Abstract
A substantial volume of literature exists linking proton pump inhibitor (PPI) use with a multitude of serious adverse events. There is uncertainty, however, over whether these associations are clinically important. Excessive concern about PPI-related adverse events may leave patients at risk of harm by leaving acid-related upper gastrointestinal disease untreated. Conversely, the risk of treatments may outweigh the benefits if any of the purported adverse events are directly caused by PPI use; this is of particular concern where indications for PPI use are not present. In this paper, we review the studies which have reported associations between adverse events and PPI use, discuss the proposed mechanisms of action, grade the confidence in whether these associations are truly causal, and provide advice regarding balancing the benefits of PPI use against their possible harms.
Similar content being viewed by others
Patients and care providers are becoming increasingly concerned about adverse events which have been associated with PPI use. |
To date, none of these associations have been shown to be causal, though for some, particularly the infectious complications, there is some degree of biologic plausibility. |
We do not recommend PPI discontinuation where there is a definite indication for continuing use. |
Physicians should try to prevent unnecessary long-term PPI use by being judicious in their initial prescribing and by regularly reassessing the need for ongoing PPI therapy when unclear indications exist. |
1 Introduction
Proton pump inhibitors (PPIs) are among the most widely prescribed medications in the world, with 14.9 million patients receiving 157 million prescriptions in 2012 in the United States alone [1]. This ignores the substantial contribution from over-the-counter sales of PPIs which are available in many countries. Sales of Nexium® (esomeprazole), the most commercially successful PPI, peaked at over $US6 billion in 2013 before it lost patent protection [2].
The success of PPIs is due to their profound ability to suppress gastric acid production which directly addresses the pathophysiologic mechanism of a number of highly prevalent upper gastrointestinal syndromes. They are approved by the Food and Drug Administration for use in treatment of gastroesophageal reflux disease (GERD), healing and maintenance of healing in erosive esophagitis, prevention of non-steroidal anti-inflammatory drug (NSAID)-induced gastric ulcer, treatment of gastric or duodenal ulcer, as part of Helicobacter pylori eradication, and in hypersecretory conditions like Zollinger–Ellison syndrome.
Nuisance side effects of PPI use are generally infrequent and mild, consisting of headache, nausea, abdominal pain, diarrhea, and flatulence. Despite their demonstrated safety profile in randomized controlled trials, there has been substantial concern expressed over the potential for long-term adverse effects. This is particularly relevant in light of their OTC availability, their widespread empiric use for nonspecific upper gastrointestinal symptoms and their administration at unapproved higher-than-standard doses.
The purpose of this review is to examine the evidence behind the numerous reported adverse effects of PPI therapy, discuss their purported pathophysiology, and to reflect on their relevance of these concerns to clinical practice.
2 Potential Mechanisms of PPI-Related Adverse Events
Proposed mechanisms for many of the adverse effects associated with PPI use summarized in this review can be subdivided into: (1) direct effects of inhibition of gastric acid secretion, (2) PPI effects on proton pump and proton pump analogues found in non-gastric tissue, (3) downstream neurohormonal responses to impaired acid secretion, and (4) interactions between PPIs and the absorption or metabolism of other medications leading to exaggerated or attenuated effects.
The most immediate and predictable effect of PPI use is the development of hypochlorhydria. Gastric acid is important in the digestion and absorption of food and drugs as well as in maintaining a relatively sterile gastric environment. Many pathogens cannot survive in a highly acidic environment, and thus reduction in gastric acid production may increase the risk of infections spread through fecal-oral contact. In addition, hypochlorhydria may theoretically interfere with the absorption of certain cations which are more soluble in acidic environments (e.g. calcium carbonate) or where acid is necessary to oxidize cations into a more absorbable form (as in the acid-induced conversion of ferrous ions to ferric ions).
A number of non-gastric H+K+-ATPases exhibiting unique functional properties have been isolated from colon, bone, kidney, prostate, and pancreas [3, 4]. These non-gastric proton pumps are thought to be important in creating hyperlocal acidic environments which may accelerate certain metabolic pathways. PPIs have demonstrated the ability to inhibit non-gastric proton pumps in vitro and in animal models, offering a potential mechanism for adverse events outside of the gastrointestinal tract [5].
