Abstract
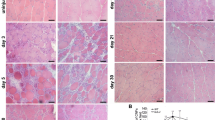
The effects of the human BCL-xL and ACR-1genes on dystrophin expression in cross-striated muscle fibers (CSMF) and on CSMF viability were studied in mdx mice after ballistic cotransfection with the human dystrophin minigene. In control mice, the proportion of dystrophin-positive (D(+)) and dying CSMF were 2.1 ± 0.1 and 2.1 ± 0.3%, respectively. Introduction of the dystrophin minigene (20 μg of the pSG5dys plasmid) increased the proportions of D(+) and dying CSMF to 5.6 ± 1.4% and 4.5 ± 0.9%, respectively. When pSG5dys was introduced along with the pSFFV-Neo plasmid carrying the BCL-xL gene (10 μg of each plasmid per shot), the death of CSMF decreased to 3.7 ± 1% and the proportion of D(+) CSMF significantly (P < 0.05) increased to 12.2 ± 2.2%. Cotransfection with the dystrophin minigene and the BCL-xL gene at 20 μg of each plasmid per shot did not stimulate generation of D(+) CSMF, but did reduce the CSMF death to 1.5 ± 0.3%. Introduction of pSG5dys along with the pRc-CMV-10.1 plasmid containing the ACR-1 gene (10 μg of each plasmid per shot) reduced the proportion of D(+) CSMF to 1.1 ± 0.5% and significantly reduced the proportion of dying CSMF to 0.9 ± 0.3% as compared with the proportions observed in intact mice or in mice subjected to transfection with pSG5dys. Introduction of the pSG5dys plasmid substantially reduced the proportion of CSMF with peripheral nuclei, suggesting disturbed CSMF differentiation. After cotransfection with the human dystrophin minigene, the BCL-xL and ACR-1 genes did not affect the extent of CSMF differentiation as compared with that observed in the case of the dystrophin minigene alone. Thus, ballistic transfection of mdx mice with the human dystrophin gene used along with the BCL-xLor ACR-1 gene was shown to suppress the death of muscle fibers and to expedite dystrophin synthesis and cell differentiation.
Similar content being viewed by others
REFERENCES
Baranov, V.S., Baranov, A.N., and Zelenin, A.V., Current State and Prospects of Gene Therapy of Duchenne Muscular Dystrophy in the World and in Russia, Genetika (Moscow), 2001, vol. 37, no. 8, pp. 1046–1053.
Zelenin, A.V., Kaigorodov, V.A., and Prasolov, V.S., Gene Therapy Today and Tomorrow, Mol. Biol. (Moscow), 1998, vol. 32, pp. 219–228.
Mikhailov, V.M., Zelenin, A.V., Stein, G.I., et al., Differentiation of Muscle Fibers of Mdx Mice Subjected to Ballistic Transfection with the Human Dystrophin Gene cDNA, Tsitologiya, 1998, vol. 40, pp. 394–400.
Mikhailov, V.M., Kazakov, V.I., Komarov, V.A., et al., Apoptosis and DNA Degradation in Cardiomyocytes of Mdx and C57Bl Mice, Tsitologiya, 1998, vol. 40, pp. 401–406.
Lefaucheur, J.P., Pastoret, C., and Sebille, A., A Phenotype of Dystrophiopathy in Old mdx Mice, Anat. Rec., 1995, vol. 242, pp. 70–76.
Sandri, M., Podhorska-Okolow, M., Geromel, V., et al., Exercise Induces Myonuclear Ubiquitination and Apoptosis in Dystrophin-Deficient Muscle of Mice, J. Neuropathol. Exp. Neurol., 1997, vol. 56, pp. 45–57.
Matsuda, R., Nishikawa, A., and Tanaka, H., Visualization of Dystrophic Muscle Fibers in Mdx Mice by Vital Staining with Evans Blue: Evidence of Apoptosis in Dystrophin-Deficient Muscle, J. Biochem. (Tokyo), 1995, vol. 118, pp. 959–964.
Dunckley, M.G., Piper, T.A., and Dickson, G., Toward a Gene Therapy for Duchenne Muscular Dystrophy, Mental Retardation and Developmental Disabilities Reviews, 1995, vol. 1, pp. 71–78.
Baranov, A., Glazkov, P., Kiselev, A., et al., Local and Distant Transfection of Mdx Muscle Fibers with Dystrophin and lacZ Genes Delivered in vivo by Synthetic Microspheres, Gene Ther., 1999, vol. 6, pp. 1406–1414.
Boise, L.H., Gonzalez-Garcia, M., Pastema, C.E., et al., BCL-xL, a BCL-2 Related Gene That Functions as a Dominant Regulator of Apoptotic Cell Death, Cell (Cambridge, Mass.), 1993, vol. 74, pp. 597–608.
Kane, D., Sarafion, T., Anton, R., et al., BCL-2 Inhibition of Neuronal Death: Decreased Generation of Reactive Oxygen Species, Science, 1993, vol. 262, pp. 1274–1277.
Boise, L.H. and Thompson, C.B., BCL-X Can Inhibit Apoptosis in Cells That Have Undergone FAS-Induced Protease Activation, Proc. Natl. Acad. Sci. USA, 1997, vol. 94, pp. 3759–3764.
