Abstract
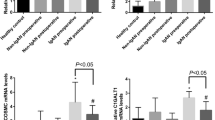
The benefits of tonsillectomy in IgA nephropathy (IgAN) are still debated. Tonsillectomy may remove pathogen sources and reduce the mucosal associated lymphoid tissue (MALT), limiting degalactosylated IgA1 (deGal-IgA1) production, which is considered to be the initiating pathogenetic event leading to IgA glomerular deposition. In the European network VALIGA, 62/1147 IgAN patients underwent tonsillectomy (TxIgAN). In a cross-sectional study 15 of these patients were tested and compared to 45 non-tonsillectomized IgAN (no-TxIgAN) and healthy controls (HC) regarding levels of deGal-IgA1, and markers of innate immunity and oxidative stress, including toll-like receptors (TLR)2, 3, 4 and 9 mRNAs, proteasome (PS) and immunoproteasome (iPS) mRNAs in peripheral blood mononuclear cells (PBMC), and advanced oxidation protein products (AOPP). Levels of deGal-IgA1 were lower in TxIgAN than in no-TxIgAN (p = 0.015), but higher than in HC (p = 0.003). TLR mRNAs were more expressed in TxIgAN than in HC (TLR4, p = 0.021; TLR9, p = 0.027), and higher in TxIgAN than in no-TxIgAN (p ≤ 0.001 for TLR2, 4, 9). A switch from PS to iPS was detected in PBMC of TxIgAN in comparison to HC and it was higher than in no-TxIgAN [large multifunctional peptidase (LMP)2/β1, p = 0.039; LPM7/β5, p < 0.0001]. The levels of AOPP were significantly higher in TxIgAN than HC (p < 0.001) and no-TxIgAN (p = 0.033). In conclusion, the activation of innate immunity via TLRs and ubiquitin–proteasome pathways and the pro-oxidative milieu were not affected by tonsillectomy, even though the levels of aberrantly galactosylated IgA1 were lower in patients with IgAN who had tonsillectomy. The residual hyperactivation of innate immunity in tonsillectomized patients may result from extra-tonsillar MALT.




Similar content being viewed by others
References
Berger J, Hinglais N (1968) Intercapillary deposits of IgA–IgG. J Urol Nephrol 74:694–695
Coppo R, Amore A, Peruzzi L, Vergano L, Camilla R (2010) Innate immunity and IgA nephropathy. J Nephrol 23:626–632
Mogensen TH (2009) Pathogen recognition and inflammatory signaling in innate immune defenses. Clin Microbiol Rev 22:240–273
Rasmussen SB, Reinert LS, Paludan SR (2009) Innate recognition of intracellular pathogens: detection and activation of the first line of defense. APMIS 117:323–337
Mellanby RJ, Thomas DC, Lamb J (2009) Role of regulatory T-cells in autoimmunity. Clin Sci (Lond) 116:639–649
Anders HJ, Muruve DA (2011) The inflammasomes in kidney disease. J Am Soc Nephrol 22:1007–1018
Rock KL, York IA, Saric T (2002) Protein degradation and the generation of MHC class I-presented peptides. Adv Immunol 80:1–70
Macagno A, Kuehn L, de Giuli R et al (2001) Pronounced up-regulation of the PA28alpha/beta proteasome regulator but little increase in the steady-state content of immunoproteasome during dendritic cell maturation. Eur J Immunol 31:3271–3280
Foersch S, Waldner MJ, Neurath MF (2013) Innate and adaptive immunity in inflammatory bowel diseases. Dig Dis 31:317–320
Kiryluk K, Li Y, Sanna-Cherchi S, Rohanizadegan M et al (2012) Geographic differences in genetic susceptibility to IgA nephropathy: GWAS replication study and geospatial risk analysis. PLoS Genet 8:e1002765
Gesualdo L, Lamm ME, Emancipator SN (1990) Defective oral tolerance promotes nephritogenesis in experimental IgA nephropathy induced by oral immunization. J Immunol 145:3684–3691
Amore A, Coppo R, Nedrud JG, Sigmund N, Lamm ME, Emancipator SN (2004) The role of nasal tolerance in a model of IgA nephropathy induced in mice by Sendai virus. Clin Immunol 113:101–108
Coppo R, Amore A (2004) Aberrant glycosylation in IgA nephropathy (IgAN). Kidney Int 65:1544–1547
Suzuki H, Fan R, Zhang Z, Brown R et al (2009) Aberrantly glycosylated IgA1 in IgA nephropathy patients is recognized by IgG antibodies with restricted heterogeneity. J Clin Invest 119:1668–1677
Suzuki H et al (2011) The pathophysiology of IgA nephropathy. J Am Soc Nephrol 22:1795–1803
Rasche FM, Schwarz A, Keller F (1999) Tonsillectomy does not prevent a progressive course in IgA nephropathy. Clin Nephrol 51:147–152
Piccoli A, Codognotto M, Tabbi MG, Favaro E, Rossi B (2010) Influence of tonsillectomy on the progression of mesangioproliferative glomerulonephritis. Nephrol Dial Transplant 25:2583–2589
KDIGO Guidelines on Glomerulonephritis (2012) Kidney International Supplement 2:218–220
Chen Y, Tang Z, Wang Q et al (2007) Long-term efficacy of tonsillectomy in Chinese patients with IgA nephropathy. Am J Nephrol 27:170–175
