Abstract
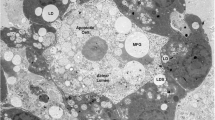
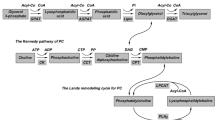
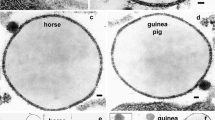
The cream fraction of milk comprises droplets oftriacylglycerol coated with cellular membranes. In thisreview, we discuss how these droplets are formed andsecreted from mammary epithelial cells during lactation. This secretory system is especiallyinteresting because the assembled lipid droplets aresecreted from the cytoplasm enveloped by cellularmembranes. In other cells, such as hepatocytes andenterocytes, lipid is secreted by exocytosis frommembrane-bounded compartments of the secretory pathway.Milk lipids originate as small droplets oftriacylglycerol, synthesized in or on the surfaces ofrough endoplasmic reticulum (ER)4 membranes. Thesedroplets are released into the cytoplasm as microlipiddroplets (MLDs) with a surface coat of protein and polarlipid. MLDs may fuse with each other to form largercytoplasmic lipid droplets (CLDs). Droplets of varyingsize, are transported to the apical cytoplasm by unknownmechanisms and are secreted from the cell coated with anouter bilayer membrane. CLDs may increase in size in all regions of the cell, especially atthe plasma membrane during secretion. Two possiblemechanisms for lipid secretion have been proposed: anapical mechanism, in which lipid droplets are enveloped with apical plasma membrane, and asecretory-vesicle mechanism, in which fat droplets aresurrounded by secretory vesicles in the cytoplasm andare released from the surface by exocytosis fromintracytoplasmic vacuoles. A combination of both mechanisms maybe possible. Following secretion, a fraction of themembrane surrounding the globules may be shed from thedroplets and give rise to membrane fragments in the skimmilk phase.
Similar content being viewed by others
References
S. Patton and R. G. Jensen (1975). Lipid metabolism and membrane functions of the mammary gland. In R. T. Holman (ed.), Progress in the Chemistry of Fats and other Lipids (Vol. XIV) Part 4, Pergamon Press, Oxford, pp. 163-277.
I. H. Mather and T. W. Keenan (1983). Function of endomembranes and the cell surface in the secretion of organic milk constituents. In T. B. Mepham (ed.), Biochemistry of Lactation Elsevier, Amsterdam, pp. 231-283.
I. H. Mather (1987). Proteins of the milk-fat-globule membrane as markers of mammary epithelial cells and apical plasma membrane. In M. C. Neville and C. W. Daniel (eds.), The Mammary Gland, Development, Regulation and Function Plenum Press, New York, pp. 217-267.
T.W. Keenan, I. H. Mather, and D. P. Dylewski (1988). Physical equilibria: Lipid phase. In N. P. Wong (ed.), Fundamentals of Dairy Chemistry, Van Nostrand Reinhold Co., New York, pp. 511-582.
T. W. Keenan and D. P. Dylewski (1994). Intracellular origin of milk lipid globules and the nature and structure of the milk lipid globule membrane. In P. F. Fox (ed.), Advanced Dairy Chemistry-2-Lipids Chapman and Hall, London, pp. 89-130.
T. W. Keenan and S. Patton (1995). The Structure of Milk: Implications for sampling and storage. The Milk Lipid Globule Membrane. In R. G. Jensen (ed.), Handbook of Milk Composition Academic Press, Inc., New York, pp. 5-50.
B. J. Kitchen (1974). A comparison of the properties of membranes isolated from bovine skim milk and cream. Biochim. Biophys. Acta 356: 257-269.
F. B. P. Wooding (1974). Milk fat globule membrane material in skim-milk. J. Dairy Res. 41: 331-337.
M. Anderson, B. E. Brooker, A. T. Andrews, and E. Alichanidis (1975). Membrane material in bovine skim-milk from udder quarters infused with endotoxin and pathogenic organisms. J. Dairy Res. 42: 401-417.
O. T. Oftedal and S. J. Iverson (1995). Comparative analysis of nonhuman milks. Phylogenetic variation in the gross composition of milks. In R. G. Jensen (ed.), Handbook of Milk Composition Academic Press, Inc., New York, pp. 749-789.
W. Bargmann and A. Knoop (1959). Uber die Morphologie der Milchsekretion. Licht-und Elektronenmikroskopische Studien an der Milchdruse der Ratte. Z. Zellforsch. 49: 344-388.
S. M. Cooper and M. R. Grigor (1980). Fatty acid specificities of microsomal acyltransferases esterifying positions-1 and-2 of acylglycerols in mammary glands from lactating rats. Biochem. J. 187: 289-295.
