amyloid beta peptide correlates with neurotoxic activity in vitro. Mol. Pharmacol. 45:373–379.
Mrak, R. E., Sheng, J. G., and Griffin, W. S. 1995. Glial cytokines in Alzheimer's disease: Review and pathogenic implications. Hum. Pathol. 26:816–823.
Wright, K. L., Hsiao, K., Duff, K., Morgan, D. G., and Gordon, M. N. 1998. Inflammation in PSAPP Alzheimer's transgenic mice bred into complement sufficient, BUB/BnJ, mouse strain. Soc. Neurosci. Abstr. 24:2430.
Ong, G. L., Baker, A. E., and Mattes, M. J. 1992. Analysis of high complement levels in Mus hortulanus and BUB mice. J. Immunol. Methods. 154:37–45.
Ong, G. L. and Mattes, M. J. 1989. Mouse strains with typical mammalian levels of complement activity. J. Immunol. Methods. 125:147–158.
Webster, S. D., Tenner, A. J., Poulos, T. L., and Cribbs, D. H. 1999. The mouse C1q A-chain sequence alters beta-amyloid-induced complement activation. Neurobiol. Aging. 20:297–304.
Hsiao, K., Chapman, P., Nilsen, S., Eckman, C., Harigaya, Y., Younkin, S., Yang, F., and Cole, G. 1996. Correlative memory deficits, Abeta elevation, and amyloid plaques in transgenic mice. Science. 274:99–102.
Duff, K., Eckman, C., Zehr, C., Yu, X., Prada, C. M., Perez-tur, J., Hutton, M., Buee, L., Harigaya, Y., Yager, D., Morgan, D., Gordon, M. N., Holcomb, L., Refolo, L., Zenk, B., Hardy, J., and Younkin, S. 1996. Increased amyloid-beta42(43) in brains of mice expressing mutant presenilin 1. Nature. 383:710–713.
Gordon, M. N., Schreier, W. A., Ou, X., Holcomb, L. A., and Morgan, D. G. 1997. Exaggerated astrocyte reactivity after nigrostriatal deafferentation in the aged rat. J. Comp. Neurol. 388:106–119.
Gordon, M. N., Holcomb, L. A., Jantzen, P. T., DiCarlo, G., Wilcock, D., Boyett, K. W., Connor, K., Melachrino, J., O'Callaghan, J. P., and Morgan, D. 2002. Time course of the development of Alzheimer-like pathology in the doubly-transgenic PS1+APP mouse. Exp. Neurol. 173:183–195.
Webster, S., Glabe, C., and Rogers, J. 1995. Multivalent binding of complement protein C1Q to the amyloid beta-peptide (A beta) promotes the nucleation phase of A beta aggregation. Biochem. Biophys. Res. Commun. 217:869–875.
Webster, S. D., Park, M., Fonseca, M. I., and Tenner, A. J. 2000. Structural and functional evidence for microglial expression of C1qR(P), the C1q receptor that enhances phagocytosis. J. Leukoc. Biol. 67:109–116.
Rozovsky, I., Morgan, T. E., Willoughby, D. A., Dugichi-Djordjevich, M. M., Pasinetti, G. M., Johnson, S. A., and Finch, C. E. 1994. Selective expression of clusterin (SGP-2) and complement C1qB and C4 during responses to neurotoxins in vivo and in vitro. Neuroscience 62:741–758.
Pasinetti, G. M., Johnson, S. A., Rozovsky, I., Lampert-Etchells, M., Morgan, D. G., Gordon, M. N., Morgan, T. E., Willoughby, D., and Finch, C. E. 1992. Complement C1qB and C4 mRNAs responses to lesioning in rat brain. Exp. Neurol. 118:117–125.
Goldsmith, S. K., Wals, P., Rozovsky, I., Morgan, T. E., and Finch, C. E. 1997. Kainic acid and decorticating lesions stimulate the synthesis of C1q protein in adult rat brain. J. Neurochem. 68:2046–2052.
Wright, K. L., Morgan, D. G., and Gordon, M. N. 1997. Complement immunoreactivity in murine brain after kainic acid treatment. Soc. Neurosci. Abstr. 23:1503.
Ferraro, T. N., Golden, G. T., Smith, G. G., and Berrettini, W. H. 1995. Differential susceptibility to seizures induced by systemic kainic acid treatment in mature DBA/2J and C57BL/6J mice. Epilepsia 36:301–307.
Grilli, M., Diodato, E., Lozza, G., Brusa, R., Casarini, M., Uberti, D., Rozmahel, R., Westaway, D., George-Hyslop, P., Memo, M., and Ongini, E. 2000. Presenilin-1 regulates the neuronal threshold to excitotoxicity both physiologically and pathologically. Proc. Natl. Acad. Sci. U.S.A. 97:12822–12827.
Weinbauer, G. F., Galhotra, M. M., and Nieschlag, E. 1985. Focal testicular destruction following intratesticular injection of glycerol in rats. Int. J. Androl. 8:365–375.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Boyett, K.W., DiCarlo, G., Jantzen, P.T. et al. Increased Fibrillar β-Amyloid in Response to Human C1q Injections into Hippocampus and Cortex of APP+PS1 Transgenic Mice. Neurochem Res 28, 83–93 (2003). https://doi.org/10.1023/A:1021600212829
Issue Date:
DOI: https://doi.org/10.1023/A:1021600212829