Abstract
Aim
To determine the clinical non-inferiority of recombinant glargine-Basalin vs glargine-Lantus, in treatment of type 2 diabetes mellitus (T2DM) using continuous glucose monitoring system (CGMS).
Methods
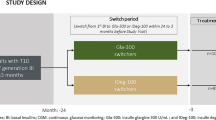
One hundred patients with T2DM were recruited. They were either regularly taking Basalin (Basalin group) or Lantus (Lantus group) (n = 50 each). CGMS was employed to real-time monitor blood glucose profile for 4 days (from day 1 to day 5). To exclude the effect of patient background, the study design was to have a blinded crossover from glargine-Basalin to glargine-Lantus on day 3, and vice versa. 24-hour mean blood glucose (24hMBG), 24-hour standard deviation of blood glucose (24hSDBG), 24-hour mean amplitude of glycemic excursion (24hMAGE), and number of glycemic excursion (NGE) every 24 h (24hNGE) were calculated for each glargine from 100 patients.
Results
No significant difference of 24hMBG, 24hSDBG, 24hMAGE, and 24hNGE (p > 0.05 for all) was found between Basalin and Lantus treatments. The glucose area under the curve and time when blood glucose was below 3.9 mmol/L, between 3.9 and 10.0 mmol/L, or above 10.0 mmol/L were similar between Basalin and Lantus treatment. The frequency of hypoglycemic episodes was also similar. However, the mean cost of Basalin was only 72% of Lantus’s in one treatment course.
Conclusion
Glargine-Basalin is non-inferior in clinical efficacy compared to glargine-Lantus. In view of the large difference in the cost of glargine-Basalin, it would be much more cost-effective for our patients.

Similar content being viewed by others
References
Liu Z, Fu C, Wang W, Xu B (2010) Prevalence of chronic complications of type 2 diabetes mellitus in outpatients—a cross-sectional hospital based survey in urban China. Health Qual Life Outcomes 8:62
Wang W, McGreevey WP, Fu C, Zhan S, Luan R, Chen W, Xu B (2009) Type 2 diabetes mellitus in China: a preventable economic burden. Am J Manag Care 15:593–601
Roglic G, Unwin N (2010) Mortality attributable to diabetes: estimates for the year 2010. Diabetes Res Clin Pract 87:15–19
Danaei G, Lawes CM, Vander Hoorn S, Murray CJ, Ezzati M (2006) Global and regional mortality from ischaemic heart disease and stroke attributable to higher-than-optimum blood glucose concentration: comparative risk assessment. Lancet 368:1651–1659
Moghissi ES, Korytkowski MT, DiNardo M, Einhorn D, Hellman R, Hirsch IB, Inzucchi SE, Ismail-Beigi F, Kirkman MS, Umpierrez GE, American Association of Clinical E, American Diabetes A (2009) American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Diabetes Care 32:1119–1131
Hirsch IB (2015) Glycemic variability and diabetes complications: does it matter? Of course it does! Diabetes Care 38:1610–1614
Brownlee M, Hirsch IB (2006) Glycemic variability: a hemoglobin A1c-independent risk factor for diabetic complications. JAMA 295:1707–1708
Wang F, Carabino JM, Vergara CM (2003) Insulin glargine: a systematic review of a long-acting insulin analogue. Clin Ther 25:1541–1577 (discussion 1539–1540)
Deiss D, Kordonouri O, Hartmann R, Hopfenmuller W, Lupke K, Danne T (2007) Treatment with insulin glargine reduces asymptomatic hypoglycemia detected by continuous subcutaneous glucose monitoring in children and adolescents with type 1 diabetes. Pediatr Diabetes 8:157–162
Goykhman S, Drincic A, Desmangles JC, Rendell M (2009) Insulin glargine: a review 8 years after its introduction. Expert Opin Pharmacother 10:705–718
Guo W, Li F, Chen P, Lin Y, Xu X (2014) Efficacy and safety of liraglutide compared with insulin glargine in patients with type 2 diabetes mellitus inadequately controlled with metformin. Chin J Endocrinol Metab 30:1086–1091
Grunberger G (2014) Insulin analogs—are they worth it? Yes! Diabetes Care 37:1767–1770
Zdarska DJ, Kvapil M, Rusavy Z, Krcma M, Broz J, Krivska B, Kadlecova P (2014) Comparison of glucose variability assessed by a continuous glucose-monitoring system in patients with type 2 diabetes mellitus switched from NPH insulin to insulin glargine: the COBIN2 study. Wien Klin Wochenschr 126:228–237
Wang XL, Lu JM, Pan CY, Mu YM, Dou JT, Ba JM, Wang X (2007) Evaluation of the superiority of insulin glargine as basal insulin replacement by continuous glucose monitoring system. Diabetes Res Clin Pract 76:30–36
Chen SW, Lu JM, Pan CY, Wang BA, Wang YZ, Li YJ, Yang G, Yan WH (2010) Studies of pharmacokinetic, pharmacodynamic properties and bioequivalence of recombinant insulin glargine injection in healthy man. Chin J Diabetes 18:387–391
Breton MD, Shields DP, Kovatchev BP (2008) Optimum subcutaneous glucose sampling and Fourier analysis of continuous glucose monitors. J Diabetes Sci Technol 2:495–500
Vashist SK (2013) Continuous glucose monitoring systems: a review. Diagnostics (Basel) 3:385–412
Ceriello A, Ihnat MA (2010) ‘Glycaemic variability’: a new therapeutic challenge in diabetes and the critical care setting. Diabet Med 27:862–867
Hsu CW (2012) Glycemic control in critically ill patients. World J Crit Care Med 1:31–39
Yang W, Lu J, Weng J, Jia W, Ji L, Xiao J, Shan Z, Liu J, Tian H, Ji Q, Zhu D, Ge J, Lin L, Chen L, Guo X, Zhao Z, Li Q, Zhou Z, Shan G, He J, China National D, Metabolic Disorders Study G (2010) Prevalence of diabetes among men and women in China. N Engl J Med 362:1090–1101
Hu H, Sawhney M, Shi L, Duan S, Yu Y, Wu Z, Qiu G, Dong H (2015) A systematic review of the direct economic burden of type 2 diabetes in China. Diabetes Ther 6:7–16
Williams R, Van Gaal L, Lucioni C, Board C-A (2002) Assessing the impact of complications on the costs of Type II diabetes. Diabetologia 45:S13–S17
Acknowledgements
The study was supported by the fund of Scientific and Technological Development Program of Jiangsu Province of China (BL2014010).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This crossover study was approved by Ethical Committee of Nanjing First Hospital, Nanjing Medical University, Nanjing, China.
Informed consent
Informed consents were signed by all patients.
Rights and permissions
About this article
Cite this article
Li, H.Q., Lu, C.F., Wang, J. et al. A comparison of clinical efficacy and economic value in Basalin- and Lantus-treated patients with type 2 diabetes using continuous glucose monitoring system. J Endocrinol Invest 41, 179–184 (2018). https://doi.org/10.1007/s40618-017-0712-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-017-0712-0