O. Stein and Y. Stein (1967). Lipid synthesis, intracellular transport, and secretion. II. Electron microscopic radioautographic study of the mouse lactating mammary gland. J. Cell Biol. 34: 251-263.
F. Daudet, C. Augeron, and M. Ollivier-Bousquet (1981). Early action of colchicine, ammonium chloride and prolactin, on secretion of milk lipids in the lactating mammary gland. Eur. J. Cell Biol. 24: 197-202.
D. P. Dylewski, C. H. Dapper, H. M. Valivullah, J. T. Deeney, and T. W. Keenan (1984). Morphological and biochemical characterization of possible intracellular precursors of milk lipid globules. Eur. J. Cell Biol. 35: 99-111.
J. T. Deeney, H. M. Valivullah, C. H. Dapper, D. P. Dylewski, and T. W. Keenan (1985). Microlipid droplets in milk secreting mammary epithelial cells: Evidence that they originate from endoplasmic reticulum and are precursors of milk lipid globules. Eur. J. Cell Biol. 38: 16-26.
M. Zaczek and T. W. Keenan (1990). Morphological evidence for an endoplasmic reticulum origin of milk lipid globules obtained using lipid-selective staining procedures. Protoplasma 159: 179-182.
D. Ghosal, N.W. Shappell, and T.W. Keenan (1994). Endoplasmic reticulum lumenal proteins of rat mammary gland. Potential involvement in lipid droplet assembly during lactation. Biochim. Biophys. Acta 1200: 175-181.
T. W. Keenan, D. P. Dylewski, D. Ghosal, and B. H. Keon (1992). Milk lipid globule precursor release from endoplasmic reticulum reconstituted in a cell-free system. Eur. J. Cell Biol. 57: 21-29.
H. M. Valivullah, D. R. Bevan, A. Peat, and T. W. Keenan (1988). Milk lipid globules: Control of their size distribution. Proc. Natl. Acad. Sci. U.S.A. 85: 8775-8779.
R. O. Scow, E. J. Blanchette-Mackie, and L. C. Smith (1980). Transport of lipid across capillary endothelium. Fed. Proc. 39: 2610-2617.
A. Peixoto de Menezes and P. Pinto da Silva (1979). Fat droplet formation in rat lactating mammary gland and mammary carcinomas viewed by freeze-fracture. Lab. Invest. 40: 545-553.
C. A. Long and S. Patton (1978). Formation of intracellular fat droplets: Interrelation of newly synthesized phosphatidyl-choline and triglyceride in milk. J. Dairy Sci. 61: 1392-1399.
H. Mulder and P. Walstra (1974). The Milk Fat Globule, Center for Agricultural Publishing and Documentation, Wageningen, The Netherlands, p. 296.
B. H. Stemberger and S. Patton (1981). Relationships of size, intracellular location, and time required for secretion of milk fat droplets. J. Dairy Sci. 64: 422-426.
B. H. Stemberger, R. M. Walsh, and S. Patton (1984). Morphometric evaluation of lipid droplet associations with secretory vesicles, mitochondria and other components in the lactating cell. Cell Tissue Res. 236: 471-475.
H. M. Valivullah, D. P. Dylewski, and T. W. Keenan (1986). Distribution of terminal transferases of acylglycerol synthesis in cell fractions from lactating mammary gland. Int. J. Bio chem. 18: 799-806.
F. B. P. Wooding (1971). The mechanism of secretion of the milk fat globule. J. Cell Sci. 9: 805-821.
D. Ghosal, D. Ankrapp, and T.W. Keenan (1993). Low molecular mass GTP-binding proteins are secreted from mammary epithelial cells in association with lipid globules. Biochim. Biophys. Acta 1168: 299-306.
H. Jamil, J. K. Dickson, C.-H. Chu, M.W. Lago, J. K. Rinehart, S. A. Biller, R. E. Gregg, and J. R. Wetterau (1995). Microsomal triglyceride transfer protein. Specificity of lipid binding and transport. J. Biol. Chem. 270: 6549-6554.
S. C. Nickerson and T. W. Keenan (1979). Distribution and orientation of microtubules in milk secreting epithelial cells of rat mammary gland. Cell Tissue Res. 202: 303-312.
M. A. Guerin and R. F. Loizzi (1980). Tubulin content and assembly states in guinea pig mammary gland during pregnancy, lactation, and weaning. Proc. Soc. Exp. Biol. Med. 165: 50-54.
S. Patton (1974). Reversible suppression of lactation by colchicine. FEBS Lett. 48: 85-87.
S. Patton, B. H. Stemberger, and C. M. Knudson (1977). The suppression of milk fat globule secretion by colchicine: An effect coupled to inhibition of exocytosis. Biochim. Biophys. Acta 499: 404-410.
