Abstract
The present study was conducted to evaluate the activity and toxicity profile of radiation (RT) and concomitant chemotherapy in patients with glioblastoma multiforme (GBM). Thirty-nine patients were treated postoperatively with RT and concomitant administration of paclitaxel. Cranial irradiation was initiated 2–3 weeks postoperatively and was administered in 2.0 fractions, one fraction per day, for 5 consecutive days per week, to a total of 60 Gy. Paclitaxel was delivered at a dose of 100 mg/m2 over 3-h once weekly for 6 weeks.
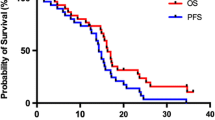
Thirty-three patients received all 6 cycles of paclitaxel according to the protocol. Totally, 217 cycles were delivered all of them at full dose. The median relative dose intensity of paclitaxel was 1 (range 0.88–1.1). Three (7.5%) patients achieved complete and 9 (23%) partial response, while 12 (30.5%) patients demonstrated stabilization of the disease. Side effects from combined chemoradiotherapy were mainly mild. Grade III toxicity included infection (7.5%) and alopecia (5%). Median time to progression was 6 (range 0.9–27) months and median survival 10.7 (range 0.9–39.5+) months.
The present study has clearly shown that 100 mg/m2 of paclitaxel in 1-h infusion weekly can be safely given concomitantly with RT in patients with GBM with manageable toxicity. However, the efficacy of this combined modality treatment does not appear to be superior to that of RT alone.
Similar content being viewed by others
References
Walker AE, Robins M, Weinfeld FD: Epidemiology of brain tumors: the national survey of intracranial neoplasm. Neurology 35: 219, 1985
Boring CC, Squires TS, Tong T, Montgomery S: Cancer statistics, 1994. CA Cancer J Clin 44: 7–26, 1994
Fine HA, Dear KB, Loeffler JS, Black PM, Canellos GP: Meta-analysis of radiation therapy with and without adjuvant chemotherapy for malignant gliomas in adults. Cancer 71: 2585–2597, 1993
Cahan MA, Walter KA, Colvin OM, Brem H: Cytotoxicity of taxol in vitro against human and rat malignant brain tumors. Cancer Chem Pharm 33: 441–444, 1994
Helson L, Helson C, Malik S, Ainsworth S, Mangiardi J: A saturation threshold for taxol cytotoxicity in human glial and neuroblastoma cells. Anti, Canc Drug 4: 487–490, 1993
Hei TK, Piao CQ, Geard CR, Hall EJ: Taxol and ionising radiation: interaction and mechanisms. Int J Radiation Oncol Biol Phys 29: 267–271, 1994
Geard CR, Jones JM, Schiff PB: Taxol and radiation. Monogr Natl Cancer Inst 15: 89–94, 1993
Nitta T, Sato K: Prognostic implication of the extent of surgical resection in patients with intracranial malignant gliomas. Cancer 75: 2727–2731, 1995
Burger PC, Vogel FS, Green SB, Strike TA: Glioblastoma multiforme and anaplastic astrocytoma. Pathologic criteria and prognostic implications. Cancer 56: 1106–1111, 1985
MacDonald DR, Cascino TL, Schold SCJ, Cairncross JG: Response criteria for phase II studies of supratentorial malignant glioma. J Neurosurg 8: 1277–1280, 1990
Levin VA, Edwards MS, Wright DC, Seager ML, Schimberg TP, Townsend JJ, Wilson CB: Modified procarbazine, CCNU and vincristine (PCV-3) combination chemotherapy in the treatment of malignant brain tumors. Cancer Treat Rep 64: 237–241, 1980
Soini Y, Niemela A, Kamel D, Herva R, Bloigu R, Paakko P, Vahakangas K: p53 immunohistochemical positivity as a prognostic marker in intracranial tumors. APMIS 102: 786–792, 1994
Kaplan EL, Meier P: Non parametric estimation from incomplete observations. J Am Stat Assoc 53: 457–481, 1958
Mantel M: Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 50: 163–170, 1966
Vokes EE, Pajak TF: Enhancing the therapeutic index of concomitant chemoradiotherapy for head and neck cancer. Ann Oncol 9: 471–473, 1998
