Abstract
Background
Extended-interval aminoglycoside (EIAG) and extended- and continuous-infusion β-lactam (EIBL and CIBL) dosing strategies are increasingly used in adults, but pediatric literature is limited.
Objective
The objective of this study was to describe the use of EIAG, EIBL, and CIBL dosing in pediatric hospitals in the USA.
Study Design, Setting, and Participants
A national survey of children’s hospitals was conducted. A single practitioner from each target hospital was identified through the Children’s Hospital Association. Practice-based survey questions identified whether hospitals utilize EIAG, EIBL, and CIBL dosing.
Main Outcome Measure
The main outcome measure was the percentage utilization of the dosing strategies, with secondary outcomes being the reasons for not using these dosing strategies.
Results
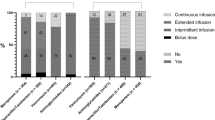
Seventy-seven of 215 identified practitioners (36 %) participated in the survey. EIAG, EIBL, and CIBL dosing were utilized in 63 %, 24 %, and 13 % of responding hospitals, respectively. The most common reasons for not using EIAG were concern regarding lack of efficacy data (56 %) and concern regarding the duration of the drug-free period (41 %). Respondents who did not utilize EIBL cited concern due to lack of pediatric EIBL efficacy data (54 %), the need for more intravenous access (54 %), intravenous medication compatibility issues (39 %), and the time during which the patient is attached to an intravenous infusion (31 %).
Conclusion
This survey of children’s hospitals indicates that EIAG is used in over 50 % of hospitals, but there is some lag in adoption of EIBL and CIBL dosing, both of which are used in fewer than 25 % of hospitals. Additional studies may provide much-needed evidence to increase the utilization of these strategies.
Similar content being viewed by others
References
Qin X, Zerr DM, Weissman SJ, Englund JA, et al. Prevalence and mechanisms of broad-spectrum beta-lactam resistance in Enterobacteriaceae: a children’s hospital experience. Antimicrob Agents Chemother. 2008;52:3909–14. doi:10.1128/AAC.00622-08.
El-Mahallawy HA, El-Wakil M, Moneer MM, Shalaby L. Antibiotic resistance is associated with longer bacteremic episodes and worse outcomes in febrile neutropenic children with cancer. Pediatr Blood Cancer. 2011;57:283–8. doi:10.1002/pbc.22926.
Downes KJ, Hahn A, Wiles J, Courter JD, Vinks AA. Dose optimization of antibiotics in children: application of pharmacokinetics/pharmacodynamics in paediatrics. Int J Antimicrob Agents. 2014;43:223–30. doi:10.1016/j.ijantimicag.2013.11.006.
Lodise TP Jr, Lomaestro B, Drusano GL. Piperacillin-tazobactam for Pseudomonas aeruginosa infection: clinical implications of an extended-infusion dosing strategy. Clin Infect Dis. 2007;44(3):357–63.
Lodise TP Jr, Lomaestro B, Drusano GL. Application of antimicrobial pharmacodynamics concepts into clinical practice: focus on β-lactam antibiotics. Pharmacotherapy. 2006;26:1320–32.
Falagas ME, Tansarli GS, Ikawa K, Vardakas KZ. Clinical outcomes with extended or continuous versus short-term intravenous infusion of carbapenems and piperacillin/tazobactam: a systematic review and meta-analysis. Clin Infect Dis. 2013;56:272–82. doi:10.1093/cid/cis857.
Nicolau DP, Freeman CD, Belliveau PP, Nightingale CH, Ross JW, Quintiliani R. Experience with an once-daily aminoglycoside program administered to 2,184 adult patients. Antimicrob Agents Chemother. 1995;39:650–5.
Contopoulos-Ioannidis DG, Giotis ND, Baliatsa DV, Ioannidis JPA. Extended-interval aminoglycoside administration for children: a meta-analysis. Pediatrics. 2004;114:e111. doi:10.1542/peds.114.1.e111.
Knoderer CA, Everett JA, Buss WF. Clinical issues surrounding once-daily aminoglycoside dosing in children. Pharmacotherapy. 2003;23:44–56.
Nichols KR, Knoderer CA, Cox EG, Kays MB. System-wide implementation of the use of an extended-infusion piperacillin/tazobactam dosing strategy: feasibility of utilization from a children’s hospital perspective. Clin Ther. 2012;34:1459–65. doi:10.1016/j.clinthera.2012.05.005.