A low intragastric pH is an important inhibitor of gastrin production. Hypochlorhydria, whether the result of PPI therapy, gastric resection, or other causes, is well documented to result in hypergastrinemia. Gastrin levels increase after as little as 2–4 weeks of PPI therapy and remain elevated with prolonged use [6]. This has potential effects outside of the primary role as a stimulant of acid secretion, as gastrin also acts as a growth hormone capable of stimulating parietal cell proliferation, cell migration and angiogenesis, and of inhibiting apoptosis [7].
3 Observational Studies and Their Limitations
The majority of studies reporting on association between PPI use and adverse outcomes are observational, with many utilizing epidemiologic databases originally intended for administrative purposes. This methodology offers the ability to analyze large cohorts with relative ease and to identify associations that may have gone unnoticed in randomized controlled trials (RCTs) as RCTs generally would have smaller numbers of subjects, shorter duration of follow-up, and stringent patient selection criteria inherent in their design. There exists, however, the potential to draw false associations when examining observational data due to confounding as well as to inappropriate study design. This is of particular concern with regard to the validity of PPI-adverse event associations, as PPI use is much more common among people with a number of medical comorbidities, and these comorbidities may actually be the underlying cause of the adverse event. This is generally referred to as confounding.
Observational studies generally attempt to adjust for the effects of confounding through statistical modeling; however, modeling cannot control for the effects of potential confounding variables which are absent from the dataset being analyzed. Furthermore, techniques which control merely for the presence of a confounding exposure that do not take into account the severity of the confounding disorder will bias the results of a study against the group with the higher proportion of people with severe disease.
As a result, observational studies are capable of establishing association between exposure and event but are unable to establish causation. While causation can be more firmly implied through an experimental design such as an RCT, performing an RCT to confirm whether PPI use causes a particular adverse event would be unfeasible due to financial, logistical, and ethical constraints. As such, we frequently must make inferences regarding the causality of an association which is detected in an observational trial. Frameworks such as the Bradford-Hill criteria are frequently used in determining that an association between an exposure and an outcome detected in an observational trial is likely to be causal, which helps care-providers and policymakers determine whether to apply these findings to clinical practice. In the studies evaluating associations between PPIs and adverse events, the criteria that are most applicable are strength of association, consistency, temporality, and plausibility.
Strength of association between exposure and event is often considered the most important of the Bradford Hill criteria due to the potential for residual confounding in observational studies. The stronger the association between an exposure and an outcome the more difficult it is to argue that the association is merely due to residual confounding or other methodological biases. Odds ratios (OR) of 0.5–2.0 are often referred to as being in the “zone of potential bias” and should be interpreted with caution.
Along similar lines, consistency in results between studies is also important. Differing results may be due to differences in study design and their effect on confounders, while results that are consistent, particularly between different study types, are more supportive of a causal association.
Temporality means that the exposure must occur before the event in question. While simple in concept, establishing temporal association in practice is complicated by the potential for protopathic bias, which occurs when a drug is prescribed for an early manifestation of a disease that has not yet been diagnosed. When the disease is ultimately diagnosed, it will appear that the drug was started before the disease occurred, when in fact the drug was prescribed in the preclinical prodrome of the disease process. This often leads to effect sizes that are larger in the initial period after starting a drug but diminish with time. This hallmark of protopathic bias is seen with a number of the adverse effects associated with PPI discussed below.
Ultimately, it is important to establish a biologically plausible mechanism through which PPI use can lead to the occurrence of a specific adverse outcome. In certain circumstances, a known mechanism of action may not be evident when an epidemiologic association is first detected, but then is discovered through further basic scientific research. However, the failure to either have an a priori mechanism or the failure to discover a mechanism within a reasonable amount of time suggests that an association seen between PPI use and a specific adverse outcome is noncausal.
For the purpose of this review, literature search was conducted using PubMed. Consideration was given to studies where an adverse association was initially noted, particularly high-quality studies addressing a particular association, and to the most recently published meta-analyses. We have included a summary of all reported side effects and complications associated with PPI use in Table 1.
