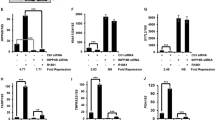
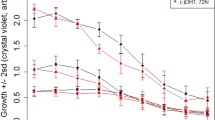
Abstract
Expression of death-signaling genes induces many biochemical cascades resulting in elimination of cells via apoptosis or programmed cell death. GC79/TRPS1 is a novel apoptosis associated gene that encodes a multitype zinc finger GATA-type transcription factor. Expression of GC79/TRPS1 is repressed in the rat ventral prostate and significantly elevated after androgen withdrawal by castration. Castration leads to regression of the prostate caused by apoptosis of androgen-dependent prostate cells. Prostate cancer consists of androgen-dependent and androgen-independent cells. The androgen-independent cells, usually present in the prostate of advanced prostate cancer patients do not have the ability to undergo apoptosis after androgen withdrawal. GC79/TRPS1 expression in androgen-dependent prostate cancer cells is repressed by androgens, while GC79/TRPS1 expression is hardly detectable in androgen-independent prostate cancer cells under cell culture conditions. This suggests that lack of GC79/TRPS1 expression could be a mechanism for the inability to induce the apoptotic pathway in androgen-independent prostate cancer cells after androgen withdrawal. This review will focus on the current knowlegde of the structure and function of GC79/TRPS1, a novel androgen-repressible apoptosis gene.
Similar content being viewed by others
References
Wingo PA, Ries LAG, Giovino GA, et al Annual report to the nation on the status of cancer, 1973–1996, with a special section on lung cancer and tobacco smoking. J Natl Cancer Inst 1999; 91: 675–690.
Cunha GR, Donjacour AA, Cooke PS, et al The endocrinology and developmental biology of the prostate. Endo Rev 1987; 8: 338–362.
Isaacs JT, Coffey DS Adaption versus selection as the mechanism responsible for the relapse to prostatic cancer to androgen ablation therapy as studied in Dunning R-3327-H adenocarcinoma. Cancer Res 1981; 41: 5070–5075.
Rinker-Schaeffer CW, Partin AW, Isaacs WB, Coffey DS, Isaacs JT Molecular and cellular changes associated with the acquisition of metastatic ability by prostatic cancer cells. Prostate 1994; 25: 249–265.
Huggins C, Hodges CV Studies on prostate cancer. I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res 1941; 1: 293–297.
Kyprianou N, Isaacs JT Activation of programmed cell death in the rat ventral prostate after castration. Endocrinology 1988; 12: 357–364.
Kyprianou N, English HF, Isaacs JT Programmed cell death during regression of PC-82 human prostate cancer following androgen ablation. Cancer Res 1990; 50: 3748–3753.
Wyllie AH, Kerr JFR, Currie AR Cell death: The significance of apoptosis. Int Rev Cytol 1980; 68: 251–306.
McDonnell TJ, Troncoso P, Brisby SM, et al Expression of the proto-oncogene bcl-2 in the prostate and its association with emergence of androgen-independent prostate cancer. Cancer Res 1992; 52: 6940–6944.
Eastman JA, Hall SJ, Sehgal I, et al In vivo gene therapy with p53 or p21 adenovirus for prostate cancer. Cancer Res 1995; 55: 5151–5155.
Montpetit ML, Lawless KR, Tenniswood M Androgenrepressed messages in the rat ventral prostate. Prostate 1986; 8: 25–36.
Buttyan R, Olsson CA, Pintar J, et al Induction of the TRPM-2 gene in cells undergoing programmed death. Mol Cell Biol 1989; 9: 3473–3481.
Kyprianou N, Isaacs JT Expression of transforming growth factor-β in the rat ventral prostate during castration-induced programmed cell death. Mol Endo 1989; 3: 1515–1522.
Sells SF, Han SS, Muthukkumar S, et al Expression and function of the leucine zipper protein par-4 in apoptosis. Mol Cell Biol 1997; 17: 3823–3832.
Buttyan R, Zakeri Z, Lockshin RA, Wolgemuth D Cascade induction of c-fos, c-myc and heat shock 70 K transcripts during regression of the rat ventral prostate gland. Mol Endo 1988; 2: 650–657.
Chang GTG, Steenbeek M, Schippers E, et al Characterization of a zinc-finger protein and its association with apoptosis in prostate cancer cells. J Natl Cancer Inst 2000; 92: 1414–1421.
Giedion A, Burdea M, Fruchter Z, Meloni T, Trosc V Autosomal-dominant transmission of the tricho-rhinophalangeal syndrome. Report of 4 unrelated families, review of 60 cases. Helv Paediatr Acta 1973; 28: 249–259.
