Abstract
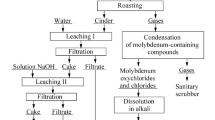
Extraction and processing constitute an important area in the metallurgy of molybdenum. Molybdenum trioxide (MoO3) production is one of the most important stages in molybdenum production. In this study, the effects of different furnace types on MoO3 production were investigated by using molybdenum concentrate as a raw material obtained during the enrichment of copper ores. Preliminary experiments have been made using a chamber type furnace, and a transition to a pilot-scale has been carried out with rotary kiln experiments. The effects of a different time and atmospheric conditions on MoO3 roasting kinetics were investigated by experimental studies conducted in the temperature range of 500–650°C. It was determined that the optimum temperature and time for sulfur (S) removal was 625°C and 180 min in the studies carried out in the rotary kiln, and it was determined that 0.61% S content was reached with an average removal efficiency of 98%. The activation energy in these sets was calculated as 89.519 kJ/mol.



















Similar content being viewed by others
Data Availability
Not applicable
Code Availability
Not applicable
References
Kim B-S, Jha MK, Jeong J, Lee J-C (2008) Leaching of impurities for the up-gradation of molybdenum oxide and cementation of copper by scrap iron. Int J Miner Process 88:7–12. https://doi.org/10.1016/j.minpro.2008.04.002
Öncel L (2020) Production of ferromolybdenum from mill scale via aluminothermic process. Sinop Üniv Fen Bilim Derg 5:64–76. https://doi.org/10.33484/sinopfbd.724720
Sheybani K, Paydar MH, Shariat MH (2019) Effect of mechanical activation on aluminothermic reduction of molybdenum trioxide. Int J Refract Met Hard Mater 82:245–254. https://doi.org/10.1016/j.ijrmhm.2019.04.015
Wang L, Zhang GH, Chou KC (2016) Study on oxidation mechanism and kinetics of MoO2 to MoO3 in air atmosphere. Int J Refract Met Hard Mater 57:115–124. https://doi.org/10.1016/j.ijrmhm.2016.03.001
Wan YP, Fincke JR, Jiang XY, Sampath S, Prasad V, Herman H (2001) Modeling of oxidation of molybdenum particles during plasma spray deposition. Metall Mater Trans B Process Metall Mater Process Sci 32:475–481. https://doi.org/10.1007/s11663-001-0033-7
Habashi F (1997) Handbook of Extractive Metallurgy III. WILEY-VCH, Weinheim
Utigard T (2009) Oxidation mechanism of molybdenite concentrate. Metall Mater Trans B Process Metall Mater Process Sci 40:490–496. https://doi.org/10.1007/s11663-009-9245-z
Mchugh LF, Shekhter LN, Lessard JD, Gribbin DG, Cankaya-Yalcin E (2015) Sulfide oxidation process for production of molybdenum oxides from molybdenite
Wang L, Zhang G, Dang J, Chou K (2015) Oxidation roasting of molybdenite concentrate. Trans Nonferrous Metals Soc China 25:4167–4174. https://doi.org/10.1016/S1003-6326(15)64067-5
Kim B-S, Lee H-I, Choi Y-Y, Kim S (2009) Kinetics of the oxidative roasting of low grade mongolian molybdenite concentrate. Mater Trans 50:2669–2674. https://doi.org/10.2320/matertrans.M2009197
Živković ŽD, Šesták J (1998) Kinetics and mechanism of the oxidation of molybdenum sulphide. J Therm Anal Calorim 53:263–267. https://doi.org/10.1023/A:1010108813595
Marin T, Utigard T, Hernandez C (2009) Roasting kinetics of molybdenite concentrates. Can Metall Q 48:73–80. https://doi.org/10.1179/cmq.2009.48.1.73
Blanco E, Sohn HY, Han G, Hakobyan KY (2007) The kinetics of oxidation of molybdenite concentrate by water vapor. Metall Mater Trans B Process Metall Mater Process Sci 38:689–693. https://doi.org/10.1007/s11663-006-9001-6
Gupta CK (2017) Extractive Metallurgy of Molybdenum. Routledge, New York
Kan S, Benzeşik K, Sönmez MŞ, Yücel O (2018) 19th International Metallurgy and Materials Congress. In: Roasting of molybdenite concentrates in pilot scale rotary furnace. Istanbul, pp 128–132
Benzeşik K, Sönmez MŞ, Yücel O (2016) 18th International Metallurgy and Materials Congress. In: Production of technical-grade MoO3 from MoS2 concentrate. Istanbul, pp 592–596
Kan S, Buğdaycı M, Benzeşik K, Yücel O (2017) XIV International Symposium on Self-Propagating High Temperature Synthesis. In: Production of ferro-molybdenum from domestic resources via metallothermic process. Tbilisi, pp 107–109
Hou L, Yu Q, Wang K, Qin Q, Wei M, Yang F (2018) Oxidation kinetics of YBaCo4O7+δ and substituted oxygen carriers. R Soc Open Sci 5:180150. https://doi.org/10.1098/rsos.180150
Khawam A, Flanagan DR (2006) Solid-state kinetic models: basics and mathematical fundamentals. J Phys Chem B 110:17315–17328. https://doi.org/10.1021/jp062746a
Taşyürek K, Buğdaycı M, Yücel O (2018) Reduction conditions of metallic calcium from magnesium production residues. Metals (Basel) 8:383. https://doi.org/10.3390/met8060383
Author information
Authors and Affiliations
Contributions
Not applicable
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kan, S., Benzeşik, K., Odabaş, Ö.C. et al. Investigation of Molybdenite Concentrate Roasting in Chamber and Rotary Furnaces. Mining, Metallurgy & Exploration 38, 1597–1608 (2021). https://doi.org/10.1007/s42461-021-00429-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42461-021-00429-4