S. C. Nickerson, J. J. Smith, and T. W. Keenan (1980). Ultrastructural and biochemical response of rat mammary epithelial cells to vinblastine sulfate. Eur. J. Cell Biol. 23: 115-121.
A. J. Henderson and M. Peaker (1980). The effects of cholchicine on milk secretion, mammary metabolism and blood flow in the goat. Quart. J. Exp. Physiol. 65: 367-378.
P. A. Amato and R. F. Loizzi (1981). The identification and localization of actin and actin-like filaments in lactating guinea pig mammary gland alveolar cells. Cell Motility 1: 329-347.
W. W. Franke, H. W. Heid, C. Grund, S. Winter, C. Freudenstein, E. Schmid, E.-D. Jarasch, and T. W. Keenan (1981). Antibodies to the major insoluble milk fat globule membrane-associated protein: Specific location in apical regions of lactating epithelial cells. J. Cell Biol. 89: 485-494.
H. L. Asch, E. Mayhew, R. O. Lazo, and B. B. Asch (1990). Lipids noncovalently associated with keratins and other cytoskeletal proteins of mouse mammary epithelial cells in primary culture. Biochim. Biophys. Acta 1034: 303-308.
W. W. Franke, M. Hergt, and C. Grund (1987). Rearrangement of the vimentin cytoskeleton during adipose conversion: Formation of an intermediate filament cage around lipid globules. Cell 49: 131-141.
G. Almahbobi and P. F. Hall (1990). The role of intermediate filaments in adrenal steroidogenesis. J. Cell Sci. 97: 679-687.
F. B. P. Wooding, M. Peaker, and J. L. Linzell (1970). Theories of milk secretion: evidence from the electron microscopic examination of milk. Nature (Lond.) 226: 762-764.
M. M. T. Janssen and P. Walstra (1982). Cytoplasmic remnants in milk of certain species. Neth. Milk Dairy J. 36: 365-368.
G. E. Huston and S. Patton (1990). Factors related to the formation of cytoplasmic crescents on milk fat globules. J. Dairy Sci. 73: 2061-2066.
F. B. P. Wooding (1973). Formation of the milk fat globule membrane without participation of the plasmalemma. J. Cell Sci. 13: 221-235.
M. Kralj and N. Pipan (1992). The role of exocytosis in the apocrine secretion of milk lipid globules in mouse mammary gland during lactogenesis. Biol. Cell 75: 211-216 (erratum: (1992). Biol. Cell 76: 288).
M. Sasaki, W. N. Eigel, and T. W. Keenan (1978). Lactose and major milk proteins are present in secretory vesicle-rich fractions from lactating mammary gland. Proc. Natl. Acad. Sci. U.S.A. 75: 5020-5024.
A. Peixoto de Menezes and P. Pinto da Silva (1978). Freeze-fracture observations of the lactating rat mammary gland. Membrane events during milk fat secretion. J. Cell Biol. 76: 767-778.
F. B. P. Wooding and P. Kemp (1975). Ultrastructure of the milk fat globule membrane with and without triglyceride. Cell Tissue Res. 165: 113-127.
W. Buchheim (1982). Paracrystalline arrays of milk fat globule membrane-associ ated proteins as revealed by freeze-fracture. Naturwissenschaft. 69: 505-507.
C. Freudenstein, T. W. Keenan, W. N. Eigel, M. Sasaki, J. Stadler, and W.W. Franke (1979). Preparation and characterization of the inner coat material associated with fat globule membranes from bovine and human milk. Exp. Cell Res. 118: 277-294.
L. R. Banghart, C. W. Chamberlain, J. Velarde, I. V. Korobko, S. L. Ogg, L. J. W. Jack, V. N. Vakharia, and I. H. Mather (1998). Butyrophilin is expressed in mammary epithelial cells from a single-sized messenger RNA as a type I membrane glycoprotein. J. Biol. Chem. 273: 4171-4179.
W. Buchheim, U. Welsch, and S. Patton (1988). Electron microscopy and carbohydrate histochemistry of the human milk fat globule membrane. In L. A. Hanson (ed.), Biology of Human Milk, Nestlé Nutrition Workshop, Vol. 15, Vevey/Raven Press Ltd., New York, pp. 27-44.
I. H. Mather and T. W. Keenan (1975). Studies on the structure of milk fat globule membrane. J. Membrane Biol. 21: 65-85.
S. Patton, S. J. Gendler, and A. P. Spicer (1995). The epithelial mucin, MUC1, of milk, mammary gland and other tissues. Biochim. Biophys. Acta 1241: 407-424.
E.-D. Jarasch, C. Grund, G. Bruder, H. W. Heid, T.W. Keenan, and W. W. Franke (1981). Localization of xanthine oxidase in mammary-gland epithelium and capillary endothelium. Cell 25: 67-82.