Carrington PR, Chen H, Altick JA Trichorhinophalangeal syndrome, type I. J Am Acad Dermatol 1994; 31: 331–336.
Itin PH, Bohn S, Mathys D, Guggenheim R, Richard G Trichorhinophalangeal syndrome type III. Dermatology 1996; 193: 349–352.
Momeni P, Glockner G, Schmidt O, et al Mutations in a new gene, encoding a zinc-finger protein, cause tricho-rhinophalangeal syndrome type I. Nat Genet 2000; 24: 71–74.
Ahn J, Ludecke HJ, Lindow S, et al Cloning of the putative tumour suppressor gene for hereditary multiple exostoses (EXT1). Nat Genet 1995; 11: 137–143.
Lüdecke HJ, Schaper J, Meinecke P, et al Genotypic and phenotypic spectrum in tricho-rhino-phalangeal syndrome types I and III. Am J Hum Genet 2001; 68: 81–91.
Malik TH, Shoichet SA, Latham P, Kroll TG, Peters LL, Shivdasani RA Transcriptional repression and developmental functions of the atypical vertebrate GATA protein TRPS1. EMBO J 2001; 20: 1715–1725.
Horoszewics JS, Leong SS, Kawinski E, et al LNCaP model of human prostatic carcinoma. Cancer Res 1983; 43: 1809–1818.
van Steenbrugge GJ, van Uffelen CJ, Bolt J, Schröder FH The human prostatic cancer cell line LNCaP and its derived sublines: An in vitro model for the study of androgen sensitivity. J Steroid Biochem Mol Biol 1991; 40: 207–214.
Chang GTG, Blok LJ, Steenbeek M, et al Differentially expressed genes in androgen-dependent and-independent prostate carcinomas. Cancer Res 1997; 57: 4075–4081.
Chang GTG, Tapsi N, Steenbeek M, et al Identification of a gene on human chromosome 8q11, that is differentially expressed during prostate cancer progression. Int J Cancer 1999; 83: 506–511.
Blok LJ, Chang GTG, Steenbeek-Slotboom M, et al Regulation of expression of Na+,K+-ATPase in androgen-dependent and androgen-independent prostate cancer. Br J Cancer 1999; 81: 28–36.
Chang GTG, Blok LJ, Steenbeek M, et al Identification and characterization of genes involved in the progression of prostate cancer. In: Belldegrun A, Kirby RS, Newling WW, eds. New Perspective in Prostate Cancer, 2nd edn. Oxford: Isis Medical Media Ltd. 2000: 73–84.
Kozak M An analysis of vertebrate mRNA sequences: Intimations of translational control. J Cell Biol 1991; 115: 887–903.
Ginsberg AM, King BO, Roeder RG Xenopus 5S gene transcription factor TFIIIA: Characterization of a cDNA clone and measurement of RNA levels throughout development. Cell 1984; 39: 479–489.
Tautz D Regulation of the Drosophila segmentation gene hunchback by two maternal morphogenetic centres. Nature 1988; 332: 281–284.
Georgopoulos K, Moore DD, Derfler B Ikaros, an early lymphoid-specific transcription factor and a putative mediator for T cell commitment. Science 1992; 258: 808–812.
Brown KE, Guest SS, Smale ST, Hahm K, Merkenschlager M, Fisher AG Association of transcriptionally silent genes with Ikaros complexes at centromeric heterochromatin. Cell 1997; 91: 845–854.
Sun L, Liu A, Georgopoulos K. Zinc finger-mediated protein interactions modulate Ikaros activity, a molecular control of lymphocyte development. EMBO J 1996; 15: 5358–5369.
Morgan B, Sun L, Avitahl N, et al Aiolos, a lymphoid restricted transcription factor that interacts with Ikaros to regulate lymphocyte differentiation. EMBO J 1997; 16: 2004–2013.
Hahm K, Cobb BS, McCarty AS, et al Helios, aTcell-restricted Ikaros family member that quantitatively associates with Ikaros at centromeric heterochromatin. Genes Dev 1998; 12: 782–796.
Kelley CM, Ikeda T, Koipally J, et al Helios, a novel dimerization partner of Ikaros expressed in the earliest hematopoietic progenitors. Curr Biol 1998; 8: 508–515.
Perdomo J, Holmes M, Chong B, Crossley M Eos, pegasus, two members of the ikaros family of proteins with distinct DNA binding activities. J Biol Chem 2000; 275: 38347–38354.
Koipally J, Renold A, Kim J, Georgopoulos K Repression by Ikaros and Aiolos is mediated through histone deacetylase complexes. EMBO J 1999; 18: 3090–3100.
