Abstract
Neuronal morphogenesis depends on the organization of cytoskeletal elements among which microtubules play a very important role. The organization of microtubules is controlled by the presence of microtubule-associated proteins (MAPs), the activity of which is modulated by phosphorylation and dephosphorylation. One of these MAPs is MAP1B, which is very abundant within growing axons of developing neurons where it is found phosphorylated by several protein kinases including CK2. The expression of MAP1B is notably decreased after neuronal maturation in parallel with a change in the localization of the protein, which becomes largely concentrated in neuronal cell bodies and dendrites. Interestingly, MAP1B remains highly phosphorylated at sites targeted by protein kinase CK2 in mature neurons.
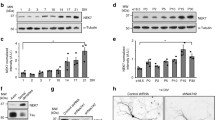
We have analyzed the expression and localization of CK2 catalytic subunits along neuronal development. CK2α subunit appears early during development whereas CK2α′ subunit appears within mature neurons at the time of dendrite maturation and synaptogenesis, in parallel with the change in the localization of MAP1B. CK2α subunit is found associated with microtubule preparations obtained from either grey matter or white matter from adult bovine brain, whereas CK2α′ subunit is highly enriched in microtubules obtained from grey matter. These results lend support to the hypothesis that CK2α′ subunit is concentrated in neuronal cell bodies and dendrites, where it associates with microtubules, thus contributing to the increased phosphorylation of MAP1B in this localization in mature neurons.
Similar content being viewed by others
References
Avila J, Domínguez J, Díaz-Nido J: Regulation of microtubule dynamics by microtubule-associated protein expression and phosphorylation during neonatal development of MAP 1.2 J Cell Biol 106: 1573–1581, 1988
Belmont LD, Mitchison TJ: Identification of a protein that interacts with tubulin dimers and increases the catastrophe rate of microtubules. Cell 84: 623–631, 1996
Marklund U, Larsson N, Gradin HM, Brattsand G, Gullberg M: Oncoprotein 18 is a phosphorylation-responsive regulator of microtubule dynamics. EMBO J 15: 5290–5298, 1996
Tucker RP, Garner CC, Matus A: In situ localization of microtubule associated protein mRNA in the developing and adult rat brain. Neuron 2: 1245–1246, 1989
Brugg B, Deddy D, Matus A: Attenuation of MAP1B (MAP5) expression by antisense oligodeoxynucleotides inhibits initiation of neurite outgrowth. Neuroscience 52: 489–496, 1993
Díaz-Nido J, Serrano L, Méndez E, Avila J: A casein kinase II-related activity is involved in phosphorylation of microtubule-associated protein MAP1B during neuroblastoma cell. J Cell Biol 106: 2057–2065, 1988
Gard DL, Kirschner MW: A polymer-dependent increase in phosphorylation of β-tubulin accompanies differentiation of a mouse neuroblastoma cell line. J Cell Biol 100: 764–774, 1985
Ulloa L, Díaz-Nido J, Avila J: Depletion of casein kinase II by antisense oligodeoxy-nucleotide prevents neuritogenesis in neuroblastoma cells. EMBO J 12: 1633–1640, 1993a
Ulloa L, Avila J, Díaz-Nido J: Heterogeinity in the phosphorylation of MAP1B during rat brain development. J Neurochem 61: 961–972, 1993b
Yu IJ, Spector DL, Bae Y-S, Marsak DR: Immunocytochemical localization of casein kinase II during interphase and mitosis. J Cell Biol 114: 1217–1232, 1991
Chester N, Yu IJ, Marshack DR: Identification and characterization of protein kinase CKII isoforms in HeLa cells. J Biol Chem 270: 7501–7514, 1995
Saneto RP, De Vellis J: Neuronal and glial cells: Cell culture nervous system. In: A.J. Turner and H.S. Bachelard (eds). Neurochemistry. A practical approach. IRL Press, Oxford, 1987, pp 27–64
Dotti CG, Sullivan CA, Banker GA: The establishment of polarity by hippocampal neurons in culture. J Neurosci 8: 1454–1468, 1988
Ulloa L, Diéz-Guerra J, Avila J, Díaz-Nido J: Localization of differentially phosphorylated isoforms of microtubule-associated protein 1B (MAP1B) into axons and dendrites of cultured rat hippocampal neurons. Neuroscience 61: 211–223, 1994
Benitez MJ, Mier G, Briones F, Moreno FJ, Jiménez JS: A surfaceplasmon-resonance analysis of polylysine interactions with a peptide substrate of protein kinase CK2 and with the enzyme. Biochem J 324: 987–994, 1997
Vallee RB, Bloom GS, Luca FC: Differential studies and distribution of the high molecular weight brain microtubule associated proteins MAP1 and MAP2. Ann NY Acad 466: 134–144, 1986
Towbin H, Staehelin T, Gordon J: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc Natl Acad Sci USA 76: 4350–4354, 1979
Ulloa L, Donbradi V, Díaz-Nido J, Szucs K, Gergely P, Fiedrich P, Avila J; Dephosphorylation of distinct sites on microtubule-associated protein MAP1B by protein phosphatases 1, 2A and 2B. FEBS Lett. 330: 85–89, 1993c
Heriché JK, Lebrin F, Rabilloud T, Leroy D, Chambaz EM, Coldbey Y: Regulation of protein phosphatase 2A by direct interaction with casein kinase 2α. Science 276: 952–955, 1997
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moreno, F.J., Díaz-Nido, J., Jiménez, J.S. et al. Distribution of CK2, its substrate MAP1B and phosphatases in neuronal cells. Mol Cell Biochem 191, 201–205 (1999). https://doi.org/10.1023/A:1006836211318
Issue Date:
DOI: https://doi.org/10.1023/A:1006836211318