Abstract
Typical catalyst inks in proton exchange membrane fuel cells (PEMFCs) are composed of a catalyst, its support, an ionomer and a solvent and are used with solution processing approaches to manufacture conventional catalyst layers (CLs). Because of this, catalyst ink formulation and deposition processes are closely related to CL structure and performance. However, catalyst inks with ideal rheology and optimized electrochemical performances remain lacking in the large-scale application of PEMFCs. To address this, this review will summarize current progress in the formulation, characterization, modeling and deposition of catalyst inks. In addition, this review will highlight recent advancements in catalyst ink materials and discuss corresponding complex interactions. This review will also present various catalyst ink dispersion methods with insights into their stability and introduce the application of small-angle scattering and cryogenic transmission electron microscopy (cryo-TEM) technologies in the characterization of catalyst ink microstructures. Finally, recent studies in the kinetic modeling and deposition of catalyst inks will be analyzed.
Graphic Abstract
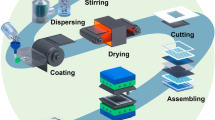
The formulation and the deposition process of catalyst inks determine the formation of catalyst. The interaction between the components of the catalyst ink governs the microstructure and processability of the ink, thereby affecting the microstructure and performance of the CL.















Similar content being viewed by others
References
Litster, S., McLean, G.: PEM fuel cell electrodes. J. Power Sources 130, 61–76 (2004). https://doi.org/10.1016/j.jpowsour.2003.12.055
Majlan, E.H., Rohendi, D., Daud, W.R.W., et al.: Electrode for proton exchange membrane fuel cells: a review. Renew. Sust. Energ. Rev. 89, 117–134 (2018). https://doi.org/10.1016/j.rser.2018.03.007
Karan, K.: PEFC catalyst layer: recent advances in materials, microstructural characterization, and modeling. Curr. Opin. Electrochem. 5, 27–35 (2017). https://doi.org/10.1016/j.coelec.2017.08.018
Huang, J., Li, Z., Zhang, J.B.: Review of characterization and modeling of polymer electrolyte fuel cell catalyst layer: the blessing and curse of ionomer. Front. Energy 11, 334–364 (2017). https://doi.org/10.1007/s11708-017-0490-6
Hou, Y., Deng, H., Pan, F., et al.: Pore-scale investigation of catalyst layer ingredient and structure effect in proton exchange membrane fuel cell. Appl. Energ. 253, 113561 (2019). https://doi.org/10.1016/j.apenergy.2019.113561
Kurihara, Y., Mabuchi, T., Tokumasu, T.: Molecular analysis of structural effect of ionomer on oxygen permeation properties in PEFC. J. Electrochem. Soc. 164, F628–F637 (2017). https://doi.org/10.1149/2.1301706jes
Wu, H.W.: A review of recent development: transport and performance modeling of PEM fuel cells. Appl. Energy 165, 81–106 (2016). https://doi.org/10.1016/j.apenergy.2015.12.075
Holdcroft, S.: Fuel cell catalyst layers: a polymer science perspective. Chem. Mater. 26, 381–393 (2014). https://doi.org/10.1021/cm401445h
Ioroi, T., Siroma, Z., Yamazaki, S.I., et al.: Electrocatalysts for PEM fuel cells. Adv. Energy Mater. 9, 1801284 (2019). https://doi.org/10.1002/aenm.201801284
Dixit, M.B., Harkey, B.A., Shen, F.Y., et al.: Catalyst layer ink interactions that affect coatability. J. Electrochem. Soc. 165, F264–F271 (2018). https://doi.org/10.1149/2.0191805jes
So, M., Ohnishi, T., Park, K., et al.: The effect of solvent and ionomer on agglomeration in fuel cell catalyst inks: simulation by the discrete element method. Int. J. Hydrog. Energy 44, 28984–28995 (2019). https://doi.org/10.1016/j.ijhydene.2019.09.012
Mashio, T., Ohma, A., Tokumasu, T.: Molecular dynamics study of ionomer adsorption at a carbon surface in catalyst ink. Electrochim. Acta 202, 14–23 (2016). https://doi.org/10.1016/j.electacta.2016.04.004
Sassin, M.B., Garsany, Y., Gould, B.D., et al.: Fabrication method for laboratory-scale high-performance membrane electrode assemblies for fuel cells. Anal. Chem. 89, 511–518 (2017). https://doi.org/10.1021/acs.analchem.6b03005
Liu, R.L., Zhou, W., Wan, L.Y., et al.: Electrostatic spraying of membrane electrode for proton exchange membrane fuel cell. Curr. Appl. Phys. 20, 11–17 (2020). https://doi.org/10.1016/j.cap.2019.09.016
Chen, G.H., Kelly, D., Shine, A., et al.: Electrospinning deposition of poly(acrylic acid): platinum/carbon catalyst ink to enhance polymer electrolyte membrane fuel cell performance. MRS Commun. 9, 1343–1348 (2019). https://doi.org/10.1557/mrc.2019.144
Ding, X.Y., Didari, S., Fuller, T.F., et al.: Membrane electrode assembly fabrication process for directly coating catalyzed gas diffusion layers. J. Electrochem. Soc. 159, B746–B753 (2012). https://doi.org/10.1149/2.103206jes
Gomes Bezerra, C.A., Deiner, L.J., Tremiliosi-Filho, G.: Unexpected performance of inkjet-printed membrane electrode assemblies for proton exchange membrane fuel cells. Adv. Eng. Mater. 21, 1900703 (2019). https://doi.org/10.1002/adem.201900703
Park, I.S., Li, W., Manthiram, A.: Fabrication of catalyst-coated membrane-electrode assemblies by doctor blade method and their performance in fuel cells. J. Power Sources 195, 7078–7082 (2010). https://doi.org/10.1016/j.jpowsour.2010.05.004
Wang, W.T., Chen, S.Q., Li, J.J., et al.: Fabrication of catalyst coated membrane with screen printing method in a proton exchange membrane fuel cell. Int. J. Hydrog. Energy 40, 4649–4658 (2015). https://doi.org/10.1016/j.ijhydene.2015.02.027
Xiong, L., Manthiram, A.: High performance membrane-electrode assemblies with ultra-low Pt loading for proton exchange membrane fuel cells. Electrochim. Acta 50, 3200–3204 (2005). https://doi.org/10.1016/j.electacta.2004.11.049
Mauger, S.A., Neyerlin, K.C., Yang-Neyerlin, A.C., et al.: Gravure coating for roll-to-roll manufacturing of proton-exchange-membrane fuel cell catalyst layers. J. Electrochem. Soc. 165, F1012–F1018 (2018). https://doi.org/10.1149/2.0091813jes
Banham, D., Ye, S.: Current status and future development of catalyst materials and catalyst layers for proton exchange membrane fuel cells: an industrial perspective. ACS Energy Lett. 2, 629–638 (2017). https://doi.org/10.1021/acsenergylett.6b00644
Lin, R., Cai, X., Zeng, H., et al.: Stability of high-performance Pt-based catalysts for oxygen reduction reactions. Adv. Mater. 30, 1705332 (2018). https://doi.org/10.1002/adma.201705332
Mahata, A., Nair, A.S., Pathak, B.: Recent advancements in Pt-nanostructure-based electrocatalysts for the oxygen reduction reaction. Catal. Sci. Technol. 9, 4835–4863 (2019). https://doi.org/10.1039/C9CY00895K
Liu, M.L., Zhao, Z.P., Duan, X.F., et al.: Nanoscale structure design for high-performance Pt-based ORR catalysts. Adv. Mater. 31, 1802234 (2019). https://doi.org/10.1002/adma.201802234
Lang, P., Yuan, N.N., Jiang, Q.Q., et al.: Recent advances and prospects of metal-based catalysts for oxygen reduction reaction. Energy Technol. 8, 1900984 (2020). https://doi.org/10.1002/ente.201900984
Yano, H., Watanabe, M., Iiyama, A., et al.: Particle-size effect of Pt cathode catalysts on durability in fuel cells. Nano Energy 29, 323–333 (2016). https://doi.org/10.1016/j.nanoen.2016.02.016
Shinozaki, K., Morimoto, Y., Pivovar, B.S., et al.: Re-examination of the Pt particle size effect on the oxygen reduction reaction for ultrathin uniform Pt/C catalyst layers without influence from nafion. Electrochim. Acta 213, 783–790 (2016). https://doi.org/10.1016/j.electacta.2016.08.001
Antolini, E.: Structural parameters of supported fuel cell catalysts: the effect of particle size, inter-particle distance and metal loading on catalytic activity and fuel cell performance. Appl. Catal. B 181, 298–313 (2016). https://doi.org/10.1016/j.apcatb.2015.08.007
