Abstract
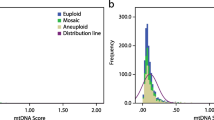
The aim of this study is to explore the relationship between mitochondrial DNA (mtDNA) copy number and embryo implantation potential in in vitro fertilization (IVF). A retrospective study of 319 blastocysts from patients undergoing preimplantation genetic testing (PGT) at Reproductive Medicine Center in Tongji Hospital from January 2016 to February 2018 was conducted. We used multiple annealing- and looping-based amplification cycles (MALBAC) technology to amplify the genetic materials from the trophectoderm cells of blastocysts, and next-generation sequencing (NGS) technology to test mitochondrial DNA copy number. Box-Cox transformation was introduced to eliminate the skewness distribution of mtDNA copy number, and the transformed data were defined as adjusted mtDNA. Subsequently, associations between adjusted mtDNA and the clinical characteristics of patients were assessed by univariate analysis and multiple linear regression. In addition, Gaussian Naive Bayes classifier was also used to predict pregnancy outcomes. We observed that only antral follicle count (AFC) was significantly associated with adjusted mtDNA without the influence of multicollinearity. What’s more, the distribution of the adjusted mtDNA of blastocysts resulting in live birth was more concentrated than that of others. The area under the curve (AUC) of the prediction model that combined adjusted mtDNA with other clinical characteristics of patients was up to 0.81, higher than that excluded adjusted mtDNA. Among patient clinical characteristics, AFC was significantly associated with adjusted mtDNA. Mitochondrial DNA copy number may help to optimize the pregnancy outcome prediction in IVF.



Similar content being viewed by others
References
Ishihara O, Adamson GD, Dyer S, de Mouzon J, Nygren KG, Sullivan EA, et al. International committee for monitoring assisted reproductive technologies: world report on assisted reproductive technologies, 2007. Fertil Steril. 2015;103:402–13.e11.
Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of Embryology. The Istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Hum Reprod. 2011;26:1270–83.
Forman EJ, Hong KH, Ferry KM, Tao X, Taylor D, Levy B, et al. In vitro fertilization with single euploid blastocyst transfer: a randomized controlled trial. Fertil Steril. 2013;100:100–7.e1.
Rubio C, Bellver J, Rodrigo L, Castillón G, Guillén A, Vidal C, et al. In vitro fertilization with preimplantation genetic diagnosis for aneuploidies in advanced maternal age: a randomized, controlled study. Fertil Steril. 2017;107:1122–9.
Scott RT, Upham KM, Forman EJ, Hong KH, Scott KL, Taylor D, et al. Blastocyst biopsy with comprehensive chromosome screening and fresh embryo transfer significantly increases in vitro fertilization implantation and delivery rates: a randomized controlled trial. Fertil Steril. 2013;100:697–703.
Simon AL, Kiehl M, Fischer E, Proctor JG, Bush MR, Givens C, et al. Pregnancy outcomes from more than 1,800 in vitro fertilization cycles with the use of 24-chromosome single-nucleotide polymorphism-based preimplantation genetic testing for aneuploidy. Fertil Steril. 2018;110:113–21.
Fragouli E, McCaffrey C, Ravichandran K, Spath K, Grifo JA, Munné S, et al. Clinical implications of mitochondrial DNA quantification on pregnancy outcomes: a blinded prospective non-selection study. Hum Reprod. 2017;32:2340–7.
Fragouli E, Spath K, Alfarawati S, Kaper F, Craig A, Michel CE, et al. Altered levels of mitochondrial DNA are associated with female age, aneuploidy, and provide an independent measure of embryonic implantation potential. PLoS Genet. 2015;11:e1005241.
May-Panloup P, Chrétien MF, Savagner F, Vasseur C, Jean M, Malthièry Y, et al. Increased sperm mitochondrial DNA content in male infertility. Hum Reprod. 2003;18:550–6.
van Blerkom J, Davis PW, Lee J. ATP content of human oocytes and developmental potential and outcome after in-vitro fertilization and embryo transfer. Hum Reprod. 1995;10:415–24.
van Blerkom J. Mitochondria in human oogenesis and preimplantation embryogenesis: engines of metabolism, ionic regulation and developmental competence. Reproduction. 2004;128:269–80.
de Los Santos MJ, Diez Juan A, Mifsud A, Mercader A, Meseguer M, Rubio C, et al. Variables associated with mitochondrial copy number in human blastocysts: what can we learn from trophectoderm biopsies? Fertil Steril. 2018;109:110–7.
Ho JR, Arrach N, Rhodes-Long K, Salem W, McGinnis LK, Chung K, et al. Blastulation timing is associated with differential mitochondrial content in euploid embryos. J Assist Reprod Genet. 2018;35:711–20.
Tan Y, Yin X, Zhang S, Jiang H, Tan K, Li J, et al. Clinical outcome of preimplantation genetic diagnosis and screening using next generation sequencing. GigaScience. 2014;3:30.
Victor AR, Brake AJ, Tyndall JC, Griffin DK, Zouves CG, Barnes FL, et al. Accurate quantitation of mitochondrial DNA reveals uniform levels in human blastocysts irrespective of ploidy, age, or implantation potential. Fertil Steril. 2017;107:34–42.e3.
