Abstract
Purpose. The purpose of this study was to relate the combination of glass transition temperature (T g) and temperature of measurement with the mechanical and compaction properties of some test materials.
Methods. Copolymers with different T gs were synthesised by free radical copolymerisation of methyl methacrylate with lauryl methacrylate. Elastic moduli were measured by dynamic mechanical analysis at different strain rates and temperatures. Compaction experiments were performed at different compaction speeds and temperatures.
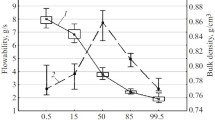
Results. The difference between temperature of measurement and T g appears to determine both elastic modulus and yield strength completely. They both decrease with decreasing difference between temperature of measurement and Tg and increase with strain rate. At temperatures of measurement higher than the T g, the elastic modulus is extremely low because the materials behave as rubbers. Consequently, the amount of energy stored during compaction decreases when the compaction temperature approaches the T g and increases with strain rate. When the compaction temperature is higher than the T g, the amount of stored energy is extremely large. The compaction experiments show that the final tablet porosity is completely determined by stress relaxation phenomena. Consequently, the final tablet porosity follows exactly the same relation as that of stored energy.
Conclusions. The final tablet porosity is unequivocally determined by the amount of stored energy. This implies that tablet production at a temperature of about 20 K under the glass transition temperature of the material yields tablets with minimum porosity.
Similar content being viewed by others
REFERENCES
R. J. Roberts and R. C. Rowe. J. Pharm. Pharmacol. 37:377–384 (1985).
C. E. Bos, G. K. Bolhuis, H. Van Doorne, and C. F. Lerk. Pharm Weekblad Sci. Ed. 9:274–282 (1987).
J. E. Rees and K. D. Tsardaka. Eur. J. Pharm. Biopharm. 40:193–197 (1994).
K. Van der Voort Maarschalk, K. H. Vromans, W. Groenendijk, G. K. Bolhuis, and C. F. Lerk. Eur. J. Pharm Biopharm. Submitted (1996).
R. C. Rowe and R. J. Roberts. In G. Alderborn and C. Nyström (eds.) Pharmaceutical powder compaction technology, Dekker, New York, 1996, pp. 283–322.
J. M. G. Cowie. Polymers; Chemistry and Physics of Modern Materials, Blackie, London, 1991.
D. W. Van Krevelen, Properties of polymers, Elsevier, Amsterdam, 1990.
R. J. Crawford. Plastics Engineering, Pergamon, Oxford, 1987.
K. Van der Voort Maarschalk, K. Zuurman, H. Vromans, G. K. Bolhuis, and C. F. Lerk. Int. J. Pharm. 140:185–193 (1996).
K. Van der Voort Maarschalk, H. Vromans, G. K. Bolhuis, and C. F. Lerk. Eur. J. Pharm. Biopharm. 42:49–55 (1996).
R. W. Heckel. Trans. Met. AIME. 221:671–675 (1961).
R. W. Heckel. Trans. Met. Soc. AIME. 221:1001–1008 (1961).
H. F. Mark, Encyclopaedia of Polymer Science and Engineering, Wiley, New York, 1987.
W. Duckworth. J. Am. Ceram. Soc. 36:68 (1953).
R. Steendam and C. F. Lerk. J. Contr. Release. Submitted.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Maarschalk, K.V.d.V., Zuurman, K., Van Steenbergen, M.J. et al. Effect of Compaction Temperature on Consolidation of Amorphous Copolymers with Different Glass Transition Temperatures. Pharm Res 14, 415–419 (1997). https://doi.org/10.1023/A:1012078928297
Issue Date:
DOI: https://doi.org/10.1023/A:1012078928297