Zamel, N.: The catalyst layer and its dimensionality: a look into its ingredients and how to characterize their effects. J. Power Sources 309, 141–159 (2016). https://doi.org/10.1016/j.jpowsour.2016.01.091
Hou, J.B., Yang, M., Ke, C.C., et al.: Platinum-group-metal catalysts for proton exchange membrane fuel cells: from catalyst design to electrode structure optimization. EnergyChem 2, 100023 (2020). https://doi.org/10.1016/j.enchem.2019.100023
Beltrán-Gastélum, M., Salazar-Gastélum, M.I., Flores-Hernández, J.R., et al.: Pt-Au nanoparticles on graphene for oxygen reduction reaction: stability and performance on proton exchange membrane fuel cell. Energy 181, 1225–1234 (2019). https://doi.org/10.1016/j.energy.2019.06.033
Beltrán-Gastélum, M., Salazar-Gastélum, M.I., Félix-Navarro, R.M., et al.: Evaluation of PtAu/MWCNT (multiwalled carbon nanotubes) electrocatalyst performance as cathode of a proton exchange membrane fuel cell. Energy 109, 446–455 (2016). https://doi.org/10.1016/j.energy.2016.04.132
Wang, J., Xue, Q., Li, B., et al.: Preparation of a graphitized-carbon-supported PtNi octahedral catalyst and application in a proton-exchange membrane fuel cell. ACS Appl. Mater. Interfaces. 12, 7047–7056 (2020). https://doi.org/10.1021/acsami.9b17248
Taniguchi, K., Shinoda, K., Cuya Huaman, J.L., et al.: Designed synthesis of highly catalytic Ni–Pt nanoparticles for fuel cell applications. SN Appl. Sci. 1, 124 (2019). https://doi.org/10.1007/s42452-018-0133-5
Xiong, X.L., Chen, W.H., Wang, W., et al.: Pt–Pd nanodendrites as oxygen reduction catalyst in polymer–electrolyte–membrane fuel cell. Int. J. Hydrog. Energy 42, 25234–25243 (2017). https://doi.org/10.1016/j.ijhydene.2017.08.162
Bharti, A., Cheruvally, G.: Surfactant assisted synthesis of Pt–Pd/MWCNT and evaluation as cathode catalyst for proton exchange membrane fuel cell. Int. J. Hydrog. Energy 43, 14729–14741 (2018). https://doi.org/10.1016/j.ijhydene.2018.06.009
Wang, R.Y., Li, D.G., Maurya, S., et al.: Ultrafine Pt cluster and RuO2 heterojunction anode catalysts designed for ultra-low Pt-loading anion exchange membrane fuel cells. Nanoscale Horiz. 5, 316–324 (2020). https://doi.org/10.1039/c9nh00533a
Sun, M.M., Lv, Y., Song, Y.J., et al.: CO-tolerant PtRu@h-BN/C core–shell electrocatalysts for proton exchange membrane fuel cells. Appl. Surf. Sci. 450, 244–250 (2018). https://doi.org/10.1016/j.apsusc.2018.04.170
Yin, Y., Liu, J., Chang, Y.F., et al.: Design of Pt–C/Fe–N–S–C cathode dual catalyst layers for proton exchange membrane fuel cells under low humidity. Electrochim. Acta 296, 450–457 (2019). https://doi.org/10.1016/j.electacta.2018.11.048
Du, X.X., He, Y., Wang, X.X., et al.: Fine-grained and fully ordered intermetallic PtFe catalysts with largely enhanced catalytic activity and durability. Energy Environ. Sci. 9, 2623–2632 (2016). https://doi.org/10.1039/c6ee01204c
Yang, D.J., Yan, Z.Y., Li, B., et al.: Highly active and durable Pt–Co nanowire networks catalyst for the oxygen reduction reaction in PEMFCs. Int. J. Hydrog. Energy 41, 18592–18601 (2016). https://doi.org/10.1016/j.ijhydene.2016.08.159
Li, J.R., Sharma, S., Liu, X.M., et al.: Hard-magnet L10–CoPt nanoparticles advance fuel cell catalysis. Joule 3, 124–135 (2019). https://doi.org/10.1016/j.joule.2018.09.016
Jung, W.S., Popov, B.N.: New method to synthesize highly active and durable chemically ordered fct-PtCo cathode catalyst for PEMFCs. ACS Appl. Mater. Interfaces. 9, 23679–23686 (2017). https://doi.org/10.1021/acsami.7b04750
Lim, T., Kim, O.H., Sung, Y.E., et al.: Preparation of onion-like Pt-terminated Pt–Cu bimetallic nano-sized electrocatalysts for oxygen reduction reaction in fuel cells. J. Power Sources 316, 124–131 (2016). https://doi.org/10.1016/j.jpowsour.2016.03.068
Wu, M.X., Wu, X., Zhang, L., et al.: Cu@Pt catalysts prepared by galvanic replacement of polyhedral copper nanoparticles for polymer electrolyte membrane fuel cells. Electrochim. Acta 306, 167–174 (2019). https://doi.org/10.1016/j.electacta.2019.03.111
Ren, X.F., Lv, Q., Liu, L.F., et al.: Current progress of Pt and Pt-based electrocatalysts used for fuel cells. Sustain. Energy Fuels 4, 15–30 (2020). https://doi.org/10.1039/c9se00460b
Kongkanand, A., Subramanian, N.P., Yu, Y.C., et al.: Achieving high-power PEM fuel cell performance with an ultralow-Pt-content core–shell catalyst. ACS Catal. 6, 1578–1583 (2016). https://doi.org/10.1021/acscatal.5b02819
Wang, K.C., Huang, H.C., Wang, C.H.: Synthesis of Pd@Pt3Co/C core-shell structure as catalyst for oxygen reduction reaction in proton exchange membrane fuel cell. Int. J. Hydrog. Energy 42, 11771–11778 (2017). https://doi.org/10.1016/j.ijhydene.2017.03.084
Hwang, C.K., Kim, J.M., Hwang, S., et al.: Porous strained Pt nanostructured thin-film electrocatalysts via dealloying for PEM fuel cells. Adv. Mater. Interfaces 7, 1901326 (2020). https://doi.org/10.1002/admi.201901326
Vorokhta, M., Khalakhan, I., Matolínová, I., et al.: PLD prepared nanostructured Pt–CeO2 thin films containing ionic platinum. Appl. Surf. Sci. 396, 278–283 (2017). https://doi.org/10.1016/j.apsusc.2016.10.119
Sui, S., Wei, Z.X., Su, K.H., et al.: Pt nanowire growth induced by Pt nanoparticles in application of the cathodes for polymer electrolyte membrane fuel cells (PEMFCs). Int. J. Hydrog. Energy 43, 20041–20049 (2018). https://doi.org/10.1016/j.ijhydene.2018.09.009
Wang, W., Lv, F., Lei, B., et al.: Tuning nanowires and nanotubes for efficient fuel-cell electrocatalysis. Adv. Mater. 28, 10117–10141 (2016). https://doi.org/10.1002/adma.201601909
Sun, R.L., Xia, Z.X., Shang, L., et al.: Hierarchically ordered arrays with platinum coated PANI nanowires for highly efficient fuel cell electrodes. J. Mater. Chem. A 5, 15260–15265 (2017). https://doi.org/10.1039/c7ta02500a
Du, H.T., Zhang, X.P., Tan, Q.Q., et al.: A Cu3P–CoP hybrid nanowire array: a superior electrocatalyst for acidic hydrogen evolution reactions. Chem. Commun. 53, 12012–12015 (2017). https://doi.org/10.1039/c7cc07802a
Dogan, D.C., Cho, S., Hwang, S.M., et al.: Highly durable supportless pt hollow spheres designed for enhanced oxygen transport in cathode catalyst layers of proton exchange membrane fuel cells. ACS Appl. Mater. Inter. 8, 27730–27739 (2016). https://doi.org/10.1021/acsami.6b08177
Pizzutilo, E., Knossalla, J., Geiger, S., et al.: The space confinement approach using hollow graphitic spheres to unveil activity and stability of Pt–Co nanocatalysts for PEMFC. Adv. Energy Mater. 7, 1700835 (2017). https://doi.org/10.1002/aenm.201700835
Wang, X.X., Swihart, M.T., Wu, G.: Achievements, challenges and perspectives on cathode catalysts in proton exchange membrane fuel cells for transportation. Nat. Catal. 2, 578–589 (2019). https://doi.org/10.1038/s41929-019-0304-9
Pan, L.J., Ott, S., Dionigi, F., et al.: Current challenges related to the deployment of shape-controlled Pt alloy oxygen reduction reaction nanocatalysts into low Pt-loaded cathode layers of proton exchange membrane fuel cells. Curr. Opin. Electrochem. 18, 61–71 (2019). https://doi.org/10.1016/j.coelec.2019.10.011
Antolini, E.: Carbon supports for low-temperature fuel cell catalysts. Appl. Catal. B 88, 1–24 (2009). https://doi.org/10.1016/j.apcatb.2008.09.030
Shao, Y.Y., Liu, J., Wang, Y., et al.: Novel catalyst support materials for PEMfuelcells: current status and future prospects. J. Mater. Chem. 19, 46–59 (2009). https://doi.org/10.1039/b808370c
Dicks, A.L.: The role of carbon in fuel cells. J. Power Sources 156, 128–141 (2006). https://doi.org/10.1016/j.jpowsour.2006.02.054