D. E. Greenwalt, R. H. Lipsky, C. F. Ockenhouse, H. Ikeda, N. N. Tandon, and G. A. Jamieson (1992). Membrane glycoprotein CD36: A review of its roles in adherence, signal transduction, and transfusion medicine. Blood. 80: 1105-1115.
L. J. W. Jack and I. H. Mather (1990). Cloning and analysis of cDNA encoding bovine butyrophilin, an apical glycoprotein expressed in mammary tissue and secreted in association with the milk-fat globule membrane during lactation. J. Biol. Chem. 265: 14481-14486.
J. Hvarregaard, M. H. Andersen, L. Berglund, J. T. Rasmussen, and T. E. Petersen (1996). Characterization of glycoprotein PAS-6/7 from membranes of bovine milk fat globules. Eur. J. Biochem. 240: 628-636.
H. W. Heid, M. Schnolzer, and T.W. Keenan (1996). Adipocyte differentiation-related protein is secreted into milk as a constituent of milk lipid globule membrane. Biochem. J. 320: 1025-1030.
R. Brandt, M. Pepperle, A. Otto, R. Kraft, F.-D. Boehmer, and R. Grosse (1988). A 13-kilodalton protein purified from milk fat globule membranes is closely related to a mammary-derived growth inhibitor. Biochemistry 27: 1420-1425.
B. L. Mondy and T. W. Keenan (1993). Butyrophilin and xanthine oxidase occur in constant molar proportions in milk lipid globule membrane but vary in amount with breed and stage of lactation. Protoplasma 177: 32-36.
H. M. Valivullah and T. W. Keenan (1989). Butyrophilin of milk lipid globule membrane contains N-linked carbohydrates and cross-links with xanthine oxidase. Int. J. Biochem. 21: 103-107.
T. Ishii, N. Aoki, A. Noda, T. Adachi, R. Nakamura, and T. Matsuda (1995). Carboxy-terminal cytoplasmic domain of mouse butyrophilin specifically associates with a 150-kDa protein of mammary epithelial cells and milk fat globule membrane. Biochim. Biophys. Acta 1245: 285-292.
A. P. Spicer, G. J. Rowse, T. K. Lidner and S. J. Gendler (1995). Delayed mammary tumor progression in Muc-1 null mice. J. Biol. Chem. 270: 30,093-30,101.
D. A. Ruddy, G. S. Kronmal, V. K. Lee, G. A. Mintier, L. Quintana, R. Domingo, Jr., N. C. Meyer, A. Irrinki, E. E. McClelland, A. Fullan, F. A. Mapa, T. Moore, W. Thomas, D. B. Loeb, C. Harmon, Z. Tsuchihashi, R. K. Wolff, R. C. Schatzman, and J. N. Feder (1997). A 1.1-Mb transcript map of the hereditary hemochromatosis locus. Genome Res. 7: 441-456.
A. F. Williams and A. N. Barclay (1988). The immunoglobulin superfamily-domains for cell surface recognition. Ann. Rev. Immunol. 6: 381-405.
D. L. Brasaemle, T. Barber, N. E. Wolins, G. Serrero, E. J. Blanchette-Mackie, and C. Londos (1997). Adipose differentiation-related protein is an ubiquitously expressed lipid storage droplet-associated protein. J. Lipid Res. 38: 2249-2263.
E. M. Rohlfs, D. S. Louie, and S. H. Zeisel (1993). Lipid synthesis and secretion by primary cultures of rat mammary epithelial cells. J. Cell. Physiol. 157: 469-480.
V. L. Spitsberg and R. C. Gorewit (1997). In vitro phosphorylated bovine milk fat globule membrane proteins. J. Nutr. Biochem. 8: 181-189.
M. D. Turner, M. E. Rennison, S. E. Handel, C. J. Wilde, and R. D. Burgoyne (1992). Proteins are secreted by both constitutive and regulated secretory pathways in lactating mouse mammary epithelial cells. J. Cell Biol. 117: 269-278.
T. H. M. Da Costa, K. Taylor, V. Ilic, and D. H. Williamson (1995). Regulation of milk lipid secretion: effects of oxytocin, prolactin and ionomycin on triacylglycerol release from rat mammary gland slices. Biochem. J. 308: 975-981.
F. B. P. Wooding (1971). The structure of the milk fat globule membrane. J. Ultrastr. Res. 37: 388-400.
Rights and permissions
About this article
Cite this article
Mather, I.H., Keenan, T.W. Origin and Secretion of Milk Lipids. J Mammary Gland Biol Neoplasia 3, 259–273 (1998). https://doi.org/10.1023/A:1018711410270
Issue Date:
DOI: https://doi.org/10.1023/A:1018711410270