Abstract
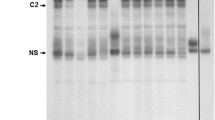
We have cloned the rat fibroblast growth factor-2 (FGF-2) promoter region including 1058 base pairs (bp) of 5′-flanking DNA. Complete sequencing of this promoter region revealed a 74 bp domain between nucleotides -793 and -720 that was greater than 97% A/G-rich. A repeat of the sequence 5′-AGGGAGGG-3′ separated by 11 bp was located at the core of this domain. A 37 bp A/G-rich oligonucleotide containing these AGGG-repeat sequences was synthesised, and tested for function on a minimal herpes simplex virus thymidine kinase (TK) promoter, fused to the firefly luciferase gene (TKp.luc), in transiently transfected neonatal rat cardiac myocytes. Promoter activity was stimulated ~3 fold in the presence of AGGG-repeat sequences. This effect was neither tissue or species-specific since TK promoter activity was increased ~11 fold in both rat and human glial tumor cells. Four specific complexes (C14) were detected between neonatal rat heart nuclear proteins and the 37 bp A/G-rich oligonucleotide by gel mobility shift assay. Competition with excess unlabelled 37 bp A/G-rich oligonucleotide revealed that two complexes represented very high affinity/specificity interactions (C2 > C4) while C1 and C3 were of lower affinity. As a result, competition with up to a 25 fold molar excess of 37 bp A/G-rich oligonucleotide led to the loss of C2 and C4, and a corresponding and transient increase in the levels of C1 and C3, which themselves were reduced with more competitor oligonucleotide. The AGGG-repeat resembles the 5′-gGGGAGGG-3′ sequence previously implicated in the response of the atrial natriuretic factor promoter to the α-adrenergic agonist, phenylephrine. Although an additional 1.5 fold increase in TK promoter activity was detected in the presence of the 37 bp A/G-rich oligonucleotide with phenylephrine treatment of transfected myocytes, this effect was not statistically significant. Furthermore, there was no difference in the gel mobility shift (C14) pattern obtained with the 37 bp A/G-rich oligonucleotide and nuclear protein isolated from neonatal rat cardiac myocytes grown in the presence or absence of norepinephrine. These data suggest that the A/G rich sequences in the rat FGF-2 gene 5′-flanking DNA, including the AGGG-repeat, are able to confer stimulatory activity on a promoter in a tissue- and species-independent manner, but alone are not able to induce a significant phenylephrine response in neonatal rat cardiac myocytes.
Similar content being viewed by others
References
Sugi Y, Sasse J, Lough J: Inhibition of precardiac mesoderm cell proliferation by antisense oligodeoxynucleotide complementary to fibroblast growth factor-2 (FGF-2). Dev Biol 157: 28–37, 1993
Mima T, Ueno H, Fischman DA, Williams LT, Mikawa T: Fibroblast growth factor receptor is required for in vivo cardiac myocyte proliferation at early embryonic stages of heart development. Proc Natl Acad Sci USA 92: 467–471, 1995
Clarke MS, Caldwell RW, Chiao H, Miyake K, McNeil PL: Contractioninduced cell wounding and release of basic fibroblast growth factor. Circ Res 76: 927–934, 1995
Kaye D, Pimental D, Prasad S, Maki S, Berger HJ, McNeil PL, Smith TW, Kelly RA: Role of transiently altered sarcolemmal membrane permeability and basic fibroblast growth factor release in the hypertrophic response of adult rat ventricular myocytes to increased mechanical activity in vitro. J Clin Invest 97: 281–291, 1996
Unger EF, Banai S, Shou M, Lazarous DF, Jaklitsch MT, Scheinowitz M, Correa R, Ningbeil C, Epstein SE: Basic fibroblast growth factor enhances myocardial collateral flow in a canine model. Am J Physiol 266 (Heart Circ Physiol 35): H158–H1595, 1994
Lazarous DF, Scheinowitz M, Shou M, Hodge E, Majanayagam MAS, Hunsberger S, Robinson Jr WG, Stiber JA, Correa R, Epstein SE, Unger EF: Effects of chronic systemic administration of basic fibroblast growth factor on collateral development in the canine heart. Circulation 91: 145–153, 1995
Fox JC, Shanley JR: Antisense inhibition of basic fibroblast growth factor induces apoptosis in vascular smooth muscle cells. J Biol Chem 271: 12578–12584, 1996
Padua RR, Sethi R, Dhalla NS, Kardami E: Basic fibroblast growth factor is cardioprotective in ischemia-reperfusion injury. Mol Cell Biochem 143: 129–135, 1995
Padua RR, Sethi R, Davey-Forgie SE, Lui L, Dhalla N, Kardami E: Cardioprotection and basic fibroblast growth factor. In: N.S. Dhalla, P.K. Singal, R.E. Beamish (eds). Heart Hypertrophy and Failure. Kluwer Academic Publishers, Boston, 1996, pp 501–518.