Samad, S., Loh, K.S., Wong, W.Y., et al.: Carbon and non-carbon support materials for platinum-based catalysts in fuel cells. Int. J. Hydrog. Energy 43, 7823–7854 (2018). https://doi.org/10.1016/j.ijhydene.2018.02.154
You, P.Y., Kamarudin, S.K.: Recent progress of carbonaceous materials in fuel cell applications: an overview. Chem. Eng. J. 309, 489–502 (2017). https://doi.org/10.1016/j.cej.2016.10.051
Mansor, M., Timmiati, S.N., Lim, K.L., et al.: Recent progress of anode catalysts and their support materials for methanol electrooxidation reaction. Int. J. Hydrog. Energy 44, 14744–14769 (2019). https://doi.org/10.1016/j.ijhydene.2019.04.100
Antolini, E.: Graphene as a new carbon support for low-temperature fuel cell catalysts. Appl. Catal. B: Environ. 123–124, 52–68 (2012). https://doi.org/10.1016/j.apcatb.2012.04.022
Luo, C., Xie, H., Wang, Q., et al.: A review of the application and performance of carbon nanotubes in fuel cells. J. Nanomater. 2015, 1–10 (2015). https://doi.org/10.1155/2015/560392
Gan, L., Du, H.D., Li, B.H., et al.: The effect of particle size on the interaction of Pt catalyst particles with a carbon black support. New Carbon Mater. 25, 53–59 (2010). https://doi.org/10.1016/s1872-5805(09)60015-9
Wang, X., Hsing, I.M., Yue, P.L.: Electrochemical characterization of binary carbon supported electrode in polymer electrolyte fuel cells. J. Power Sources 96, 282–287 (2001). https://doi.org/10.1016/S0378-7753(00)00625-X
Padgett, E., Andrejevic, N., Liu, Z.Y., et al.: Editors’ choice: connecting fuel cell catalyst nanostructure and accessibility using quantitative cryo-STEM tomography. J. Electrochem. Soc. 165, F173–F180 (2018). https://doi.org/10.1149/2.0541803jes
Cetinbas, F.C., Ahluwalia, R.K., Kariuki, N.N., et al.: Effects of porous carbon morphology, agglomerate structure and relative humidity on local oxygen transport resistance. J. Electrochem. Soc. 167, 013508 (2019). https://doi.org/10.1149/2.0082001jes
Park, Y.C., Tokiwa, H., Kakinuma, K., et al.: Effects of carbon supports on Pt distribution, ionomer coverage and cathode performance for polymer electrolyte fuel cells. J. Power Sources 315, 179–191 (2016). https://doi.org/10.1016/j.jpowsour.2016.02.091
Yu, P.T., Liu, Z.Y., Makharia, R.: Investigation of carbon corrosion behavior and kinetics in proton exchange membrane fuel cell cathode electrodes. J. Electrochem. Soc. 160, F645–F650 (2013). https://doi.org/10.1149/2.120306jes
Devrim, Y., Arıca, E.D.: Investigation of the effect of graphitized carbon nanotube catalyst support for high temperature PEM fuel cells. Int. J. Hydrog. Energy 45, 3609–3617 (2020). https://doi.org/10.1016/j.ijhydene.2019.01.111
Zhu, S.Y., Zheng, J.S., Huang, J., et al.: Fabrication of three-dimensional buckypaper catalyst layer with Pt nanoparticles supported on polyelectrolyte functionalized carbon nanotubes for proton exchange membrane fuel cells. J. Power Sources 393, 19–31 (2018). https://doi.org/10.1016/j.jpowsour.2018.04.100
Berber, M.R., Hafez, I.H., Fujigaya, T., et al.: A highly durable fuel cell electrocatalyst based on double-polymer-coated carbon nanotubes. Sci. Rep. 5, 16711 (2015). https://doi.org/10.1038/srep16711
Skupov, K.M., Ponomarev, I.I., Razorenov, D.Y., et al.: Carbon nanofiber paper electrodes based on heterocyclic polymers for high temperature polymer electrolyte membrane fuel cell. Macromol. Symp. 375, 1600188 (2017). https://doi.org/10.1002/masy.201600188
Chung, S., Shin, D., Choun, M., et al.: Improved water management of Pt/C cathode modified by graphitized carbon nanofiber in proton exchange membrane fuel cell. J. Power Sources 399, 350–356 (2018). https://doi.org/10.1016/j.jpowsour.2018.07.126
Li, M.W., Wu, X., Zeng, J.H., et al.: Heteroatom doped carbon nanofibers synthesized by chemical vapor deposition as platinum electrocatalyst supports for polymer electrolyte membrane fuel cells. Electrochim. Acta 182, 351–360 (2015). https://doi.org/10.1016/j.electacta.2015.09.122
Yarlagadda, V., Carpenter, M.K., Moylan, T.E., et al.: Boosting fuel cell performance with accessible carbon mesopores. ACS Energy Lett. 3, 618–621 (2018). https://doi.org/10.1021/acsenergylett.8b00186
Prithi, J.A., Rajalakshmi, N., Ranga Rao, G.: Nitrogen doped mesoporous carbon supported Pt electrocatalyst for oxygen reduction reaction in proton exchange membrane fuel cells. Int. J. Hydrog. Energy 43, 4716–4725 (2018). https://doi.org/10.1016/j.ijhydene.2017.11.137
Sultana, K.N., Worku, D., Hossain, M.T.Z., et al.: Synthesis of graphitic mesoporous carbon from metal impregnated silica template for proton exchange membrane fuel cell application. Fuel Cells 19, 27–34 (2019). https://doi.org/10.1002/fuce.201800034
Liu, S.G., Huang, S.P.: Theoretical insights into the activation of O2 by Pt single atom and Pt4 nanocluster on functionalized graphene support: critical role of Pt positive polarized charges. Carbon 115, 11–17 (2017). https://doi.org/10.1016/j.carbon.2016.12.094
Devrim, Y., Arıca, E.D., Albostan, A.: Graphene based catalyst supports for high temperature PEM fuel cell application. Int. J. Hydrog. Energy 43, 11820–11829 (2018). https://doi.org/10.1016/j.ijhydene.2018.03.047
Balu, R., Choudhury, N.R., Mata, J.P., et al.: Evolution of the interfacial structure of a catalyst ink with the quality of the dispersing solvent: a contrast variation small-angle and ultrasmall-angle neutron scattering investigation. ACS Appl. Mater. Interfaces. 11, 9934–9946 (2019). https://doi.org/10.1021/acsami.8b20645
Orfanidi, A., Rheinländer, P.J., Schulte, N., et al.: Ink solvent dependence of the ionomer distribution in the catalyst layer of a PEMFC. J. Electrochem. Soc. 165, F1254–F1263 (2018). https://doi.org/10.1149/2.1251814jes
Song, C., Park, J.: Effect of dispersion solvents in catalyst inks on the performance and durability of catalyst layers in proton exchange membrane fuel cells. Energies 12, 549 (2019). https://doi.org/10.3390/en12030549
Koh, B.S., Yi, S.C.: Effect of organic solvents on catalyst structure of PEM fuel cell electrode fabricated via electrospray deposition. J. Ceram. Process Res. 18, 810–814 (2017)
Kim, T.H., Yi, J.Y., Jung, C.Y., et al.: Solvent effect on the Nafion agglomerate morphology in the catalyst layer of the proton exchange membrane fuel cells. Int. J. Hydrog. Energy 42, 478–485 (2017). https://doi.org/10.1016/j.ijhydene.2016.12.015
Kim, Y.S., Welch, C.F., Mack, N.H., et al.: Highly durable fuel cell electrodes based on ionomers dispersed in glycerol. Phys. Chem. Chem. Phys. 16, 5927–5932 (2014). https://doi.org/10.1039/c4cp00496e
Yang, F., Xin, L., Uzunoglu, A., et al.: Investigation of solvent effects on the dispersion of carbon agglomerates and nafion ionomer particles in catalyst inks using ultra small angle X-ray scattering method. ECS Trans. 75, 361–371 (2016). https://doi.org/10.1149/07514.0361ecst
Huang, D.C., Yu, P.J., Liu, F.J., et al.: Effect of dispersion solvent in catalyst ink on proton exchange membrane fuel cell performance. Int. J. Electrochem. Sci. 6, 2551–2565 (2011)
Shin, S.J., Lee, J.K., Ha, H.Y., et al.: Effect of the catalytic ink preparation method on the performance of polymer electrolyte membrane fuel cells. J. Power Sources 106, 146–152 (2002). https://doi.org/10.1016/S0378-7753(01)01045-X
Shukla, S., Bhattacharjee, S., Weber, A.Z., et al.: Experimental and theoretical analysis of ink dispersion stability for polymer electrolyte fuel cell applications. J. Electrochem. Soc. 164, F600–F609 (2017). https://doi.org/10.1149/2.0961706jes
Sharma, R., Grahl-Madsen, L., Andersen, S.M.: Influence of dispersion media on Nafion ionomer distribution in proton exchange membrane fuel cell catalyst carbon support. Mater. Chem. Phys. 226, 66–72 (2019). https://doi.org/10.1016/j.matchemphys.2019.01.015