Miura N, Imai H, Kikuchi S et al (2009) Tonsillectomy and steroid pulse (TSP) therapy for patients with IgA nephropathy: a nationwide survey of TSP therapy in Japan and an analysis of the predictive factors for resistance to TSP therapy. Clin Exp Nephrol 13:460–466
Tsuchiya T, Ito S, Yamaguchi Y, Moriyama Y, Ehara H, Deguchi T (2010) Tonsillectomy and steroid pulse therapy for recurrent IgA nephropathy in renal allograft. Clin Nephrol 73:68–71
Coppo R, Troyanov S, Bellur S, et al, on behalf of the VALIGA study of the ERA–EDTA Immunonephrology Working Group (2014) The predictive value of the Oxford classification of IgA nephropathy in cohorts with different presentations and receiving different treatments: the VALIGA study. Kidney Int [Epub ahead of print]
Coppo R, Camilla R, Amore A et al (2010) Toll-like receptor 4 expression is increased in circulating mononuclear cells of patients with immunoglobulin A nephropathy. Clin Exp Immunol 159:73–81
Coppo R, Camilla R, Alfarano A et al (2009) Upregulation of the immunoproteasome in peripheral blood mononuclear cells of patients with IgA nephropathy. Kidney Int 75:536–541
Camilla R, Suzuki H, Daprà V et al (2011) Oxidative stress and galactose-deficient IgA1 as markers of progression in IgA nephropathy. Clin J Am Soc Nephrol 6:1903–1911
Descamps-Latscha B, Witko-Sarsat V, Nguyen-Khoa T et al (2004) Early prediction of IgA nephropathy progression: proteinuria and AOPP are strong prognostic markers. Kidney Int 66:1606–1612
Cattran DC, Coppo R, Cook HT et al (2009) The Oxford classification of IgA nephropathy: rationale, clinicopathological correlations, and classification. Kidney Int 76:534–545
Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D, Modification of Diet in Renal Disease Study Group (1999) A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Ann Intern Med 130:461–470
Amore A, Cirina P, Conti G, Brusa P, Peruzzi L, Coppo R (2001) Glycosylation of circulating IgA in patients with IgA nephropathy modulates proliferation and apoptosis of mesangial cells. J Am Soc Nephrol 12:1862–1871
Wyatt RJ, Julian BA (2013) IgA nephropathy. N Engl J Med 368:2402–2414
Hellings P, Jorissen M, Ceuppens JL (2000) The Waldeyer’s ring. Acta Otorhinolaryngol 54:237–241
Russell MW, Mestecky J, Julian BA, Galla JH (1986) IgA-associated renal diseases: antibodies to environmental antigens in sera and deposition of immunoglobulins and antigens in glomeruli. J Clin Immunol 6:74–86
Wang J, Anders RA, Wu Q et al (2004) Dysregulated LIGHT expression on T cells mediates intestinal inflammation and contributes to IgA nephropathy. J Clin Invest 113:826–835
Smerud HK, Fellström B, Hällgren R et al (2009) Gluten sensitivity in patients with IgA nephropathy. Nephrol Dial Transplant 24:2476–2481
Emancipator SN, Gallo GR, Lamm ME (1983) Experimental IgA nephropathy induced by oral immunization. J Exp Med 157:572–582
Coppo R, Mazzucco G, Martina G et al (1989) Gluten-induced experimental IgA glomerulopathy. Lab Invest 60:499–506
Kloster Smerud H, Fellström B, Hällgren R, Osagie S, Venge P, Kristjánsson G (2010) Gastrointestinal sensitivity to soy and milk proteins in patients with IgA nephropathy. Clin Nephrol 74:364–371
López Castro J, Mouronte Sánchez MC, Pérez Carral O, Núñez Calvo L (2004) Celiac disease and IgA nephropathy: a double face coin? Med Clin 122:158–159
Ramezani A, Raj DS (2014) The gut microbiome, kidney disease, and targeted interventions. J Am Soc Nephrol 25:657–670
Anders HJ, Banas B, Schlöndorff D (2004) Signaling danger: toll-like receptors and their potential roles in kidney disease. J Am Soc Nephrol 15:854–867
McCarthy DD, Kujawa J, Wilson C et al (2011) Mice overexpressing BAFF develop a commensal flora-dependent, IgA-associated nephropathy. J Clin Invest 121:3991–4002
Brown HJ, Lock HR, Wolfs TG et al (2007) Toll-like receptor 4 ligation on intrinsic renal cells contributes to the induction of antibody-mediated glomerulonephritis via CXCL1 and CXCL2. J Am Soc Nephrol 18:1732–1739
Yoon HJ, Shin JH, Yang SH, Chae DW et al (2003) Association of the CD14 gene-159C polymorphism with progression of IgA nephropathy. J Med Genet 40:104–108
McCarthy DD, Chiu S, Gao Y et al (2006) BAFF induces a hyper-IgA syndrome in the intestinal lamina propria concomitant with IgA deposition in the kidney independent of LIGHT. Cell Immunol 241:85–94
Conflict of interest
No author has any conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vergano, L., Loiacono, E., Albera, R. et al. Can tonsillectomy modify the innate and adaptive immunity pathways involved in IgA nephropathy?. J Nephrol 28, 51–58 (2015). https://doi.org/10.1007/s40620-014-0086-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-014-0086-8