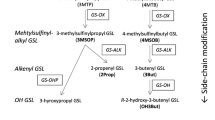
Abstract
Glucosinolates are known to elicit responses from Brassica herbivores in laboratory studies. To study their importance in interactions with herbivores in the field, glucosinolate profiles and levels of herbivory were ascertained for wild cabbage plants growing in four neighboring populations in the UK. Glucosinolate profiles differed between plant populations, but not between different habitats within populations. Within habitats, there was no link between individual plant glucosinolate profiles and herbivory by Pieris spp., slugs and snails, flea beetles or aphids. Plants attacked by the micromoth, Selania leplastriana, contained higher levels of 2–hydroxy-3–butenylglucosinolate and 3–indolylmethylglucosinolate than plants within the same population that were not attacked. It is concluded that the differences in glucosinolate profiles between the plant populations are unlikely to be due to differential selection pressures from herbivores feeding on the mature plants over the two years studies.
Similar content being viewed by others
REFERENCES
Bartlet, E., Kiddle, G., Williams, I., and Wallsgrove, R. 1999. Wound-induced increases in the glucosinolate content of oilseed rape and their effect on subsequent herbivory by a crucifer specialist. Entomol. Exp. Appl. 91:164–167.
Bennet, R. N., and Wallsgrove, R. M. 1994. Tansley review no. 72. Secondary metabolites in plant defense mechanisms. New Phytol. 127:617–633.
Bradley, J. D., Tremewan, W. G., and Smith, A. 1979. British Tortricoid Moths: Tortricidae: Olethreutinae. The Ray Society, London.
Broadway, R. M., and Colvin, A. A. 1992. Influence of cabbage proteinase inhibitors in situ on the growth of larval Trichoplusia ni and Pieris rapae. J. Chem. Ecol. 18:1009–1024.
Chew, F. S. 1988. Biological effects of glucosinolates, pp. 155–181, in H. Cutler (ed.). Biologically Active Natural Products. American Chemical Society, Washington, DC.
Chew, F. S., and Courtney, S. P. 1991. Plant apparency and evolutionary escape from insect herbivory. Am. Nat. 138:729–750.
Cook, R. T., Bailey, S. E. R., and McCrohan, C. R. 1997. The potential for common weeds to reduce slug damage to winter wheat: Laboratory and field studies. J. Appl. Ecol. 34:79–87.
Courtney, S. P. 1982. Coevolution of pierid butterflies and their cruciferous foodplants IV. Crucifer apparency and Anthocharis cardamines (L.) oviposition. Oecologia 52:258–265.
Giamoustaris, A., and Mithen, R. 1995. The effect of modifying the glucosinolate content of oilseed rape (Brassica napus ssp. olerifera) on its interaction with specialist and generalist pests. Ann. Appl. Biol. 126:347–363.
Giamoustaris, A., and Mithen, R. 1996a. Genetics of aliphatic glucosinolates. IV. Side-chain modification in Brassica oleracea. Theor. Appl. Genet. 93:1006–1010.
Giamoustaris, A., and Mithen, R. 1996b. The effect of flower colour and glucosinolates on the interaction between oilseed rape and pollen beetles. Entomol. Exp. Appl. 80:206–208.
Glen, D. M., Jones, H., and Fieldsend, J. K. 1990. Damage to oilseed rape seedlings by the field slug Deroceras reticulatum in relation to glucosinolate concentration. Ann. Appl. Biol. 117:197–207.
Gurevitch, J., and Collins, S. L. 1994. Experimental manipulation of natural plant communities. Trends Ecol. Evol. 9:94–97.
Herms, D. A., and Mattson, W. J. 1992. The dilemma of plants: To grow or defend. Q. Rev. Biol. 67:283–335.
Hogge, L. R., Reed, D. W., and Underhill, E. W. 1988. The identification of desulphoglucosinolates using thermospray liquid chromatography/mass spectrometry. J. Chromatogr. Sci. 26:348–351.
Huang, X., and Renwick, J. A. A. 1994. Relative activities of glucosinolates as oviposition stimulants for Pieris rapae and P. napi oleracea. J. Chem. Ecol. 20:1025–1037.
Jones, R. E. 1977. Movement patterns and egg distribution in cabbage butterflies. J. Anim. Ecol. 46:195–212.
Kareiva, P. 1982. Experimental and mathematical analyses of herbivore movement: Quantifying the influence of plant spacing and quality on foraging discrimination. Ecol. Monogr. 53:261–282.
Kerney, M. P., Cameron, R. A. D., and Riley, G. 1987. Land Snails of Britain and North-West Europe. HarperCollins, Hong Kong.
Kirk, W. D. J., and Gray, M. 1992. Insect on Cabbages and Oilseed Rape. The Richmond Publishing Co. Ltd., Slough, UK.
Koritsas, V. M., Lewis, J. A., and Fenwick, G. R. 1989. Accumulation of indole glucosinolates in Psylliodes chrysocephala L.-infested, or-damaged tissues of oilseed rape (Brassica napus L.). Experientia 45:493–495.
Louda, S., and Mole, S. 1991. Glucosinolates: Chemistry and ecology, pp. 123–164, in G. A. Rosenthal and M. R. Berenbaum (eds.). Herbivores: Their Interactions with Secondary Plant Metabolites. Academic Press, New York.
Louda, S. M., and Rodman, J. E. 1983. Ecological patterns in the glucosinolate content of a native mustard, Cardamine cordifolia, in the Rocky mountains. J. Chem. Ecol. 9:397–423.