Jorge, M., Lue, L.: The dielectric constant: reconciling simulation and experiment. J. Chem. Phys. 150, 084108 (2019). https://doi.org/10.1063/1.5080927
Uchida, M., Aoyama, Y., Eda, N., et al.: New preparation method for polymer–electrolyte fuel cells. J. Electrochem. Soc. 142, 463–468 (1995). https://doi.org/10.1149/1.2044068
Shukla, S., Bhattacharjee, S., Secanell, M.: Rationalizing catalyst inks for PEMFC electrodes based on colloidal interactions. ECS Trans. 58, 1409–1428 (2013). https://doi.org/10.1149/05801.1409ecst
Ngo, T.T., Yu, T.L., Lin, H.L.: Nafion-based membrane electrode assemblies prepared from catalyst inks containing alcohol/water solvent mixtures. J. Power Sources 238, 1–10 (2013). https://doi.org/10.1016/j.jpowsour.2013.03.055
Therdthianwong, A., Ekdharmasuit, P., Therdthianwong, S.: Fabrication and performance of membrane electrode assembly prepared by a catalyst-coated membrane method: effect of solvents used in a catalyst ink mixture. Energy Fuels 24, 1191–1196 (2010). https://doi.org/10.1021/ef901105k
Song, W., Yu, H.M., Hao, L.X., et al.: Effect of catalytic ink on sub-freezing endurance of PEMFCs. Int. J. Hydrog. Energy 35, 11129–11137 (2010). https://doi.org/10.1016/j.ijhydene.2010.07.041
Chisaka, M., Daiguji, H.: Effect of organic solvents on catalyst layer structure in polymer electrolyte membrane fuel cells. J. Electrochem. Soc. 156, B22–B26 (2009). https://doi.org/10.1149/1.3000592
Fernández, R., Ferreira-Aparicio, P., Daza, L.: PEMFC electrode preparation: influence of the solvent composition and evaporation rate on the catalytic layer microstructure. J. Power Sources 151, 18–24 (2005). https://doi.org/10.1016/j.jpowsour.2005.02.048
Yang, T.H., Yoon, Y.G., Park, G.G., et al.: Fabrication of a thin catalyst layer using organic solvents. J. Power Sources 127, 230–233 (2004). https://doi.org/10.1016/j.jpowsour.2003.09.018
Kim, J.H., Ha, H.Y., Oh, I.H., et al.: Influence of the solvent in anode catalyst ink on the performance of a direct methanol fuel cell. J. Power Sources 135, 29–35 (2004). https://doi.org/10.1016/j.jpowsour.2004.03.058
Lee, S.J., Yu, T.L., Lin, H.L., et al.: Solution properties of nafion in methanol/water mixture solvent. Polymer 45, 2853–2862 (2004). https://doi.org/10.1016/j.polymer.2004.01.076
Welch, C.F., Labouriau, A., Hjelm, R.P., et al.: Nafion in dilute solvent systems: dispersion or solution? ACS Macro Lett. 1, 1403–1407 (2012). https://doi.org/10.1021/mz3005204
Kim, T.H., Yoo, J.H., Maiyalagan, T., et al.: Influence of the Nafion agglomerate morphology on the water-uptake behavior and fuel cell performance in the proton exchange membrane fuel cells. Appl. Surf. Sci. 481, 777–784 (2019). https://doi.org/10.1016/j.apsusc.2019.03.113
Ngo, T.T., Yu, T.L., Lin, H.L.: Influence of the composition of isopropyl alcohol/water mixture solvents in catalyst ink solutions on proton exchange membrane fuel cell performance. J. Power Sources 225, 293–303 (2013). https://doi.org/10.1016/j.jpowsour.2012.10.055
Sharma, R., Andersen, S.M.: Zoom in catalyst/ionomer interface in polymer electrolyte membrane fuel cell electrodes: impact of catalyst/ionomer dispersion media/solvent. ACS Appl. Mater. Interfaces. 10, 38125–38133 (2018). https://doi.org/10.1021/acsami.8b14622
Kusoglu, A., Weber, A.Z.: New insights into perfluorinated sulfonic-acid ionomers. Chem. Rev. 117, 987–1104 (2017). https://doi.org/10.1021/acs.chemrev.6b00159
Liu, Y., Horan, J.L., Schlichting, G.J., et al.: A small-angle X-ray scattering study of the development of morphology in films formed from the 3 M perfluorinated sulfonic acid ionomer. Macromolecules 45, 7495–7503 (2012). https://doi.org/10.1021/ma300926e
Kim, O.H., Oh, S.H., Ahn, C.Y., et al.: Enhanced performance of ionomer binder with shorter side-chains, higher dispersibility, and lower equivalent weight. Fuel Cells 18, 711–722 (2018). https://doi.org/10.1002/fuce.201800083
Mauritz, K., Moore, R.: State of understanding of Nafion. Chem. Rev. 104, 4535–4585 (2004)
Karimi, M.B., Mohammadi, F., Hooshyari, K.: Recent approaches to improve Nafion performance for fuel cell applications: a review. Int. J. Hydrog. Energy 44, 28919–28938 (2019). https://doi.org/10.1016/j.ijhydene.2019.09.096
Lade, H., Kumar, V., Arthanareeswaran, G., et al.: Sulfonated poly(arylene ether sulfone) nanocomposite electrolyte membrane for fuel cell applications: a review. Int. J. Hydrog. Energy 42, 1063–1074 (2017). https://doi.org/10.1016/j.ijhydene.2016.10.038
Xiao, L., Chen, X., Xu, J.J., et al.: Synthesis and properties of novel side-chain sulfonated poly(arylene ether sulfone)s for proton exchange membranes. J. Polym. Sci. Part A: Polym. Chem. 57, 2304–2313 (2019). https://doi.org/10.1002/pola.29533
Oh, K.H., Kim, W.K., Choo, M.J., et al.: Partially perfluorinated hydrocarbon ionomer for cathode catalyst layer of polymer electrolyte membrane fuel cell. Electrochim. Acta 125, 314–319 (2014). https://doi.org/10.1016/j.electacta.2014.01.116
Sahin, A., Tasdemir, H.M., Ar, İ.: Improved performance and durability of sulfonated polyether ether ketone/cerium phosphate composite membrane for proton exchange membrane fuel cells. Ionics 25, 5163–5175 (2019). https://doi.org/10.1007/s11581-019-03109-y
Omata, T., Tanaka, M., Miyatake, K., et al.: Preparation and fuel cell performance of catalyst layers using sulfonated polyimide ionomers. ACS Appl. Mater. Interfaces. 4, 730–737 (2012). https://doi.org/10.1021/am201360j
Yao, Z.L., Zhang, Z.H., Hu, M., et al.: Perylene-based sulfonated aliphatic polyimides for fuel cell applications: performance enhancement by stacking of polymer chains. J. Membr. Sci. 547, 43–50 (2018). https://doi.org/10.1016/j.memsci.2017.10.032
Peron, J., Shi, Z., Holdcroft, S.: Hydrocarbon proton conducting polymers for fuel cell catalyst layers. Energy Environ. Sci. 4, 1575–1591 (2011). https://doi.org/10.1039/C0EE00638F
Aldebert, P., Dreyfus, B., Gebel, G., et al.: Rod like micellar structures in perfluorinated ionomer solutions. J. Phys. France 49, 2101–2109 (1988). https://doi.org/10.1051/jphys:0198800490120210100
Aldebert, P., Dreyfus, B., Pineri, M.: Small-angle neutron scattering of perfluorosulfonated ionomers in solution. Macromolecules 19, 2651–2653 (1986). https://doi.org/10.1021/ma00164a035
Szajdzinska-Pietek, E., Schlick, S., Plonka, A.: Self-assembling of perfluorinated polymeric surfactants in nonaqueous solvents. Electron spin resonance spectra of nitroxide spin probes in nafion solutions and swollen membranes. Langmuir 10, 2188–2196 (1994). https://doi.org/10.1021/la00019a026
Szajdzinska-Pietek, E., Schlick, S., Plonka, A.: Self-assembling of perfluorinated polymeric surfactants in water. Electron-spin resonance spectra of nitroxide spin probes in nafion solutions and swollen membranes. Langmuir 10, 1101–1109 (1994). https://doi.org/10.1021/la00016a023
Loppinet, B., Gebel, G., Williams, C.E.: Small-angle scattering study of perfluorosulfonated ionomer solutions. J. Phys. Chem. B 101, 1884–1892 (1997). https://doi.org/10.1021/jp9623047
Xu, F., Zhang, H., Ilavsky, J., et al.: Investigation of a catalyst ink dispersion using both ultra-small-angle X-ray scattering and cryogenic TEM. Langmuir 26, 19199–19208 (2010). https://doi.org/10.1021/la1028228
Yamaguchi, M., Matsunaga, T., Amemiya, K., et al.: Dispersion of rod-like particles of nafion in salt-free water/1-propanol and water/ethanol solutions. J. Phys. Chem. B 118, 14922–14928 (2014). https://doi.org/10.1021/jp506814m
Berlinger, S.A., McCloskey, B.D., Weber, A.Z.: Inherent acidity of perfluorosulfonic acid ionomer dispersions and implications for ink aggregation. J. Phys. Chem. B 122, 7790–7796 (2018). https://doi.org/10.1021/acs.jpcb.8b06493
