Abstract
Introduction
Pharmacovigilance (PV) systems around the world vary in capacity and maturity. Many factors can influence PV.
Objectives
This systematic scoping review aims to explore the opportunities for effective PV in the Middle East and North Africa (MENA) region by (1) using the pharmacogovernance (PG) framework to evaluate the PV systems and (2) determining the factors influencing PV, including regulatory governance, PG, and international actors influence.
Methods
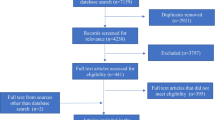
In accordance with Cochrane Collaboration guidelines, the STARLITE criteria were used to guide the systematic literature search and review process. The search included English and Arabic language databases, search engines, and grey literature and a supplementary search of the reference lists of included articles. Inclusion criteria were pertinent to the influence of PV, governance and health, PG framework domains, international actors, and MENA countries. Data synthesis followed the meta-aggregation process.
Results
From the 32 included records, the majority of PV-enhanced actions were from Saudi Arabia, Iran, and Morocco. The ‘policy, law, and regulations’ domain was the strongest in the MENA, whereas public ‘participation and representation’ was lacking. Further, the PG framework allowed the identification of shortcomings, including a lack of public inclusiveness and participation, resource scarcity, misalignment of resource allocation, issues with existing regulations, and deficiencies in ‘accountability and transparency’.
Conclusion
Most of the existing systems are still in their infancy, and disparities exist at national and regional levels. The operationalization of effective PV systems requires actions that assure (1) the development and enforcement of comprehensive regulations, (2) adequate and equitable aid for core PV processes, and (3) accountability and participation from all stakeholders.


Similar content being viewed by others
References
World Health Organization. The importance of pharmacovigilance-safety monitoring of medicinal products. Geneva: WHO; 2002.
Pharmacovigilance WH. Ensuring the safe use of medicines, WHO policy perspective of medicine. Geneva: WHO; 2004. p. 1222–39.
Supporting pharmacovigilance in developing Countries: the systems perspective. 1st ed. Arlington: Management Sciences for Health; 2009 [cited 8 Mar 2017]. http://apps.who.int/medicinedocs/documents/s18813en/s18813en.pdf.
Olsson S, Pal SN, Stergachis A, Couper M. Pharmacovigilance activities in 55 low-and-middle-income countries. Drug Saf. 2010;33(8):689–703.
Olsson S, Pal SN, Dodoo A. Pharmacovigilance in resource-limited countries. Expert Rev Clin Pharmacol. 2015;8(4):449–60.
The Middle East and North Africa. Worldbank.org. 2017 [cited 25 Mar 2017]. http://www.worldbank.org/en/region/mena.
Ahmad SR. Pharmacovigilance bolstered in the Arab world. Lancet. 2014;384(9960):e63.
Bham B. The first Eastern Mediterranean region/Arab countries meeting of pharmacovigilance. Drugs Real World Outcomes. 2015;2(1):111–5.
Moscou K, Kohler JC, MaGahan A. Governance and pharmacovigilance in Brazil: a scoping review. J Pharm Policy Pract. 2016;9(1):3.
Mackey TK, Liang BA. Combating healthcare corruption and fraud with improved global health governance. BMC Int Health Hum Rights. 2012;12(1):23.
Baghdadi‐Sabeti G, Cohen-Kohler J, Wondemagegnehu E. Measuring transparency in the public pharmaceutical sector-Assessment instrument. Geneva: World Health Organization, Departments of Essential Medicines and Pharmaceutical Policies and Ethics, Equity, Trade and Human Rights; 2009.
Moscou K, Kohler JC. Matching safety to access: global actors and pharmacogovernance in Kenya—a case study. Global Health. 2017;13(1):20.
United Nations Development Programme. Governance for sustainable human development: a UNDP policy document. New York: United Nations Development Programme; 1997.
Annan KA. Partnerships for global community: annual report on the work of the organization, 1998. United Nations, Department of Public Information; 1998.
Siddiqi S, Masud TI, Nishtar S, Peters DH, Sabri B, Bile KM, Jama MA. Framework for assessing governance of the health system in developing countries: gateway to good governance. Health Policy. 2009;90(1):13–25.
Anello E. A framework for good governance in the public pharmaceutical sector. Geneva: World Health Organization, Department of Essential Medicines and Pharmaceutical policies; 2008.
Ratanawijitrasin S, Wondemagegnehu E. Effective drug regulation. A multicountry study. Geneva: World Health Organization; 2002.
Moscou K, Kohler JC, Lexchin J. Drug safety and corporate governance. Glob Health Gov. 2013;7(1):68–9.