Shibata F, Baird A, Florkiewicz RZ: Functional characterization of the human basic fibroblast growth factor promoter. Growth Factors 4: 277–287, 1991
Pasumarthi KBS, Jin Y, Cattini PA: Cloning of the rat fibroblast growth factor-2 promoter region and its response to mitogenic stimuli in glioma C6 cells. J Neurochem 68: 898–908, 1997
Ueba T, Nosaka T, Takahashi JA, Shibata F, Florkiewicz RZ, Vogelstein B, Oda Y, Kikuchi H, Hatanaka M: Transcriptional regulation of basic fibroblast growth factor gene by p53 in human glioblastma and hepatocellular carcinoma cells. Proc Natl Acad Sci USA 91: 9009–9013, 1994
Biesiada E, Razandi M, Levin ER: Egr-1 activates basic fibroblast growth factor transcription. J Biol Chem 271: 18576–18581, 1996
Ardati A, Nemer M A nuclear pathway for α1-adrenergic receptor signaling in cardiac cells. EMBO J 12: 5131–5139, 1993
Pasumarthi KBS, Kardami E, Cattini PA: High and low molecular weight fibroblast growth factor-2 increase proliferation of neonatal rat cardiac myocytes but have differential effects on binucleation and nuclear morphology: Evidence for both paracrine and intracrine actions of fibroblast growth factor-2. Circ Res 78: 126–136, 1996
Dignam JD, Lebovitz RM, Roeder RG: Accurate transcription initiation by RNA polymerase II in a soluble extract from isolated mammalian nuclei. Nucl Acids Res 11: 1475–1489, 1983
Andrews NC, Faller DV: A rapid micropreparation technique for extraction of DNAbinding proteins from limiting numbers of mammalian cells. Nucl Acids Res 19: 2499, 1991
Nordeen SK: Luciferase reporter gene vectors for analysis of promoters and enhancers. Biotechniques 6: 454–458, 1988
Smith PK, Krohn RI, Hermansen GT, Mallia AK, Gartner FH, Fugimoto EK, Goeke NM, Olson BJ, Klenk DC: Measurement of protein using bicinchoninic acid. Anal Biochem 150: 76–85, 1985
Baldwin AS: Analysis of sequence-specific DNA-binding proteins by the gel mobility shift assay. DNA Protein Eng Tech 2: 73–76, 1990
Lytras A, Cattini PA: Human chorionic somatomammotropin gene enhancer activity is dependent on the blockade of a repressor mechanism. Mol Endocrinol 8: 478–489, 1994
Boulikas T: Homeodomain protein binding sites, inverted repeats, and nuclear matrix attachment regions along the human β-globin gene complex. J Cell Biochem 52: 23–36, 1993
Kardami E, Liu L, Padua RR, Fandrich RR, Pasumarthi SKB, Cattini PA: Regulation of basic fibroblast growth factor (bFGF) and FGF receptors in the heart. Ann NY Acad Sci 752: 353–369, 1995
Bikfalvi A, Klein S, Pintucci G, Rifkin DB: Biological roles of fibroblast growth factor-2. Endocrine Rev 18: 26–45, 1997
Ornitz DM, Xu J, Colvin JS, McEwen DG, MacArthur CA, Coulier F, Gao G, Goldfarb M: Receptor specificity of the fibroblast growth factor family. J Biol Chem 271: 15292–15297, 1996
Sprenkle AB, Murray SF, Glembotski CC: Involvement of multiple cis-elements in basal and α-adrenergic agonist-inducible ANF transcription. Roles for serum response elements and an Sp-l-like element. Circ Res 77: 1060–1069, 1995
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Detillieux, K.A., Meyers, A.F., Meij, J.T. et al. An A/G-rich motif in the rat fibroblast growth factor-2 gene confers enhancer activity on a heterologous promoter in neonatal rat cardiac myocytes. Mol Cell Biochem 188, 169–176 (1998). https://doi.org/10.1023/A:1006886307083
Issue Date:
DOI: https://doi.org/10.1023/A:1006886307083