Choy H, Akerley W, Safran H, Clark J, Rege V, Papa A, Glantz M, Puthawala Y, Soderberg C, Leone L: Phase I study trial of outpatient weekly paclitaxel and concurrent radiation therapy for advanced non-small cell lung cancer. J Clin Oncol 12: 2682–2686, 1994
Prados MD, Schold SC, Spence AM, Berger MS, McAllister LD, Mehta MP, Gilbert MR, Fulton D, Kuhn J, Lamborn K, Rector DJ, Chang SM: Phase II study of paclitaxel in patients with recurrent malignant glioma. J Clin Oncol 14: 2316–2321, 1996
Chamberlain MC, Kormanik P: Salvage chemotherapy with paclitaxel for recurrent primary brain tumors. J Clin Oncol 13: 2066–2071, 1995
Chang SM, Kuhn JG, Rizzo J, Robins HI, Schold C Jr, Spence AM, Berger MS, Mehta MP, Bozik ME, Pollack I, Gilbert M, Fulton D, Rankin C, Malec M, Prados M: Phase I study of paclitaxel in patients with recurrent malignant glioma: a North American Brain Tumor Consortium report. J Clin Oncol 16: 2188–2194, 1998
Briasoulis E, Tzamakou E, Nicolaides C, Karavasilis V, Pavlidis N: Long-term weekly administration of paclitaxel in chemotherapy-naive patients, phase I study: single versus cumulative dose limiting toxicity (Abstr). Proc Am Soc Clin Oncol 16: 812, 1997
Glantz MJ, Choy H, Kearns CM, Cole BF, Mills P, Zuhowski EG, Saris S, Rhodes CH, Stopa E, Egorin MJ: Phase I study of weekly outpatient paclitaxel and concurrent cranial irradiation in adults with astrocytomas. J Clin Oncol 14: 600–609, 1996
Brandes AA, Scelzi E, Salmistraro G, Ermani M, Carollo C, Berti F, Zampieri P, Baiocchi C, Friorentino MV: Incidence of risk of thromboembolism during treatment of high-grade gliomas: a prospective study. Eur J Cancer 33: 1592–1596, 1997
Cheruku R, Tapazoglou E, Ensley J, Kish JA, Cummings GD, Al-Sarraf M: The incidence and signifi-cance of thromboembolic complications in patients with high-grade gliomas. Cancer 68: 2621–2624, 1991
Goldman CK, Kim J, Wong WL, King V, Brock T, Gillespie GY: Epidermal growth factor stimulates vascular endothelial growth factor production by human malignant glioma cells: a model of glioblastoma multiforme pathophysiology. Mol Biol Cell 4: 121–133, 1993
Kornowski R, Pines A, Constantini S: Persistent activation of thrombocytes in neurosurgical patients operated for primary brain tumors. Acta Neurochir 121: 146–148, 1993
Hainsworth JD, Erland JB, Kalman LA, Schreeder MT, Greco FA: Carcinoma of unknown primary site: treatment with 1-hour paclitaxel, carboplatin, and extended-schedule etoposide. J Clin Oncol 15: 2385–2393, 1997
Bhattacharjee MB, Bruner JM: 11–6 p53 protein in pediatric malignant astrocytomas: a study of 21 patients. J Neuro-Oncol 32: 225–233, 1997
Kordek R, Biernat W, Alwasiak J, Maculewicz R, Yanagihara R, Liberski PP:p53 protein and epidermal growth factor receptor expression in human astrocytomas. J Neuro-Oncol 26: 11–16, 1995
Haas-Kogan DA, Yount G, Haas M, Levi D, Kogan SS, Hu L, Vidair C, Deen DF, Dewey WC, Israel MA: p53-dependent G1 arrest and p53-independent apoptosis influence the radiobiologic response of glioblastoma. Int J Radiat Oncol Biol Phys 36: 95–103, 1996
Kondo S, Barnett GH, Hara H, Morimura T, Takeuchi J: MDM2 protein confers the resistance of a human glioblastoma cell line to cisplatin-induced apoptosis.Oncogene 18: 2001–2006, 1995
Pollack IF, Hamilton RL, Finkelstein SD, Campbell JW, Martinez AJ, Sherwin RN, Bozik ME, Gollin SM: The relationship between TP53 mutations and overexpression of p53 and prognosis in malignant gliomas of childhood. Cancer Res 57: 304–309,1997
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fountzilas, G., Karavelis, A., Capizzello, A. et al. Radiation and Concomitant Weekly Administration of Paclitaxel in Patients with Glioblastoma Multiforme. A Phase II Study. J Neurooncol 45, 159–165 (1999). https://doi.org/10.1023/A:1006386114104
Issue Date:
DOI: https://doi.org/10.1023/A:1006386114104