Rentz ED, Lewis L, Mujica OJ, Barr DB, Schier JG, Weerasekera G, Kuklenyik P, McGeehin M, Osterloh J, Wamsley J, Lum W. Outbreak of acute renal failure in Panama in 2006: a case–control study. Bull World Health Organ. 2008;86(10):749–56.
WHO Uppsala Monitoring Centre (UMC). The WHO Programme. [cited 15 Mar 2017]. http://www.who-umc.org.
Tsintis P, La Mache E. CIOMS and ICH initiatives in pharmacovigilance and risk management: overview and implications. Drug Saf. 2004;27(8):509–17.
Olsson S. The role of the WHO programme on international drug monitoring in coordinating worldwide drug safety efforts. Drug Saf. 1998;19(1):1.
Nwokike J. Technical assistance for the establishment of a pharmacovigilance and medicine safety system in Rwanda. Submitted to the US Agency for International Development by the Strengthening Pharmaceutical Systems (SPS) Program. Arlington: Management Sciences for Health; 2009.
Higgins JP, Green S, editors. Cochrane handbook for systematic reviews of interventions. New York: Wiley; 2011.
Supplemental Handbook Guidance | Cochrane Qualitative and Implementation. Methods.cochrane.org. 2017 [cited 15 May 2017]. http://methods.cochrane.org/qi/supplemental-handbook-guidance.
Armstrong R, Hall BJ, Doyle J, Waters E. ‘Scoping the scope’of a Cochrane review. J Public Health (Oxf). 2011;33(1):147–50.
Moher D, Liberati A, Tetzlaff J, Altman DG, Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097.
Schöpfel J, Farace DJ. Grey literature. Encyclopedia of library and information sciences. 3rd ed. Boca Raton, FL: CRC Press; 2009. p. 2029–39.
Munro S, Lewin S, Smith H, Engel M, Fretheim A, Volmink J. Adherence to tuberculosis treatment: a qualitative systematic review of stakeholder perceptions. PLOS Medicine. 2007;4(7):e238.
Noyes J, Lewin S. Chapter 5: extracting qualitative evidence. Supplementary guidance for inclusion of qualitative research in Cochrane systematic reviews of interventions. Version. 2011;1:1–24.
Wilbur K. Pharmacovigilance in Qatar: a survey of pharmacists/Pharmacovigilance au Qatar: enquete aupres des pharmaciens. East Mediterr Health J. 2013;19(11):930.
Wilbur K. Pharmacovigilance in the Middle East. Drug Saf. 2013;36(1):25–30.
Shalviri G, Mohammad K, Majdzadeh R, Gholami K. Applying quantitative methods for detecting new drug safety signals in pharmacovigilance national database. Pharmacoepidemiol Drug Saf. 2007;16(10):1136–40.
Bencheikh RS, Benabdallah G. Medication errors: pharmacovigilance centres in detection and prevention. Br J Clin Pharmacol. 2009;67(6):687–90.
Baniasadi S, Habibi M, Haghgoo R, Gamishan MK, Dabaghzadeh F, Farasatinasab M, Farsaei S, Gharekhani A, Kafi H, Karimzadeh I, Kharazmkia A. Increasing the number of adverse drug reactions reporting: the role of clinical pharmacy residents. Iran J Pharm Res. 2014;13(1):291.
Baniasadi S, Fahimi F, Shalviri G. Developing an adverse drug reaction reporting system at a teaching hospital. Basic Clin Pharmacol Toxicol. 2008;102(4):408–11.
Al-Worafi YM. Comment on: “Pharmacovigilance in the Middle East”. Drug Saf. 2014;37(8):651.
Alshammari TM, Wa’ad H, Le Louet H, Aljadhey HS. Completeness of adverse drug reactions reports of the Saudi adverse event reporting system. Saudi Med J. 2015;36(7):821–8.
Wilbur K. How to improve suspected ADR reporting among pharmacists in the GCC? Saudi Pharm J. 2016;24(6):718.
Khan LM, Al-Harthi SE, Alkreathy HM, Osman AM, Ali AS. Detection of adverse drug reactions by medication antidote signals and comparison of their sensitivity with common methods of ADR detection. Saudi Pharm J. 2015;23(5):515–22.
Alshammari T, Alshakka M, Aljadhey H. Pharmacovigilance system in Saudi Arabia. Saudi Pharm J. 2017;25(3):299–305.
Aljadhey H, Mahmoud MA, Alshammari TM, Al-Dhaeefi M, Le Louet H, Perez-Gutthann S, Pitts PJ. A qualitative exploration of the major challenges facing pharmacovigilance in Saudi Arabia. Saudi Med J. 2015;36(9):1097–102.
Swamy S, Mourya M, Kadhe G, Mane A, Sawant S. Safety reporting through a comprehensive and pragmatic pharmacovigilance process for India and emerging markets: an industry perspective. Expert Opin Drug Saf. 2015;14(9):1409–20.