4 Gastrointestinal Complications of PPI Use
4.1 Alteration of the Gastric Microbiome
In persons with normal gastric acid physiology, the gastric microenvironment is inhospitable to many bacterial and fungal organisms, with the notable exception of Helicobacter pylori (HP). The majority of untreated subjects have no culturable organisms in gastric juices, while those who do generally do not exceed 106 colony-forming units (CFU) per mL [8].
PPIs are capable of altering the gastric microbiome through two mechanisms. First, PPIs promote alkalization of the gastric microenvironment, which may promote the growth of non-HP organisms. CFU counts of culturable bacteria in stomach contents of healthy volunteers given a two-week course of omeprazole increased 200-fold from baseline [9]. Similar effect has been observed with culture of gastric mucosa [8]. Prolonged PPI treatment for > 12 months further increases CFU counts of non-HP organisms by a factor of 106 [10]. PPIs can also have direct effects on bacterial and fungal proton pumps [11]. The clinical relevance of this second mechanism is largely unexplored, although super-physiologic doses of pantoprazole have been shown to inhibit growth of Lactobacillus species in vitro [12].
Despite their increased numbers, the diversity of the gastric microbiome does not appear to be affected. 16S RNA sequencing of corpus biopsy specimens from PPI-treated subjects and untreated controls demonstrated similar bacterial profiles [13]. Whether or not the increase in gastric bacterial burden is of clinical relevance remains to be determined.
4.2 Small Intestinal Bacterial Overgrowth
PPI-induced hypochlorhydria is thought to predispose to development of small intestinal bacterial overgrowth (SIBO) by allowing the increased survival of ingested microorganisms [14]. Persons with SIBO may develop bloating, diarrhea, and abdominal pain, and may suffer from carbohydrate malabsorption as well as micronutrient deficiency. It is unclear whether PPIs promote the development of SIBO in the absence of other predisposing factors such as intestinal dysmotility [15], structural abnormalities [16], or immune deficiency [17].
The majority of studies evaluating the prevalence of SIBO have used some form of metabolic breath test to establish the diagnosis. The range of breath tests available introduces significant heterogeneity as well as inaccuracy, as the gold standard for diagnosis remains duodenal aspirate and culture. A 2012 meta-analysis of 11 studies found no association between PPI use and SIBO but did find a significant association when the analysis was restricted to the five studies using duodenal aspirates [18]. A subsequent meta-analysis in 2018 including 19 studies with 2175 cases of SIBO in 7055 subjects demonstrated a modest association between PPI use and SIBO in both glucose-hydrogen breath test (OR 1.84, 1.03–3.30) and duodenal aspirate and culture studies (OR 2.22, 1.33–3.68), but not in lactulose hydrogen breath test or D-xylose breath test studies [19].
Further research is needed to investigate whether PPI discontinuation or a course of antibiotic treatment results in resolution of SIBO. The substantial differences in study design as well as the limited role that duodenal aspirate and culture play in routine clinical practice limit the relevance of the currently available data.
4.3 Enteric Infection
Gastric acid serves an important immune function by eliminating ingested pathogens from the digestive tract. Impairment of acid production by PPIs and histamine H2-receptor antagonists (H2RAs) has been implicated in the development of Clostridium difficile infection (CDI) and other bacterial infections of the gut.
4.3.1 C. difficile Infection
CDI is the most common cause of hospital-acquired diarrhea [20]. Antibiotic use is strongly associated with development of CDI, but a substantial proportion of CDI cases are community acquired and occur in subjects without typical risk factors. C. difficile is a spore-forming organism which makes it relatively resistant to degradation by gastric acid [21], yet a substantial body of evidence exists documenting an association between PPI use and development of infection. In light of the acid-resistant nature of C. difficile it seems unlikely that predisposition to CDI afforded by PPI use would be the result of pH effect. More plausible is that the association results from alteration to the colonic microbiome [22]. Antibiotic use is known to cause CDI by depleting commensal bacteria that normally prevent C. difficile proliferation and reducing microbial diversity [23]. Freedberg et al. demonstrated that healthy volunteers exposed to PPI therapy for four weeks develop within-individual increases in Enterococcaceae and Streptococcaceae, taxa that have been associated with exposure to antibiotics and increased risk for CDI, offering support for this mechanism [24].