Knoderer CA, Morris JL, Cox EG. Continuous infusion of nafcillin for sternal osteomyelitis in an infant following cardiac surgery. J Pediatr Pharmacol Ther. 2010;15:49–54.
Walker MC, Lam WM, Manasco KB. Continuous and extended-infusions of β-lactam antibiotics in the pediatric population. Ann Pharmacother. 2012;46:1537–46. doi:10.1345/aph.1R216.
Courter JD, Kuti JL, Girotto JE, Nicolau DP. Optimizing bactericidal exposure for β-lactams using prolonged and continuous infusions in the pediatric population. Pediatr Blood Cancer. 2009;53:379–85. doi:10.1002/pbc.22051.
Cies JJ, Shankar V, Schlichting C, Kuti JL. Population pharmacokinetics of piperacillin/tazobactam in critically ill chidren. Pediatr Infec Dis J. 2014;33:168–73. doi:10.1097/INF.0b013e3182a743c7.
Prescott WA Jr. National survey of extended-interval aminoglycoside dosing in pediatric cystic fibrosis pulmonary exacerbations. J Pediatr Pharmacol Ther. 2011;16:262–9. doi:10.5863/1551-6776-16.4.262.
Chuck SK, Raber SR, Rodvold KA, Areff D. National survey of extended-interval aminoglycoside dosing. Clin Infect Dis. 2000;30:433–9. doi:10.1086/313692.
Tamma PD, Turnbull AE, Milstone AM, Hsu AJ, Carroll KC, Cosgrove SE. Does the piperacillin minimum inhibitory concentration for Pseudomonas aeruginosa influence clinical outcomes of children with pseudomonal bacteremia? Clin Infect Dis. 2012;55:799–806. doi:10.1093/cid/cis545.
Newland JG, Hersh AL. Purpose and design of antimicrobial stewardship programs in pediatrics. Pediatr Infect Dis J. 2010;29:862–3. doi:10.1097/INF.0b013e3181ef2507.
Dellit TH, Owens RC, McGowan JE Jr, et al. Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America guidelines for developing an hospital program to enhance antimicrobial stewardship. Clin Infect Dis. 2007;44:159–77.
Kearns GL, Abdel-Rahman SM, Alander SW, Blowey DL, Leeder JS, Kauffman RE. Developmental pharmacology—drug disposition, action, and therapy in infants and children. N Engl J Med. 2003;349:1157–67.
Craig WA. Pharmacokinetic/pharmacodynamic parameters: rationale for antimicrobial dosing of mice and men. Clin Infect Dis. 1998;26:1–10.
Rubino CM, Bradley JS. Optimizing therapy with antimicrobial agents: use of pharmacokinetic-pharmacodynamic principles in pediatrics. Paediatr Drugs. 2007;9:361–9.
Lopez SA, Mulla H, Durward A, Tibby SM. Extended-interval gentamicin: population pharmacokinetics in pediatric critical illness. Pediatr Crit Care Med. 2010;11:267–74. doi:10.1097/PCC.0b013e3181b80693.
DeRyke AC, Lee SY, Kuti JL, Nicolau DP. Optimising dosing strategies of antimicrobials utilising pharmacodynamic principles: impact on the development of resistance. Drugs. 2006;66:1–14.
Schumock GT, Raber SR, Crawford SY, Naderer OJ, Rodvold KA. National survey of once-daily dosing of aminoglycoside antibiotics. Pharmacotherapy. 1995;15(2):201–9.
Kraus DM, Pai MP, Rodvold KA. Efficacy and tolerability of extended-interval aminoglycoside administration in pediatric patients. Paediatr Drugs. 2002;4:468–84.
Nestaas E, Bangstad HJ, Sandvik L, Wathne KO. Aminoglycoside extended interval dosing in neonates is safe and effective: a meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2005;90:F294–300. doi:10.1136/adc.2004.056317.
Vogelman BS, Craig WA. Postantibiotic effects. J Antimicrob Chemother. 1985;15(suppl A):37–46.
Van der Auwera P, Klastersky J. Serum bactericidal activity and postantibiotic effect in serum of patients with urinary tract infection receiving high-dose amikacin. Antimicrob Agents Chemother. 1987;31(7):1061–1068.
Conflicts of interest
C.A. Knoderer, K.R. Nichols, and E.G. Fox have no potential conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Knoderer, C.A., Nichols, K.R. & Cox, E.G. Optimized Antimicrobial Dosing Strategies: A Survey of Pediatric Hospitals. Pediatr Drugs 16, 523–529 (2014). https://doi.org/10.1007/s40272-014-0093-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-014-0093-1