Diez-Juan A, Rubio C, Marin C, Martinez S, Al-Asmar N, Riboldi M, et al. Mitochondrial DNA content as a viability score in human euploid embryos: less is better. Fertil Steril. 2015;104:534–41.e1.
Treff NR, Zhan Y, Tao X, Olcha M, Han M, Rajchel J, et al. Levels of trophectoderm mitochondrial DNA do not predict the reproductive potential of sibling embryos. Hum Reprod. 2017;32:954–62.
Humaidan P, Kristensen SG, Coetzee K. Mitochondrial DNA, a new biomarker of embryonic implantation potential: fact or fiction? Fertil Steril. 2018;109:61–2.
Wells D. Mitochondrial DNA quantity as a biomarker for blastocyst implantation potential. Fertil Steril. 2017;108:742–7.
Zegers-Hochschild F, Adamson GD, Dyer S, Racowsky C, de Mouzon J, Sokol R, et al. The International Glossary on Infertility and Fertility Care, 2017. Hum Reprod. 2017;32:1786–801.
Zong C, Lu S, Chapman AR, Xie XS. Genome-wide detection of single-nucleotide and copy-number variations of a single human cell. Science. 2012;338:1622–6.
Wells D, Ravichandran K, McCaffrey C, Grifo J, Morales A, Perloe M, et al. Reply: mitochondrial DNA quantification-the devil in the detail. Hum Reprod. 2017;32:2150–1.
Lee Y, Chen C, Lin S, Lin Y, Tzeng C. Adjusted mitochondrial DNA quantification in human embryos may not be applicable as a biomarker of implantation potential. J Assist Reprod Genet. 2019;36(9):1855–65.
Huang B, Qian K, Li Z, Yue J, Yang W, Zhu G, et al. Neonatal outcomes after early rescue intracytoplasmic sperm injection: an analysis of a 5-year period. Fertil Steril. 2015;103:1432–7.e1.
Jin L, Wang M, Yue J, Zhu GJ, Zhang B. Association between TSH level and pregnancy outcomes in euthyroid women undergoing IVF/ICSI: a retrospective study and meta-analysis. Curr Med Sci. 2019;39:631–7.
Hou Y, Fan W, Yan L, Li R, Lian Y, Huang J, et al. Genome analyses of single human oocytes. Cell. 2013;155:1492–506.
Shang W, Zhang Y, Shu M, Wang W, Ren L, Chen F, et al. Comprehensive chromosomal and mitochondrial copy number profiling in human IVF embryos. Reprod BioMed Online. 2018;36:67–74.
Yamaguchi Y, Maruo K, Partlett C, Riley RD. A random effects meta-analysis model with Box-Cox transformation. BMC Med Res Methodol. 2017;17:109.
Joshi D, Mishra A, Anand S. A naïve Gaussian Bayes classifier for detection of mental activity in gait signature. Comput Methods Biomech Biomed Engin. 2012;15:411–6.
Ravichandran K, McCaffrey C, Grifo J, Morales A, Perloe M, Munne S, et al. Mitochondrial DNA quantification as a tool for embryo viability assessment: retrospective analysis of data from single euploid blastocyst transfers. Hum Reprod. 2017;32:1282–92.
Lu S, Zong C, Fan W, Yang M, Li J, Chapman AR, et al. Probing meiotic recombination and aneuploidy of single sperm cells by whole-genome sequencing. Science. 2012;338:1627–30.
van Blerkom J. Mitochondrial function in the human oocyte and embryo and their role in developmental competence. Mitochondrion. 2011;11:797–813.
Motta PM, Nottola SA, Makabe S, Heyn R. Mitochondrial morphology in human fetal and adult female germ cells. Hum Reprod. 2000;15:129–47.
Sathananthan AH, Trounson AO. Mitochondrial morphology during preimplantational human embryogenesis. Hum Reprod. 2000;15:148–59.
Lin DP, Huang CC, Wu HM, Cheng TC, Chen CI, Lee MS. Comparison of mitochondrial DNA contents in human embryos with good or poor morphology at the 8-cell stage. Fertil Steril. 2004;81:73–9.
Houghton FD. Energy metabolism of the inner cell mass and trophectoderm of the mouse blastocyst. Differentiation. 2006;74:11–8.
Acknowledgments
We would like to express heartfelt gratitude to Yue Sun and his colleagues from Yikon Genomics for helping us process the data of the mitochondrial DNA during the MALBAC-NGS process. We also want to thank Yuqi Cui from Huazhong University of Science and Technology in China for helping in developing our machine learning algorithms.
Funding
This work was supported by the National Key Research and Development Project (2018YFC1002103) and the Chinese Medical Association (16020520668) as well as the Natural Science Foundation of Hubei Province (2017CFB752).
Author information
Authors and Affiliations
Contributions
LJ and LZ conceived the study; JL and MW wrote the paper; ZF and ZL collected data; JL, MW, and FZ analyzed data.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no conflict of interest.
Ethics Approval and Consent to Participate
The study was approved by the Ethics of Committee of Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology. All the participants have signed the consent forms.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 15 kb)
Rights and permissions
About this article
Cite this article
Zhu, L., Li, J., Wang, M. et al. Normalized Mitochondrial DNA Copy Number Can Optimize Pregnancy Outcome Prediction in IVF. Reprod. Sci. 28, 1439–1446 (2021). https://doi.org/10.1007/s43032-020-00422-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00422-0