Berlinger, S.A., McCloskey, B.D., Weber, A.Z.: Understanding binary interactions in fuel-cell catalyst-layer inks. ECS Trans. 80, 309–319 (2017). https://doi.org/10.1149/08008.0309ecst
Yang, F., Xin, L., Uzunoglu, A., et al.: Investigation of the interaction between nafion ionomer and surface functionalized carbon black using both ultrasmall angle X-ray scattering and cryo-TEM. ACS Appl. Mater. Interfaces. 9, 6530–6538 (2017). https://doi.org/10.1021/acsami.6b12949
Ma, C.H., Yu, T.L., Lin, H.L., et al.: Morphology and properties of Nafion membranes prepared by solution casting. Polymer 50, 1764–1777 (2009). https://doi.org/10.1016/j.polymer.2009.01.060
Lin, H.L., Yu, T.L., Huang, C.H., et al.: Morphology study of Nafion membranes prepared by solutions casting. J. Polym. Sci. B Polym. Phys. 43, 3044–3057 (2005). https://doi.org/10.1002/polb.20599
Yeo, R.S.: Dual cohesive energy densities of perfluorosulphonic acid (Nafion) membrane. Polymer 21, 432–435 (1980). https://doi.org/10.1016/0032-3861(80)90015-4
Kim, Y.S., Welch, C.F., Hjelm, R.P., et al.: Origin of toughness in dispersion-cast nafion membranes. Macromolecules 48, 2161–2172 (2015). https://doi.org/10.1021/ma502538k
Ishikawa, H., Sugawara, Y., Inoue, G., et al.: Effects of Pt and ionomer ratios on the structure of catalyst layer: a theoretical model for polymer electrolyte fuel cells. J. Power Sources 374, 196–204 (2018). https://doi.org/10.1016/j.jpowsour.2017.11.026
Doo, G., Lee, J.H., Yuk, S., et al.: Tuning the ionomer distribution in the fuel cell catalyst layer with scaling the ionomer aggregate size in dispersion. ACS Appl. Mater. Interfaces. 10, 17835–17841 (2018). https://doi.org/10.1021/acsami.8b01751
Morawietz, T., Handl, M., Oldani, C., et al.: Quantitative in situ analysis of ionomer structure in fuel cell catalytic layers. ACS Appl. Mater. Interfaces. 8, 27044–27054 (2016). https://doi.org/10.1021/acsami.6b07188
Uchida, M., Park, Y.C., Kakinuma, K., et al.: Effect of the state of distribution of supported Pt nanoparticles on effective Pt utilization in polymer electrolyte fuel cells. Phys. Chem. Chem. Phys. 15, 11236 (2013). https://doi.org/10.1039/c3cp51801a
Andersen, S.M., Grahl-Madsen, L.: Interface contribution to the electrode performance of proton exchange membrane fuel cells—impact of the ionomer. Int. J. Hydrog. Energy 41, 1892–1901 (2016). https://doi.org/10.1016/j.ijhydene.2015.11.101
Ott, S., Orfanidi, A., Schmies, H., et al.: Ionomer distribution control in porous carbon-supported catalyst layers for high-power and low Pt-loaded proton exchange membrane fuel cells. Nat. Mater. 19, 77–85 (2020). https://doi.org/10.1038/s41563-019-0487-0
Thoma, M., Lin, W., Hoffmann, E., et al.: Simple and reliable method for studying the adsorption behavior of aquivion ionomers on carbon black surfaces. Langmuir 34, 12324–12334 (2018). https://doi.org/10.1021/acs.langmuir.8b02726
Andersen, S.M.: Nano carbon supported platinum catalyst interaction behavior with perfluorosulfonic acid ionomer and their interface structures. Appl. Catal. B 181, 146–155 (2016). https://doi.org/10.1016/j.apcatb.2015.07.049
Andersen, S.M., Borghei, M., Dhiman, R., et al.: Interaction of multi-walled carbon nanotubes with perfluorinated sulfonic acid ionomers and surface treatment studies. Carbon 71, 218–228 (2014). https://doi.org/10.1016/j.carbon.2014.01.032
Andersen, S.M., Borghei, M., Dhiman, R., et al.: Adsorption behavior of perfluorinated sulfonic acid ionomer on highly graphitized carbon nanofibers and their thermal stabilities. J. Phys. Chem. C 118, 10814–10823 (2014). https://doi.org/10.1021/jp501088d
Koestner, R., Roiter, Y., Kozhinova, I., et al.: AFM imaging of adsorbed Nafion polymer on mica and graphite at molecular level. Langmuir 27, 10157–10166 (2011). https://doi.org/10.1021/la201283a
Koestner, R., Roiter, Y., Kozhinova, I., et al.: Effect of local charge distribution on graphite surface on nafion polymer adsorption as visualized at the molecular level. J. Phys. Chem. C 115, 16019–16026 (2011). https://doi.org/10.1021/jp203392m
Kudo, K., Jinnouchi, R., Morimoto, Y.: Humidity and temperature dependences of oxygen transport resistance of nafion thin film on platinum electrode. Electrochim. Acta 209, 682–690 (2016). https://doi.org/10.1016/j.electacta.2016.04.023
Kodama, K., Jinnouchi, R., Suzuki, T., et al.: Increase in adsorptivity of sulfonate anions on Pt (111) surface with drying of ionomer. Electrochem. Commun. 36, 26–28 (2013). https://doi.org/10.1016/j.elecom.2013.09.005
Subbaraman, R., Strmcnik, D., Paulikas, A.P., et al.: Oxygen reduction reaction at three-phase interfaces. ChemPhysChem 11, 2825–2833 (2010). https://doi.org/10.1002/cphc.201000190
Kodama, K., Motobayashi, K., Shinohara, A., et al.: Effect of the side-chain structure of perfluoro-sulfonic acid ionomers on the oxygen reduction reaction on the surface of Pt. ACS Catal. 8, 694–700 (2018). https://doi.org/10.1021/acscatal.7b03571
Kusano, T., Hiroi, T., Amemiya, K., et al.: Structural evolution of a catalyst ink for fuel cells during the drying process investigated by CV-SANS. Polym. J. 47, 546–555 (2015). https://doi.org/10.1038/pj.2015.36
Shibayama, M., Matsunaga, T., Kusano, T., et al.: SANS studies on catalyst ink of fuel cell. J. Appl. Polym. Sci. 131, 39842 (2014). https://doi.org/10.1002/app.39842
Takahashi, S., Shimanuki, J., Mashio, T., et al.: Observation of ionomer in catalyst ink of polymer electrolyte fuel cell using cryogenic transmission electron microscopy. Electrochim. Acta 224, 178–185 (2017). https://doi.org/10.1016/j.electacta.2016.12.068
Yoshimune, W., Harada, M.: Effect of Pt loading on the adsorption of perfluoro-sulfonic acid ionomer in catalyst ink for polymer electrolyte fuel cells. Chem. Lett. 48, 487–490 (2019). https://doi.org/10.1246/cl.190017
Takahashi, S., Mashio, T., Horibe, N., et al.: Analysis of the microstructure formation process and its influence on the performance of polymer electrolyte fuel-cell catalyst layers. ChemElectroChem 2, 1560–1567 (2015). https://doi.org/10.1002/celc.201500131
Shimanuki, J., Takahashi, S., Tohma, H., et al.: Microstructural observation of fuel cell catalyst inks by Cryo-SEM and Cryo-TEM. Microscopy (Oxf) 66, 204–208 (2017). https://doi.org/10.1093/jmicro/dfx001
Khan, Z.U., Kausar, A., Ullah, H.: a review on composite papers of graphene oxide, carbon nanotube, polymer/GO, and polymer/CNT: processing strategies, properties, and relevance. Polym. Plast. Technol. Eng. 55, 559–581 (2016). https://doi.org/10.1080/03602559.2015.1098693
Long, C.M., Nascarella, M.A., Valberg, P.A.: Carbon black vs. black carbon and other airborne materials containing elemental carbon: physical and chemical distinctions. Environ Pollut 181, 271–286 (2013). https://doi.org/10.1016/j.envpol.2013.06.009
Soboleva, T., Zhao, X.S., Malek, K., et al.: On the micro-, meso-, and macroporous structures of polymer electrolyte membrane fuel cell catalyst layers. ACS Appl. Mater. Interfaces. 2, 375–384 (2010). https://doi.org/10.1021/am900600y
Lauterborn, W., Ohl, C.D.: Cavitation bubble dynamics. Ultrason. Sonochem. 4, 65–75 (1997). https://doi.org/10.1016/s1350-4177(97)00009-6
Pollet, B.G., Goh, J.T.E.: The importance of ultrasonic parameters in the preparation of fuel cell catalyst inks. Electrochim. Acta 128, 292–303 (2014). https://doi.org/10.1016/j.electacta.2013.09.160
Pollet, B.G.: Let’s not ignore the ultrasonic effects on the preparation of fuel cell materials. Electrocatalysis 5, 330–343 (2014). https://doi.org/10.1007/s12678-014-0211-4