Kim J, Sif S, Jones B, et al Ikaros DNA-binding proteins direct formation of chromatin remodeling complexes in lymphocytes. Immunity 1999; 10: 345–355.
Wang JH, Nichogiannopoulou A, Wu L, et al Selective defects in the development of the fetal and adult lymphoid system in mice with an Ikaros null mutation. Immunity 1996; 5: 537–549.
Koipally J, Georgopoulos K Ikaros interactions with CtBP reveal a repression mechanism that is independent of histone deacetylase activity. J Biol Chem 2000; 275: 19594–19602.
Orkin SH GATA-binding transcription factors in hematopoietic cells. Blood 1992; 80: 575–581.
Charron F, Nemer M GATA transcription factors and cardiac development. Semin Cell Dev Biol 1999; 10: 85–91.
Trainor CD, Ghirlando R, Simpson MA GATA zinc finger interactions modulateDNAbinding and transactivation. J Biol Chem 2000; 275: 28157–28166.
Molkentin JD The zinc finger-containing transcription factors GATA-4,-5, and-6. J Biol Chem 2000; 275: 38949–38952.
Lowry JA, Atchley WR Molecular evolution of the GATA family of transcription factors: Conservation within the DNAbinding domain. J Mol Evol 2000; 50: 103–115.
Hastie N The genetics of Wilms' tumor: A case of disrupted development. Annu Rev Genet 1994; 28: 523–558.
Laverriere AC, MacNeill C, Mueller C, Poelman RE, Burch JBE, Evans T GATA4/5/6, a subfamily of three transcription factors transcribed in developing heart and gut. J Biol Chem 1994; 269: 23177–23184.
Tong Q, Dalgin G, Xu H, Ting CN, Leiden JM, Hotamisligil GS Function of GATA transcription factors in preadipocyteadipocyte transition. Science 2000; 290: 134–138.
Yomogida K, Ohtani H, Harigae H, et al Developmental stageand spermatogenic cycle-specific expression of transcription factor GATA-1 in mouse Sertoli cells. Development 1994; 120: 1759–1766.
Arceci RJ, King AA, Simon MC, Orkin SH, Wilson DB Mouse GATA-4: A retinoic acid-inducible GATA-binding transcription factor expressed in endodermally derived tissues and heart. Mol Cell Biol 1993; 13: 2235–2246.
Heikinheimo M, Ermolaeva M, Bielinska M, et al Expression and hormonal regulation of transcription factors GATA-4 and GATA-6 in the mouse ovary. Endocrinology 1997; 138: 3505–3514.
Laitinen MP, Anttonen M, Ketola I, et al Transcription factors GATA-4 andGATA-6 and aGATA family cofactor, FOG-2, are expressed in human ovary and sex cord-derived ovarian tumors. J Clin Endocrinol Metab 2000; 85: 3476–3483.
Ketola I, Rahman N, Toppari J, et al Expression and regulation of transcription factors GATA-4 and GATA-6 in developing mouse testis. Endocrinology 1999; 40: 1470–1480.
Ketola I, Pentikainen V, Vaskivuo T, et al Expression of transcription factorGATA-4 during human testicular development and disease. J Clin Endocrinol Metab 2000; 85: 3925–3931.
Zhou Y, Kim KC, Onodera K, et al Rescue of the embryonic lethal hematopoietic defect reveals a critical role for GATA-2 in urogenital development. EMBO J 1998; 17: 6689–6700.
Lakshmanan G, Lieuw KH, Lim KC Localization of distant urogenital system-, central nervous system-, and endocardiumspecific transcriptional regulatory elements in the GATA-3 locus. Mol Cell Biol 1999; 19: 1558–1568.
Molkentin JD, Tymitz KM, Richardson JA, Olson EN Abnormalities of the genitourinary tract in female mice lacking GATA5. Mol Cell Biol 2000; 20: 5256–5260.
Hoch RV, Thompson DA, Baker RJ, Weigel RJ GATA-3 is expressed in association with estrogen receptor in breast cancer. Int J Cancer 1999; 84: 122–128.
Siltanen S, Anttonen M, Heikkila P, et al Transcription factor GATA-4 is expressed in pediatric yolk sac tumors. Am J Pathol 1999; 155: 1823–1829.
Feng ZM, Wu AZ, Chen CLC Testicular GATA-1 factor regulates the promoter activity of rat inhibin α-subunit in MA-10 Leydig tumor cells. Mol Endo 1998; 12: 378–390.
Feng ZM, Wu AZ, Zhang Z, Chen CL GATA-1 and GATA-4 transactivate inhibin/activin beta-B-subunit gene transcription in testicular cells. Mol Endo 2000; 14: 1820–1835.