Mauricio, R., and Rausher, M. D. 1997. Experimental manipulation of putative selective agents provides evidence for the role of natural enemies in the evolution of plant defense. Evolution 51:1435–1444.
Milford, G. F. J., Porter, A. J. R., Fieldsend, J. K., Miller, C. A., Leach, J. E., and Williams, I. H. 1989. Glucosinolates in oilseed rape (Brassica napus) and the incidence of pollen beetles (Meligethes aeneus). Ann. Appl. Biol. 115::375–380.
Mithen, R., Raybould, A. F., and Giamoustaris, A. 1995. Divergent selection for secondary metabolites between wild populations of Brassica oleracea and its implication for plant-herbivore interactions. Heredity 75:472–484.
Moens, R., Couvreur, R., and Cors, F. 1992. Influence de la teneur en glucosinolates des variétés de colza d'hiver sur les dégâts de limaces. Bull. Rech. Agron. Gemblaux 27:289–307.
Moyes, C. L. 1997. The role of glucosinolate production in populations of the wild cabbage. PhD dissertation. University of Liverpool, Liverpool, UK.
Nottingham, S. F., Hardie, J., Dawson, G. W., Hick, A. J., Picektt, J. A., Wadhams, L. J., and Woodcock, C. M. 1991. Behavioral and electrophysiological responses of aphids to host and nonhost plant volatiles. J. Chem. Ecol. 17:1231–1242.
Pivnick, K. A., Lamb, R. J., and Reed, D. 1992. Response of flea beetles, Phyllotreta spp., to mustard oils and nitriles in field trapping experiments. J. Chem. Ecol. 18:863–873.
Read, D. P., Feeny, P. P., and Root, R. B. 1970. Habitat selection by the aphid parasite Diaeretialla rapae (Hymenoptera: Braconidae) and hyperparasite Charips brassicae (Hymenoptera: Cynipidae). Can. Entomol. 102:1567–1578.
Renwick, J. A. A. 1988. Comparative mechanisms of host selection by insects attacking pine trees and crucifers, pp. 303–316, in K. C. Spencer (ed.). Chemical Mediation on Coevolution. Harcourt Brace Jovanovitch, San Diego.
Renwick, J. A. A., Radke, C. D., Sachdev-Gupta, K., and StÄdler, E. 1992. Leaf surface chemicals stimulating oviposition by Pieris rapae (Lepidoptea: pieridae) on cabbage. Chemoecology 3:33–38.
Rodman, J. E., nad Chew, F. S. 1980. Phytochemical correlates of herbivory in a community of native and naturalized Cruciferae. Biochem. Syst. Ecol. 8:43–50.
RÜcker, B., and RÖbbelson, G. 1994. Inheritance of total and individual glucosinolate contents in seeds of winter oilseed rape (Brassica napus L.). Plant Breed. 113:206–216.
StÄdler, E., Renwick, J. A. A., Radke, C. D., and Sachdev-Gupta, K. 1995. Tarsal contact chemoreceptor response to glucosinolates and cardenolides mediating oviposition in Pieris rapae. Physiol. Entomol. 20:175–187.
Teyke, T. 1995. Food attraction conditioning in the snail, Helix pomatia. J. Comp. Physiol. 177:409–414.
Toroser, D., Thormann, C. E., Osborn, T. C., and Mithen, R. 1995. RFLP mapping of quantitative trait loci controlling seed aliphatic glucosinolate content in oilseed rape (Brassica napus L.). Theor. Appl. Genet. 91:802–808.
Traynier, R. M. M. 1986. Visual learning in assays of sinigrin solution as an oviposition releaser for the cabbage butterfly, Pieris rapae. Entomol. Exp. Appl. 40:25–33.
Traynier, R. M. M., and Truscott, R. G. W. 1991. Potent natural egg-laying stimulant for the cabbage butterfly, Pieris rapae. J. Chem. Ecol. 17:1371–1380.
Usher, B. F., and Feeny, P. 1983. Atypical secondary compounds in the family Cruciferae: Tests for toxicity to Pieris rapae, an adapted crucifer-feeding insect. Entomol. Exp. Appl. 34:257–262.
Van Loon, J. J. A., Blaakmeer, A., Griepink, F. C., Van Beek, T. A., Schooven, L. M., and De Groot, A. 1992. Leaf surface compound from Brassica oleracea (Cruciferae) induces oviposition by Pieris brassicae (Lepidoptera: Pieridae). Chemoecology 3:39–44.
Vincent, C., and Stewart, R. K. 1984. Effect of allyl isothiocyanate on field behaviour of cruciferfeeding flea beetles (Coleoptera: Chrysomelidae). J. Chem. Ecol. 10:33–39.
Visser, J. H., Piron, P. G. M., and Hardie, J. 1996. The aphids' peripheral perception of plant volatiles. Entomol. Exp. Appl. 80:35–38.
Williams, I. H. 1989. Pest incidence on single low and double low oilseed rape cultivars. Aspects Appl. Biol. 23:277.
Williams, M. R., and Abbott, I. 1991. Quantifying average defoliation using leaf level measurements. Ecology 72:1510–1511.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moyes, C.L., Collin, H.A., Britton, G. et al. Glucosinolates and Differential Herbivory in Wild Populations of Brassica oleracea. J Chem Ecol 26, 2625–2641 (2000). https://doi.org/10.1023/A:1005549115751
Issue Date:
DOI: https://doi.org/10.1023/A:1005549115751