Abstract
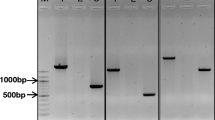
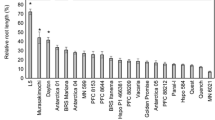
We have made use of a genetic approach to develop homozygous, near-isogenic germplasm for investigating aluminium (Al) resistance in Triticum aestivum L. A conventional backcross program was used to transfer Al resistance from the Al-resistant cultivar, Maringa, to a locally-adapted, Al-sensitive cultivar, Katepwa. At the third backcross stage, a single, resistant isoline (Alikat = Katepwa*3/Maringa) was chosen on the basis of superior root growth after 14 days of exposure to a broad range of Al concentrations (0 to 600 µM). Genetic analysis of doubled-haploid lines (DH) developed from this isoline suggested that resistance is controlled by a single dominant gene. Crosses between DH Alikat and DH Katepwa yielded an Al-resistant F1 population. Backcrossing this F1 population to DH Katepwa produced a population which segregated 1:1 for Al resistance, while selfing produced a population segregating 3 : 1 for Al resistance. Under conditions of Al stress, Al-resistant F2 plants released a suite of novel low molecular weight polypeptides into the rhizosphere. One of these polypeptides (23 kD) shows substantive Al-binding capacity and segregates with the resistant phenotype. While the precise mechanisms that mediate Al resistance are still unknown, this research has provided support for a possible role of the 23 kD exudate polypeptide in mediating resistance to Al. To more fully understand the role that this polypeptide plays in Al-resistance, we are attempting to clone this gene from microsequence data obtained from purified protein.
Similar content being viewed by others
References
Archambault, D J, Zhang G, and Taylor G J 1996a A comparison of the kinetics of aluminum (Al) uptake and distribution in roots of wheat (Triticum aestivumL.) using different aluminum sources. A revision of the operational definition of symplastic Al. Physiol. Plant. 98, 578-586.
Archambault, D J, Zhang G, and Taylor G J 1996b Accumulation of aluminum in root mucilage of an Al-resistant and an Al-sensitive cultivar of wheat (Triticum aestivumL.). Plant Physiol. 112, 1471-1478.
Archambault, D J, Zhang G, and Taylor G J 1997 Spatial variation in the kinetics of aluminum (Al) uptake in root segments of wheat (Triticum aestivum) exhibiting differential resistance to Al. Further evidence for metabolism-dependent exclusion of Al. J. Plant Physiol. (in press).
Basu, U, Godbold D, and Taylor G J. 1994a Aluminum resistance in Triticum aestivumis associated with enhanced exudation of malate. J. Plant Physiol. 144, 747-753.
Basu, U, Basu A, and Taylor G J 1994b Differential exudation of polypeptides by roots of aluminum-resistant and aluminum-sensitive cultivars of Triticum aestivumL. in response to aluminum stress. Plant Physiol. 106, 151-158.
Delhaize, E, Ryan P R, and Randall P J 1993 Aluminum tolerance in wheat (Triticum aestivumL.). 2. Aluminum-stimulated excretion of malic acid from root apices. Plant Physiol. 103, 695-702.
Gustafson, J P and Ross K 1990 Control of alien gene expression for aluminium tolerance in wheat. Genome 33, 9-12.
Lacmmli U K 1970 Cleavage of the structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680-685.
McKendry, A L. Tangue D N, and Somers D J 1996 Aluminum tolerance of 1BL.1RS and 1AL.1RS near-isolines of soft red winter wheat. Crop Sci. 34, 987-990.
Miyasaka S C, Buta J G, Howell R K, Foy C D 1991 Mechanisms of aluminum tolerance in snapbeans. Root exudation of citric acid. Plant Physiol. 96, 737-743.
Ownby, J D 1993 Mechanisms of reaction of hematoxylin with aluminium-treated wheat roots. Physiol. Plant. 87, 371-380.
Putterill J, Gardner R 1988 Proteins with the potential to protect plants from Al3+toxicity. Biochim. Biophys. Acta. 964, 137-145.
Ryan, P R, DiTomaso J M, and Kochian L V 1993 Aluminium toxicity in roots: an investigation of spatial sensitivity and the role of the root cap. J. Exp. Bot. 44, 437-446.
Taylor G J 1989 Multiple metal stress in Triticum aestivum. Differentiation between additive, multiplicative, antagonistic, and synergistic effects. Can. J. Bot. 67, 2272-2276.
Taylor G J 1995 Overcoming barriers to understanding the cellular basis of aluminum resistance. Plant and Soil 171, 89-103.
Tremaine M, and Miller R H 1983 Extracellular polysaccharides as a potential aluminum tolerance mechanism of Rhizobia. InProceedings of the 9thAmerican Rhizobium Conference. Boyce Thompson Institute, Ithaca, N.Y. Abstract L1.
Von Uexkull H R and Mutert E 1995 Global extent, development and economic impact of acid soils. Plant and Soil 171, 1-15.
Zhang, G, Hoddinott J, and Taylor G J 1994 Characterization of 1,3-b-D-glucan (callose) synthesis in roots of Triticum aestivumin response to aluminum toxicity. J. Plant Physiol. 144, 229-234.
Zhang, G and Taylor G J 1991 Effects of biological inhibitors on kinetics of aluminium uptake by excised roots and purified cell wall material of aluminium-tolerant and aluminium-sensitive cultivars of Triticum aestivumL. J. Plant Physiol. 138, 533-539.
Zhang, G and Taylor G J 1989 Kinetics of aluminum uptake by excised roots of aluminum-tolerant and aluminum-sensitive cultivars of Triticum aestivumL. Plant Physiol. 91, 1094-1099.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Basu, U., McDonald-Stephens, J.L., Archambault, D.J. et al. Genetic and physiological analysis of doubled-haploid, aluminium-resistant lines of wheat provide evidence for the involvement of a 23 kD, root exudate polypeptide in mediating resistance. Plant and Soil 196, 283–288 (1997). https://doi.org/10.1023/A:1004274629441
Issue Date:
DOI: https://doi.org/10.1023/A:1004274629441