Perez-Stable CM, Pozas A, Roos BA Arole forGATAtranscription factors in the androgen regulation of the prostate-specific antigen gene enhancer. Mol Cell Endocrinol 2000; 167: 43–53.
Lawson MA, Whyte DB, Mellon PL GATAfactors are essential for activity of the neuron-specific enhancer of the gonadotropinreleasing hormone gene. Mol Cell Biol 1996; 16: 3596–3605.
Davis DL, Burch JB The chicken vitellogenin II gene is flanked by a GATA factor-dependent estrogen response unit. Mol Endo 1996; 10: 937–944.
Silverman E, Eimerl S, Orly J CCAAT enhancer-binding protein beta and GATA-4 binding regions within the promoter of the steroidogenic acute regulatory protein (StAR) gene are required for transcription in rat ovarian cells. J Biol Chem 1999; 274: 17987–17996.
Viger RS, Mertineit C, Trasler JM, Nemer M Transcription factor GATA-4 is expressed in a sexually dimorphic pattern during mouse gonadal development and is a potent activator of the Müllerian inhibiting substance promoter. Development 1998; 125: 2665–2675.
Tremblay JJ, Viger RS Transcription factor GATA-4 enhances Müllerian inhibiting substance gene transcription through a direct interaction with the nuclear receptor SF-1. Mol Endo 1999; 13: 1388–1401.
Watanabe K, Clarke TR, Lane AH, Wang X, Donahoe PK Endogenous expression of Müllerian inhibiting substance in early postnatal rat sertoli cells requires multiple steroidogenic factor-1 and GATA-4-binding sites. Proc Natl Acad Sci USA 2000; 97: 1624–1629.
Beau C, Rauch M, Joulin V, Jegou B, Guerrier D GATA-1 is a potential repressor of anti-Müllerian hormone expression during the establishment of puberty in the mouse. Mol Reprod Dev 2000; 56: 124–138.
De Santa Barbara P, Moniot B, Poulat F, Boizet B, Berta P Steroidogenic factor-1 regulates transcription of the human anti-M¨ ullerian hormone receptor. J Biol Chem 1998; 273: 29654–29660.
Blobel GA, Sieff CA, Orkin SH Ligand-dependent repression of the erythroid transcription factor GATA-1 by the estrogen receptor. Mol Cell Biol 1995; 15: 3147–3153.
Chang TJ, Scher BM, Waxman S, Scher W Inhibition of mouse GATA-1 function by the glucocorticoid receptor: Possible mechanism of steroid inhibition of erythroleukemia cell differentiation. Mol Endo 1993; 7: 528–542.
Trainor CD, Evans T, Felsenfeld G Negative regulation of chicken GATA-1 promoter activity mediated by a hormone response element. Mol Endo 1995; 9: 1135–1146.
Zhang M, Magit D, Sager R Expression of maspin in prostate cells is regulated by a positive ets element and a negative hormonal responsive element site recognized by androgen receptor. Proc Natl Acad Sci USA 1997; 94: 5673–5678.
Wolf DA, Kohlhuber F, Schulz P, Fittler F, Eick D Transcriptional down-regulation of c-myc in human prostate carcinoma cells by the synthetic androgen mibolerone. Br J Cancer 1992; 65: 376–382.
Prendergast GC Mechanisms of apoptosis by c-myc. Oncogene 1999; 18: 2967–2987.
Fleming WH, Hamel A, MacDonald R, et al Expression of the c-myc protooncogen in human prostatic carcinoma and benign prostatic hyperplasia. Cancer Res 1986; 46: 1535–1538.
Jenkins RB, Qian J, Lieber MM, Bostwick DG Detection of c-myc oncogene amplification and chromosomal anomalies in metastatic prostatic carcinoma by fluorescence in situ hybridization. Cancer Res 1997; 57: 524–531.
Sato K, Qian J, Slezak JM, et al Clinical significance of alterations of chromosome 8 in high-grade, advanced, nonmetastatic prostate carcinoma. J Nat Cancer Inst 1999; 91: 1574–1580.
Isaacs JT Apoptosis: Translating theory to therapy for prostate cancer. J Natl Cancer Inst 2000; 92: 1367–1369.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chang, G.T.G., van den Bemd, GJ.C.M., Jhamai, M. et al. Structure and function of GC79/TRPS1, a novel androgen-repressible apoptosis gene. Apoptosis 7, 13–21 (2002). https://doi.org/10.1023/A:1013504710343
Issue Date:
DOI: https://doi.org/10.1023/A:1013504710343