Yang, E. and Korsmeyer, S.J., Molecular Thanatopsis: A Discourse on the BCL-2 Family and Cell Death, Blood, 1996, vol. 88, pp. 386–401.
Vander Heiden, M. and Thompson, C., BCL-2 Proteins: Regulators of Apoptosis or of Mitochondrial Homeostasis, Nat. Cell. Biol., 1999, pp. 209–216.
Mikhailov, V.M., Zelenin, A.V., Krutilina, R.I., et al., Ballistic Transfection of Cross-Striated Muscle Fibers of Mdx Mice with the Human BCL-2 and BCL-xL Genes Changes Dystrophin Gene Expression in Muscle Fibers and Prevents Their Death, Tsitologiya, 2001, vol. 43, pp. 365–366.
Kropotov, A.V., Analysis of the Human Proteins Controlling Transcription of Retroposons of the Alu Family, Cand. Sci. (Biol.) Dissertation, St. Petersburg, 1999.
Kropotov, A., Sedova, V., Ivanov, V., et al., A Novel Human DNA-Binding Protein with Sequence Similarity to a Subfamily of Redox Proteins Which Is Able to Repress RNA-Polymerase-III-Driven Transcription of the Alu-Family Retroposons in vitro, Eur. J. Biochem., 1999, vol. 260, pp. 336–346.
Kolesnikov, V.A., Zelenina, I.A., Semenova, M.L., et al., Ballistic Transfection of Mammalian Cells in vivo, Ontogenez (Moscow), 1995, vol. 26, pp. 467–480.
Grinchuk, T.M., Pavlenko, M.A., Pugovkina, N.A., et al., The Effect of Ectopic Overexpression of the Anti-apoptotic BCL-2 Gene on the Level and Character of Karyotype Instability in Cells of CHL V-79 RJK Chinese Hamster, Tsitologiya, 2000, vol. 43, pp. 1004–1014.
Zelenin, A., Kolesnikov, V., Tarasenko, O., et al., Bacterial b-Galactosidase and Human Dystrophin Gene Are Expressed in Mouse Skeletal Muscle Fibers after Ballistic Transfection, FEBS Lett., 1997, vol. 414, pp. 319–322.
Zelenin, A.V., Tarasenko, O.V., Kolesnikov, V.A., et al., Expression of the Human Dystrophin Gene in Skeletal Muscles of Mdx Mice after Ballistic Transfection, Genetika (Moscow), 1998, vol. 34, no. 5, pp. 595–600.
Stein, G.I., Panteleev, V.G., Povarkova, A.V., and Kudryavtsev, B.N., Potentialities of the Videotest Micro-photometric Analyzer for Microphotometric Assays in Cytology, Tsitologiya, 1998, vol. 40, pp. 914–916.
Bornman, L., Rossouw, H., Gericke, G.S., and Polla, B.S., Effect of Iron Deprivation on the Pathology and Stress Expression in Murine X-Linked Muscular Dystrophy, Biochem. Pharmacol., 1998, vol. 56, pp. 751–757.
Wang, B., Li, J., and Xiao, X., Adeno-Associated Virus Vector Carrying Human Minidystrophin Genes Effectively Ameliorates Muscular Dystrophy in Mdx Mouse Model, Proc. Natl. Acad. Sci. USA, 2000, vol. 97, pp. 13714–13719.
Hochenbary, D.M., Oltvai, Z.N., Yin, X.-M., et al., BCL-2 Functions in the Antioxidant Pathway to Prevent Apoptosis, Cell (Cambridge, Mass.), 1993, vol. 75, pp. 241–251.
Brocheriou, V., Hagege, A.A., Oubenaissa, A., et al., Cardiac Functional Improvement by Human BCL-2 Transgene in Mouse Model of Ischemia/Reperfusion Injury, J. Gene Med., 2000, vol. 2, pp. 326–333.
Manna, S.K., Haridas, V., and Aggarwal, B.B., BCL-xL Suppresses Apoptosis and Activation of Nuclear Factor kB, Activation Protein-1, and C-Jun N-Terminal Kinases, J. Interferon Cytokine Res., 2000, vol. 20, pp. 725–735.
Lakics, V., Medvedev, A.E., Okada, S., and Vogel, S.N., Inhibition of LPS-Induced Cytokines by BCL-xL in a Murine Macrophage Cell Line, J. Immunol., 2000, vol. 165, pp. 2729–2737.
Zhou, Y., Kok, K.H., Chun, A.C.S., et al., Mouse Peroxiredoxin V Is Thioredoxin That Inhibits Apoptosis, Biochem. Biophys. Res. Commun., 2000, vol. 268, pp. 921–927.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mikhailov, V.M., Kropotov, A.V., Zelenin, A.V. et al. The BCL-xL and ACR-1 Genes Promote Differentiation and Reduce Apoptosis in Muscle Fibers of mdx Mice. Russian Journal of Genetics 38, 1221–1225 (2002). https://doi.org/10.1023/A:1021134708365
Issue Date:
DOI: https://doi.org/10.1023/A:1021134708365