Zhang, B., Chen, T.J.: Study of ultrasonic dispersion of graphene nanoplatelets. Materials 12, 1757 (2019). https://doi.org/10.3390/ma12111757
Wang, M., Park, J.H., Kabir, S., et al.: Impact of catalyst ink dispersing methodology on fuel cell performance using in situ X-ray scattering. ACS Appl. Energy Mater. 2, 6417–6427 (2019). https://doi.org/10.1021/acsaem.9b01037
Adamski, M., Peressin, N., Holdcroft, S., et al.: Does power ultrasound affect Nafion® dispersions? Ultrason. Sonochem. 60, 104758 (2020). https://doi.org/10.1016/j.ultsonch.2019.104758
Tiwary, C.S., Verma, A., Biswas, K., et al.: Preparation of ultrafine CsCl crystallites by combined cryogenic and room temperature ball milling. Ceram. Int. 37, 3677–3686 (2011). https://doi.org/10.1016/j.ceramint.2011.06.029
Shin, H., Lee, S., Suk Jung, H., et al.: Effect of ball size and powder loading on the milling efficiency of a laboratory-scale wet ball mill. Ceram. Int. 39, 8963–8968 (2013). https://doi.org/10.1016/j.ceramint.2013.04.093
Burmeister, C.F., Kwade, A.: Process engineering with planetary ball Mills. Chem. Soc. Rev. 42, 7660 (2013). https://doi.org/10.1039/c3cs35455e
Hashimasa, Y., Numata, T.: Comparison of test results on load cycle durability of polymer electrolyte fuel cell cathode catalysts. Int. J. Hydrog. Energy 40, 11543–11549 (2015). https://doi.org/10.1016/j.ijhydene.2015.04.031
Khandavalli, S., Park, J.H., Kariuki, N.N., et al.: Rheological investigation on the microstructure of fuel cell catalyst inks. ACS Appl. Mater. Interfaces. 10, 43610–43622 (2018). https://doi.org/10.1021/acsami.8b15039
Tamaki, T., Koshiishi, A., Sugawara, Y., et al.: Evaluation of performance and durability of platinum–iron–copper with L10 ordered face-centered tetragonal structure as cathode catalysts in polymer electrolyte fuel cells. J. Appl. Electrochem. 48, 773–782 (2018). https://doi.org/10.1007/s10800-018-1193-3
Uemura, S., Kameya, Y., Iriguchi, N., et al.: Communication: investigation of catalyst ink degradation by X-ray CT. J. Electrochem. Soc. 165, F142–F144 (2018). https://doi.org/10.1149/2.0411803jes
Uemura, S., Yoshida, T., Koga, M., et al.: Ink degradation and its effects on the crack formation of fuel cell catalyst layers. J. Electrochem. Soc. 166, F89–F92 (2019). https://doi.org/10.1149/2.0411902jes
Fritz, G., Schadler, V., Willenbacher, N., et al.: Electrosteric stabilization of colloidal dispersions. Langmuir 18, 6381–6390 (2002). https://doi.org/10.1021/la015734j
Zhang, H.N., Pan, J.J., He, X.C., et al.: Zeta potential of Nafion molecules in isopropanol–water mixture solvent. J. Appl. Polym. Sci. 107, 3306–3309 (2008). https://doi.org/10.1002/app.27473
Hoffmann, E., Zhang, S., Thoma, M., et al.: Formulation of carbon black-ionomer dispersions for thin film formation in fuel cells. Particuology 44, 7–21 (2019). https://doi.org/10.1016/j.partic.2018.08.001
Dixit, M., Hatzell, K.B.: Understanding binary interactions and aging effects in catalyst layer inks for controlled manufacturing. ECS Trans. 80, 301–307 (2017). https://doi.org/10.1149/08008.0301ecst
Uemura, S., Kameya, Y., Iriguchi, N., et al.: Time lapse visualization of catalyst ink degradation by X-ray CT. ECS Trans. 80, 403–407 (2017). https://doi.org/10.1149/08008.0403ecst
Kameya, Y., Iriguchi, N., Ohki, M., et al.: MRI and 1H/19F NMR investigation of dispersion state of PEFC catalyst ink. ECS Trans. 80, 819–827 (2017). https://doi.org/10.1149/08008.0819ecst
Koga, M., Tokita, M., Masunaga, H., Hikima, T., Sugimori, H., Sasabe, T., Yoshida, T., Shinohara, K., Hirai, S.: Investigation of drying process of catalyst ink for polymer electrolyte fuel cells by grazing-incidence X-ray scattering. ECS Trans. 86 (2018). https://doi.org/10.1149/ma2018-02/41/1393
Suzuki, T., Tsushima, S., Hirai, S.: Effects of Nafion® ionomer and carbon particles on structure formation in a proton-exchange membrane fuel cell catalyst layer fabricated by the decal-transfer method. Int. J. Hydrog. Energy 36, 12361–12369 (2011). https://doi.org/10.1016/j.ijhydene.2011.06.090
Inoue, G., Kawase, M.: Effect of porous structure of catalyst layer on effective oxygen diffusion coefficient in polymer electrolyte fuel cell. J. Power Sources 327, 1–10 (2016). https://doi.org/10.1016/j.jpowsour.2016.07.037
Ohnishi, T., So, M., Takenaka, S., et al.: Performance of carbon-supported Pt nanoparticles covered by silica layers with low ionomer in polymer electrolyte fuel cells. ECS Trans. 86, 453–460 (2018). https://doi.org/10.1149/08613.0453ecst
Zhao, J., Shahgaldi, S., Alaefour, I., et al.: Pore structure and effective diffusion coefficient of catalyzed electrodes in polymer electrolyte membrane fuel cells. Int. J. Hydrog. Energy 43, 3776–3785 (2018). https://doi.org/10.1016/j.ijhydene.2018.01.019
Inoue, G., Kawase, M.: Understanding formation mechanism of heterogeneous porous structure of catalyst layer in polymer electrolyte fuel cell. Int. J. Hydrog. Energy 41, 21352–21365 (2016). https://doi.org/10.1016/j.ijhydene.2016.08.029
Chisaka, M., Daiguji, H.: Effect of glycerol on micro/nano structures of catalyst layers in polymer electrolyte membrane fuel cells. Electrochim. Acta 51, 4828–4833 (2006). https://doi.org/10.1016/j.electacta.2006.01.018
Jiang, J.K., Oberdörster, G., Biswas, P.: Characterization of size, surface charge, and agglomeration state of nanoparticle dispersions for toxicological studies. J. Nanopart. Res. 11, 77–89 (2009). https://doi.org/10.1007/s11051-008-9446-4
Xu, R.L., Wu, C.F., Xu, H.Y.: Particle size and zeta potential of carbon black in liquid media. Carbon 45, 2806–2809 (2007). https://doi.org/10.1016/j.carbon.2007.09.010
Bevan, M.A., Prieve, D.C.: Direct measurement of retarded van der waals attraction. Langmuir 15, 7925–7936 (1999). https://doi.org/10.1021/la981381l
Runkana, V., Somasundaran, P., Kapur, P.C.: A population balance model for flocculation of colloidal suspensions by polymer bridging. Chem. Eng. Sci. 61, 182–191 (2006). https://doi.org/10.1016/j.ces.2005.01.046
Liu, J., Cui, S., Keffer, D.J.: Molecular-level investigation of critical gap size between catalyst particles and electrolyte in hydrogen proton exchange membrane fuel cells. Fuel Cells 8, 422–428 (2008). https://doi.org/10.1002/fuce.200800001
Balbuena, P.B., Lamas, E.J., Wang, Y.X.: Molecular modeling studies of polymer electrolytes for power sources. Electrochim. Acta 50, 3788–3795 (2005). https://doi.org/10.1016/j.electacta.2005.02.063
Lamas, E.J., Balbuena, P.B.: Molecular dynamics studies of a model polymer-catalyst-carbon interface. Electrochim. Acta 51, 5904–5911 (2006). https://doi.org/10.1016/j.electacta.2006.03.033
Damasceno Borges, D., Franco, A.A., Malek, K., et al.: Inhomogeneous transport in model hydrated polymer electrolyte supported ultrathin films. ACS Nano 7, 6767–6773 (2013). https://doi.org/10.1021/nn401624p
Sugaya, Y., Tokumasu, T.: Molecular dynamics study of oxygen permeation of ionomer of hydrocarbon. ECS Trans. 58, 1165–1174 (2013). https://doi.org/10.1149/05801.1165ecst
Jinnouchi, R., Kudo, K., Kitano, N., et al.: Molecular dynamics simulations on O2 permeation through nafion ionomer on platinum surface. Electrochim. Acta 188, 767–776 (2016). https://doi.org/10.1016/j.electacta.2015.12.031
Tarokh, A., Karan, K., Ponnurangam, S.: Atomistic MD study of nafion dispersions: role of solvent and counterion in the aggregate structure, ionic clustering, and acid dissociation. Macromolecules 53, 288–301 (2020). https://doi.org/10.1021/acs.macromol.9b01663
Malek, K., Mashio, T., Eikerling, M.: Microstructure of catalyst layers in PEM fuel cells redefined: a computational approach. Electrocatalysis 2, 141–157 (2011). https://doi.org/10.1007/s12678-011-0047-0