Seven meta-analyses have been published on PPI use and CDI over the past 8 years and all have reported a positive association. The most recent of these included over 350,000 cases of CDI and found that cases were almost twice as likely to have been exposed to PPI therapy as controls (OR 1.99, 1.73–2.30, p < 0.001). Limiting analysis to the seven highest-quality studies yielded similar results (OR 1.88, 1.55–2.28). Number needed to harm was estimated at 63 (48–78) using the reported incidence of CDI of 1.67% within 14 days of hospital admission [25]. An earlier meta-analysis by Kwok et al. reported similar risk of CDI with concurrent use of PPIs and antibiotics (OR 1.96, 1.03–3.70) as with PPIs alone (1.74, 1.47–2.05) [26].
PPIs have also been associated with a higher risk of recurrent CDI. Studies have varied somewhat in their definition of the timeframe that defines recurrence, but meta-analysis has found an increased risk of recurrent CDI within 60 days (OR 1.54, 1.04–2.28) and 90 days (OR 1.53, 1.07–2.19) of initial successful treatment among PPI users [27].
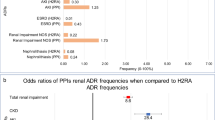
Despite the number of publications documenting association between PPI use and CDI the quality of evidence is low, consisting of nonrandomized retrospective cohort studies. Significant heterogeneity exists between studies although the direction of effect does consistently show increased risk. Of note, studies that have compared the magnitude of risk conferred by H2RA use in comparison with PPI use have shown greater risk with PPIs (OR 2.10, 1.66–2.66) than with H2RAs (OR 1.50, 1.23–1.83) [26].
4.3.2 Other Enteric Infections
Unlike C. difficile, which is capable of forming acid-resistant spores, gastric acid acts as a physiologic barrier against the oro-colonic survival of intestinal pathogens commonly spread through fecal-oral transmission, such as Salmonella, Campylobacter, and pathogenic Escherichia coli. As a consequence, persons with PPI-induced hypochlorhydria may be at increased risk of developing enteric infections with these organisms.
A 2007 meta-analysis found a significant association between PPI use and non-C. difficle bacterial enteric infections (OR 3.33, 1.84–6.02). This was numerically greater than the OR seen with H2RA use and non-C. difficile enteric infections and the OR for PPI use and C. difficile infection, although neither reached the level of statistical significance [28].
A subsequently published nested case-control study within The Rotterdam Cohort prospectively examined cases of Campylobacter, Salmonella, Yersinia, and Shigella among non-hospitalized subjects. Participants with bacterial gastroenteritis were twofold more likely to be current users of PPIs than were controls with negative stool cultures (AOR 1.94, 1.15–3.25). Importantly, this was markedly lower than the estimated sixfold increase in risk of bacterial gastroenteritis generated by comparison with the overall cohort (i.e. no stool sample taken), indicating the importance of an adequate control group in avoiding case ascertainment bias [29].
The biological plausibility of enteric infectious complications of PPI use hinges on the direct effect of increased gastric pH. While PPIs are potent acid suppressors, they do not achieve the achlorhydria seen with pernicious anemia. The magnitude of effect seen in this population would therefore represent the upper limit of effect size potentially attributable to acid suppression. There is a paucity of data on infectious complications in this population, therefore it is uncertain whether the effect sizes seen with PPI use are within this range.
Risk of precipitating enteric infection should be considered when prescribing PPIs, particularly for patients with other risk factors for CDI. Similarly, development of CDI should prompt reevaluation of the need for ongoing PPI use. If an indication continues to exist, the benefit of continued PPI use is likely warranted, particularly as alternative treatments are not without risk.