Cheaib N. Pharmacovigilance in clinical trials: current practice and challenges. Account Res. 2016;23(1):23–30.
Batniji R, Khatib L, Cammett M, Sweet J, Basu S, Jamal A, Wise P, Giacaman R. Governance and health in the Arab world. Lancet. 2014;383(9914):343–55.
Baniasadi S, Fahimi F, Namdar R. Development of an adverse drug reaction bulletin in a teaching hospital. Formulary. 2009;44:333–5.
Yadav S. Status of adverse drug reaction monitoring and pharmacovigilance in selected countries. Indian J Pharmacol. 2008;40(Suppl 1):S4–9.
Peiravian F. Parallel import: is it worth? Iran J Pharm Res. 2014;13(4):1111.
Shalviri G, Valadkhani M, Dinarvand R. Ten years pharmacovigilance activities in Iran. Iran J Public Health. 2009;38(Suppl 1):162–5.
Shankar P, Jha N, Aljadhey H, Pitts P. A qualitative exploration of the major challenges facing pharmacovigilance in Saudi Arabia. Saudi Med J. 2015;36(11):1369–70.
Wilby KJ, Hazi HM, Ashour MA. Development, implementation and evaluation of a medication safety programme for schoolchildren in Qatar/Developpement, mise en oeuvre et evaluation d’un programme de surete des medicaments pour les ecoliers au Qatar. East Mediterr Health J. 2015;21(1):62.
Gholami K, Shalviri G, Zarbakhsh A, Daryabari N, Yousefian S. New guideline for tramadol usage following adverse drug reactions reported to the Iranian Pharmacovigilance Center. Pharmacoepidemiol Drug Saf. 2007;16(2):229–37.
Fatak A. Rules of procedure applicable to medicines used in human medicine to ensure their safety under Algerian law. Egypt Contemp Mag. 2008;100(489):477–507.
Mur S. Medicine and the obligations imposed in its scope. Leg Stud Period Rev. 2013;18:9–28.
Khan LM, Al-Harthi SE, Saadah OI. Adverse drug reactions in hospitalized pediatric patients of Saudi Arabian University Hospital and impact of pharmacovigilance in reporting ADR. Saudi Pharm J. 2013;21(3):261–6.
Wilbur K. Pharmacovigilance in Qatar hospitals. Pharm Med. 2012;26(1):23–5.
Al-Essa R, Al-Rubaie M, Walker S, Salek S. Pharmaceutical regulatory environment: challenges and opportunities in the Gulf region. 1st ed. Cham: Springer International Publishing; 2015.
Fathelrahman A, Ibrahim M, Wertheimer A. Pharmacy practice in developing countries. 1st ed. London: Academic Press; 2016.
Qato DM. Current state of pharmacovigilance in the Arab and Eastern Mediterranean region: results of a 2015 survey. Int J Pharm Pract. 2017. doi:10.1111/ijpp.12372.
Kohler JC, Ovtcharenko N. Good governance for medicines initiatives: exploring lessons learned. U4 Anti-Corrupt Resour Centre. 2013;3:1-26.
Naffa S, Nuseirat A. Medicine transparency alliance Jordan Workplan. [cited 30 Sept 2017]. http://www.medicinestransparency.org/fileadmin/uploads/Renee/MeTA__Jordan_Workplan.pdf.
World Health Organization. Minimum requirements for a functional pharmacovigilance system. Geneva: WHO; 2010.
Lexchin J. Harmony in drug regulation, but who’s calling the tune? An examination of regulatory harmonization in health Canada. Int J Health Serv. 2012;42(1):119–36.
Arthur N, Bentsi-Enchill A, Couper R, Duclos P, Edwards I, Fushimi T, Hartigan-Go K, Folb P, Kshirsagar N, Lazdins-Helds J, Lepahkin V. The importance of pharmacovigilance-safety monitoring of medicinal products. Geneva: World Health Organization; 2002.
Forman L, Kohler JC. Access to medicines as a human right: implications for pharmaceutical industry responsibility. Toronto: University of Toronto Press; 2012.
World Health Organization. WHO pharmacovigilance indicators: a practical manual for the assessment of pharmacovigilance systems. Geneva, Switzerland: WHO; 2015.
Acknowledgements
English language editing of the manuscript was undertaken by American Journal Experts.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This systematic scoping review was funded by Qatar University (Grant number: QUST-2-CPH-2017-7).
Conflict of interest
MI and AH have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hamid, A.A., Mohamed Ibrahim, M.I. A Systematic Scoping Review of the State of Pharmacovigilance and Governance in the MENA Region: Challenges and Opportunities. Pharm Med 31, 437–454 (2017). https://doi.org/10.1007/s40290-017-0212-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40290-017-0212-8