Malek, K., Eikerling, M., Wang, Q.P., et al.: Self-organization in catalyst layers of polymer electrolyte fuel cells. J. Phys. Chem. C 111, 13627–13634 (2007). https://doi.org/10.1021/jp072692k
Xiao, Y., Dou, M.L., Yuan, J.L., et al.: Fabrication process simulation of a PEM fuel cell catalyst layer and its microscopic structure characteristics. J. Electrochem. Soc. 159, B308–B314 (2012). https://doi.org/10.1149/2.064203jes
Lee, J.H., Doo, G., Kwon, S.H., et al.: Dispersion-solvent control of ionomer aggregation in a polymer electrolyte membrane fuel cell. Sci. Rep. 8, 10739 (2018). https://doi.org/10.1038/s41598-018-28779-y
Forouzan, M.M., Chao, C.W., Bustamante, D., et al.: Experiment and simulation of the fabrication process of lithium-ion battery cathodes for determining microstructure and mechanical properties. J. Power Sources 312, 172–183 (2016). https://doi.org/10.1016/j.jpowsour.2016.02.014
Munekata, T., Suzuki, T., Yamakawa, S., et al.: Effects of viscosity, surface tension, and evaporation rate of solvent on dry colloidal structures: a lattice Boltzmann study. Phys. Rev. E 88, 052314 (2013). https://doi.org/10.1103/physreve.88.052314
Akhgar, A., Khalili, B., Moa, B., et al.: Lattice–Boltzmann simulation of multi-phase phenomena related to fuel cells. In: Abstracts of the International Conference on Numerical Analysis and Applied Mathematics, Rhodes, Greece, 19–25 September 2016. https://doi.org/10.1063/1.4992192
Song, S.Y., Zhou, X., Li, L., et al.: Numerical simulation and experimental validation of SiC nanoparticle distribution in magnesium melts during ultrasonic cavitation based processing of magnesium matrix nanocomposites. Ultrason. Sonochem. 24, 43–54 (2015). https://doi.org/10.1016/j.ultsonch.2014.12.010
Choi, Y.J., Djilali, N.: Direct numerical simulations of agglomeration of circular colloidal particles in two-dimensional shear flow. Phys. Fluids 28, 013304 (2016). https://doi.org/10.1063/1.4939501
Bhattacharjee, S.: DLS and zeta potential: What they are and what they are not? J. Control Release 235, 337–351 (2016). https://doi.org/10.1016/j.jconrel.2016.06.017
Urban, C., Schurtenberger, P.: Characterization of turbid colloidal suspensions using light scattering techniques combined with cross-correlation methods. J. Colloid Interface Sci. 207, 150–158 (1998). https://doi.org/10.1006/jcis.1998.5769
Hassan, P.A., Rana, S., Verma, G.: Making sense of Brownian motion: colloid characterization by dynamic light scattering. Langmuir 31, 3–12 (2015). https://doi.org/10.1021/la501789z
Kaler, E.W.: Small-angle scattering from colloidal dispersions. J. Appl. Crystallogr. 21, 729–736 (1988). https://doi.org/10.1107/s0021889888005230
Ren, Y., Zuo, X.B.: Synchrotron X-ray and neutron diffraction, total scattering, and small-angle scattering techniques for rechargeable battery research. Small Methods 2, 1800064 (2018). https://doi.org/10.1002/smtd.201800064
Fan, Y.X., Wang, Y.L.: Applications of small-angle X-ray scattering/small-angle neutron scattering and cryogenic transmission electron microscopy to understand self-assembly of surfactants. Curr. Opin. Colloid Interface Sci. 42, 1–16 (2019). https://doi.org/10.1016/j.cocis.2019.02.011
Kuntsche, J., Horst, J.C., Bunjes, H.: Cryogenic transmission electron microscopy (cryo-TEM) for studying the morphology of colloidal drug delivery systems. Int. J. Pharm. 417, 120–137 (2011). https://doi.org/10.1016/j.ijpharm.2011.02.001
Suzuki, T., Okada, S., Tsushima, S.: Characterization of catalyst inks by rheology and microscopic particle properties. ECS Trans. 86, 193–198 (2018). https://doi.org/10.1149/08613.0193ecst
Bonifácio, R.N., Paschoal, J.O.A., Linardi, M., et al.: Catalyst layer optimization by surface tension control during ink formulation of membrane electrode assemblies in proton exchange membrane fuel cell. J. Power Sources 196, 4680–4685 (2011). https://doi.org/10.1016/j.jpowsour.2011.01.010
Deiner, L.J., Reitz, T.L.: Inkjet and aerosol jet printing of electrochemical devices for energy conversion and storage. Adv. Eng. Mater. 19, 1600878 (2017). https://doi.org/10.1002/adem.201600878
Kuroki, H., Onishi, K., Asami, K., et al.: Catalyst slurry preparation using a hydrodynamic cavitation dispersion method for polymer electrolyte fuel cells. Ind. Eng. Chem. Res. 58, 19545–19550 (2019). https://doi.org/10.1021/acs.iecr.9b02111
Glüsen, A., Müller, M., Stolten, D.: Slot-Die coating: a new preparation method for direct methanol fuel cells catalyst layers. J. Fuel Cell Sci. Technol. 10, 044503 (2013). https://doi.org/10.1115/1.4024607
de las Heras, A., Vivas, F.J., Segura, F., et al.: From the cell to the stack. A chronological walk through the techniques to manufacture the PEFCs core. Renew. Sustain. Energy Rev. 96, 29–45 (2018). https://doi.org/10.1016/j.rser.2018.07.036
Somalu, M.R., Muchtar, A., Daud, W.R.W., et al.: Screen-printing inks for the fabrication of solid oxide fuel cell films: a review. Renew. Sustain. Energy Rev. 75, 426–439 (2017). https://doi.org/10.1016/j.rser.2016.11.008
Somalu, M.R., Yufit, V., Brandon, N.P.: The effect of solids loading on the screen-printing and properties of nickel/scandia-stabilized-zirconia anodes for solid oxide fuel cells. Int. J. Hydrog. Energy 38, 9500–9510 (2013). https://doi.org/10.1016/j.ijhydene.2012.06.061
Somalu, M.R., Muchtar, A., Baboli, M.G., et al.: Understanding the relationship between ink rheology and film properties for screen-printed nickel/scandia-stabilized-zirconia anodes. ECS Trans. 57, 1321–1330 (2013). https://doi.org/10.1149/05701.1321ecst
Khandavalli, S., Park, J.H., Kariuki, N.N., et al.: Investigation of the microstructure and rheology of iridium oxide catalyst inks for low-temperature polymer electrolyte membrane water electrolyzers. ACS Appl. Mater. Interfaces. 11, 45068–45079 (2019). https://doi.org/10.1021/acsami.9b14415
Zhao, J., Li, X.G., Liu, Z.S.: The effect of ink dilution and evaporation on the microstructures of catalyst layers in polymer electrolyte membrane fuel cells. Int. J. Energy Res. 43, 6799–6811 (2019). https://doi.org/10.1002/er.4671
Su, H.N., Jao, T.C., Barron, O., et al.: Low platinum loading for high temperature proton exchange membrane fuel cell developed by ultrasonic spray coating technique. J. Power Sources 267, 155–159 (2014). https://doi.org/10.1016/j.jpowsour.2014.05.086
Navaei Alvar, E., Zhou, B., Eichhorn, S.H.: Composite-supported Pt catalyst and electrosprayed cathode catalyst layer for polymer electrolyte membrane fuel cell. Int. J. Energy Res. 41, 1626–1641 (2017). https://doi.org/10.1002/er.3746
Chan, S., Jankovic, J., Susac, D., et al.: Electrospun carbon nanofiber catalyst layers for polymer electrolyte membrane fuel cells: fabrication and optimization. J. Mater. Sci. 53, 11633–11647 (2018). https://doi.org/10.1007/s10853-018-2411-4
Si, D., Zhang, S., Huang, J., et al.: Electrochemical characterization of pre-conditioning process of electrospun nanofiber electrodes in polymer electrolyte fuel cells. Fuel Cells 18, 576–585 (2018). https://doi.org/10.1002/fuce.201700209
Shukla, S., Stanier, D., Saha, M.S., et al.: Analysis of inkjet printed PEFC electrodes with varying platinum loading. J. Electrochem. Soc. 163, F677–F687 (2016). https://doi.org/10.1149/2.1111607jes
Daud, W.R.W., Mohamad, A.B., Kadhum, A.A.H., et al.: Performance optimisation of PEM fuel cell during MEA fabrication. Energy Convers. Manag. 45, 3239–3249 (2004). https://doi.org/10.1016/j.enconman.2004.01.011
Bredol, M., Szydło, A., Radev, I., et al.: How the colloid chemistry of precursor electrocatalyst dispersions is related to the polymer electrolyte membrane fuel cell performance. J. Power Sources 402, 15–23 (2018). https://doi.org/10.1016/j.jpowsour.2018.09.005
Chingthamai, N., Sombatmankhong, K., Laoonual, Y.: Experimental investigation of electrospray coating technique for electrode fabrication in PEMFCs. Energy Proc. 105, 1806–1812 (2017). https://doi.org/10.1016/j.egypro.2017.03.523
Millington, B., Whipple, V., Pollet, B.G.: A novel method for preparing proton exchange membrane fuel cell electrodes by the ultrasonic-spray technique. J. Power Sources 196, 8500–8508 (2011). https://doi.org/10.1016/j.jpowsour.2011.06.024
Bender, G., Zawodzinski, T.A., Saab, A.P.: Fabrication of high precision PEFC membrane electrode assemblies. J. Power Sources 124, 114–117 (2003). https://doi.org/10.1016/S0378-7753(03)00735-3
Engle, R.: Maximizing the use of platinum catalyst by ultrasonic spray application. J. Fuel Cell Sci. Technol. 9, 014501 (2012). https://doi.org/10.1115/1.4004462
Huang, X.Y., Rigdon, W.A., Neutzler, J., et al.: High performance membrane electrode assembly fabricated by ultrasonic spray technique. ECS Trans. 41, 901–907 (2019). https://doi.org/10.1149/1.3635624
Huang, T.H., Shen, H.L., Jao, T.C., et al.: Ultra-low Pt loading for proton exchange membrane fuel cells by catalyst coating technique with ultrasonic spray coating machine. Int. J. Hydrog. Energy 37, 13872–13879 (2012). https://doi.org/10.1016/j.ijhydene.2012.04.108
Jao, T.C., Jung, G.B., Shen, H.L., et al.: Ultrasonic spray coating for proton exchange membrane fuel cell. Open J. Acoust. 3, 33–37 (2013). https://doi.org/10.4236/oja.2013.33a006
Chaparro, A.M., Folgado, M.A., Ferreira-Aparicio, P., et al.: Properties of catalyst layers for PEMFC electrodes prepared by electrospray deposition. J. Electrochem. Soc. 157, B993–B999 (2010). https://doi.org/10.1149/1.3425740
Wang, J., Xu, H., Huo, Y., et al.: Progress of electrospray and electrospinning in energy applications. Nanotechnology 31, 132001 (2020). https://doi.org/10.1088/1361-6528/ab52bb
Soares, R.M.D., Siqueira, N.M., Prabhakaram, M.P., et al.: Electrospinning and electrospray of bio-based and natural polymers for biomaterials development. Mater. Sci. Eng. C 92, 969–982 (2018). https://doi.org/10.1016/j.msec.2018.08.004
Levitt, A.S., Vallett, R., Dion, G., et al.: Effect of electrospinning processing variables on polyacrylonitrile nanoyarns. J. Appl. Polym. Sci. 135, 46404 (2018). https://doi.org/10.1002/app.46404
Son, W.K., Youk, J.H., Lee, T.S., et al.: The effects of solution properties and polyelectrolyte on electrospinning of ultrafine poly(ethylene oxide) fibers. Polymer 45, 2959–2966 (2004). https://doi.org/10.1016/j.polymer.2004.03.006
Baturina, O.A., Wnek, G.E.: Characterization of proton exchange membrane fuel cells with catalyst layers obtained by electrospraying. Electrochem. Solid-State Lett. 8, A267 (2005). https://doi.org/10.1149/1.1895267
Benítez, R., Soler, J., Daza, L.: Novel method for preparation of PEMFC electrodes by the electrospray technique. J. Power Sources 151, 108–113 (2005). https://doi.org/10.1016/j.jpowsour.2005.02.047
Chaparro, A.M., Benítez, R., Gubler, L., et al.: Study of membrane electrode assemblies for PEMFC, with cathodes prepared by the electrospray method. J. Power Sources 169, 77–84 (2007). https://doi.org/10.1016/j.jpowsour.2007.01.044
Chaparro, A.M., Gallardo, B., Folgado, M.A., et al.: PEMFC electrode preparation by electrospray: optimization of catalyst load and ionomer content. Catal. Today 143, 237–241 (2009). https://doi.org/10.1016/j.cattod.2008.12.003
Úbeda, D., Cañizares, P., Ferreira-Aparicio, P., et al.: Life test of a high temperature PEM fuel cell prepared by electrospray. Int. J. Hydrog. Energy 41, 20294–20304 (2016). https://doi.org/10.1016/j.ijhydene.2016.09.109
Yoo, J.H., Choi, B.H., Koh, B.S., et al.: Fabrication of practical PEMFC electrode with ultralow mass loading of platinum via electro-spray deposition technique. J. Ceram. Process. Res. 18, 203–206 (2017)
Waldrop, K., Wycisk, R., Pintauro, P.N.: Application of electrospinning for the fabrication of proton-exchange membrane fuel cell electrodes. Curr. Opin. Electrochem. 21, 257–264 (2020). https://doi.org/10.1016/j.coelec.2020.03.007
Park, J.H., Ju, Y.W., Park, S.H., et al.: Effects of electrospun polyacrylonitrile-based carbon nanofibers as catalyst support in PEMFC. J. Appl. Electrochem. 39, 1229–1236 (2009). https://doi.org/10.1007/s10800-009-9787-4
Chan, S., Jankovic, J., Susac, D., et al.: Electrospun carbon nanofiber catalyst layers for polymer electrolyte membrane fuel cells: structure and performance. J. Power Sources 392, 239–250 (2018). https://doi.org/10.1016/j.jpowsour.2018.02.001
Wang, L.B., Chen, S., Shu, T., et al.: Functional inks for printable energy storage applications based on 2 D materials. Chemsuschem 13, 1330–1353 (2020). https://doi.org/10.1002/cssc.201902019
Santangelo, P., Cannio, M., Romagnoli, M.: Review of catalyst-deposition techniques for PEMFC electrodes. Techn. Italian J. Eng. Sci. 63, 65–72 (2019). https://doi.org/10.18280/ti-ijes.630109
Fromm, J.E.: Numerical calculation of the fluid dynamics of drop-on-demand jets. IBM J. Res. Dev. 28, 322–333 (1984). https://doi.org/10.1147/rd.283.0322
Towne, S., Viswanathan, V., Holbery, J., et al.: Fabrication of polymer electrolyte membrane fuel cell MEAs utilizing inkjet print technology. J. Power Sources 171, 575–584 (2007). https://doi.org/10.1016/j.jpowsour.2007.07.017
Taylor, A.D., Kim, E.Y., Humes, V.P., et al.: Inkjet printing of carbon supported platinum 3-D catalyst layers for use in fuel cells. J. Power Sources 171, 101–106 (2007). https://doi.org/10.1016/j.jpowsour.2007.01.024
Saha, M.S., Paul, D., Malevich, D., et al.: Preparation of ultra-thin catalyst layers by piezo-electric printer for PEMFCs applications. ECS Trans. 25, 2049–2059 (2019). https://doi.org/10.1149/1.3210761
Malevich, D., Saha, M.S., Halliop, E., et al.: Performance characteristics of PEFCs with patterned electrodes prepared by piezo-electric printing. ECS Trans. 50, 423–427 (2013). https://doi.org/10.1149/05002.0423ecst
Shukla, S., Domican, K., Secanell, M.: Effect of electrode patterning on PEM fuel cell performance using ink-jet printing method. ECS Trans. 64, 341–352 (2014). https://doi.org/10.1149/06403.0341ecst
Yazdanpour, M., Esmaeilifar, A., Rowshanzamir, S.: Effects of hot pressing conditions on the performance of Nafion membranes coated by ink-jet printing of Pt/MWCNTs electrocatalyst for PEMFCs. Int. J. Hydrog. Energy 37, 11290–11298 (2012). https://doi.org/10.1016/j.ijhydene.2012.04.139
Saha, M.S., Tam, M., Berejnov, V., et al.: Characterization and performance of catalyst layers prepared by inkjet printing technology. ECS Trans. 58, 797–806 (2013). https://doi.org/10.1149/05801.0797ecst
Shukla, S., Domican, K., Karan, K., et al.: Analysis of low platinum loading thin polymer electrolyte fuel cell electrodes prepared by inkjet printing. Electrochim. Acta 156, 289–300 (2015). https://doi.org/10.1016/j.electacta.2015.01.028
Shukla, S., Domican, K., Secanell, M.: Analysis of kinetic parameters and effect of Pt loading on cell performance of PEFC electrodes prepared by inkjet printing. ECS Trans. 69, 761–772 (2015). https://doi.org/10.1149/06917.0761ecst
Acknowledgements
This work was financially supported by the National Natural Science Foundation (No. 21676204) and the Program of Ministry of Science and Technology of China (No. 2018YFB0106503).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Guo, Y., Pan, F., Chen, W. et al. The Controllable Design of Catalyst Inks to Enhance PEMFC Performance: A Review. Electrochem. Energ. Rev. 4, 67–100 (2021). https://doi.org/10.1007/s41918-020-00083-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41918-020-00083-2