4.4 Spontaneous Bacterial Peritonitis
Alteration to the normal gut flora may be of particular relevance in the cirrhotic population where slowed intestinal transit, immune dysfunction, and hypoproteinemia all predispose to translocation of enteric bacteria and development of spontaneous bacterial peritonitis (SBP).
Conflicting data exist on whether PPI use is clinically relevant in this context. A 2014 meta-analysis of 12 published studies and five abstracts found a significant association between PPI use and SBP in cirrhotics with ascites (OR 2.17, 1.46–3.23; p < 0.05) [30]. This was supported by a post-hoc analysis of three multinational RCTs originally conducted to determine whether satavaptan reduced ascites formation in cirrhotics that reported an adjusted hazard ratio (HR) of 1.72 (1.10–2.69) for development of SBP with PPI use. Furthermore, there was an increased risk of developing a first episode of hepatic encephalopathy with PPI use (adjusted HR 1.36, 1.01–1.84) and a higher incidence of severe hepatic encephalopathy (55% vs 43% of episodes West Haven grade 2 or higher, p = 0.02) among PPI users [31].
In contrast, two large cohort studies of decompensated cirrhotics admitted to hospital in Argentina and Germany found no difference in the prevalence of prior PPI use between subjects diagnosed with SBP and those with other diagnoses [32, 33]. Despite this, PPI users in the German study who did develop SBP had a more severe clinical course than non-users, with higher rates of acute kidney injury (71% vs 43%; p = 0.002), hepatic encephalopathy (15% vs 0%; p = 0.04), and 28-day mortality (24% vs 0%; p = 0.008) [33].
Plausible mechanism and reasonable quality evidence exist to support a causal relationship between PPI use and development of SBP. PPIs should be used judiciously among cirrhotics with ascites as the consequences of SBP are often severe. Indications for PPI use should be clearly defined and frequently reevaluated. Of particular interest is the practice of starting PPI therapy following esophageal variceal ligation to prevent bleeding from mucosal ulcerations. This has never been demonstrated to reduce bleeding from ligation-related ulcers. If PPIs are started for this purpose the intention for them to be used for a short period of time should be clearly communicated.
4.5 Gastric Cancer
HP infection and resulting chronic gastric inflammation is a widely accepted risk factor for gastric cancer [34]. The effects of PPI use on HP are complex, with HP augmenting the acid-suppressive effect of PPIs and PPIs exhibiting bacteriostatic effects through inhibition of bacterial P-type ATPase and bacterial urease activity [35, 36]. Only a small proportion of HP subjects chronically treated with PPI monotherapy clear the infection. Rather, a change in disease distribution occurs with a shift in HP colonization and inflammatory cell infiltration from antrum to corpus after as little as four weeks of acid-suppression [37,38,39]. A number of studies have documented development of diffuse atrophic gastritis with long-term PPI use [40,41,42,43]. Occurrence is related to the effect of acid suppression on coexisting HP infection, as illustrated by the observation that 31% of HP-positive GERD subjects developed atrophic gastritis after five years of omeprazole treatment versus 0% of HP-positive subjects with surgically treated GERD (p < 0.001). In comparison, only 4% of HP-negative omeprazole subjects developed atrophic gastritis [44]. Whether the interaction between HP and PPIs further impacts the carcinogenic process is unknown, as thus far the evidence of a clinically meaningful effect is unconvincing. Further high-quality studies are needed to address this issue.
The largest and most robust study to date to examine PPI use and gastric cancer described a Swedish cohort of 797,067 subjects on chronic PPI therapy, of whom 2219 developed gastric cancer during median follow-up of 4.9 years. Standardized incidence ratio (SIR) of all gastric cancers and of gastric adenocarcinoma specifically, were both increased threefold. Risk remained elevated after excluding early gastric cancers (SIR 1.61, 1.51–1.71) and was most pronounced among individuals receiving PPIs for less than one year (SIR 12.82, 12.19–13.47) and among subjects aged < 40 years (SIR 22.76, 15.94–31.52). SIR of gastric cancer was increased across all 10 studied indications for PPI therapy including ulcer prophylaxis in NSAID users. Similar effects were not observed among H2RA users [45]. Two meta-analyses on gastric cancer in PPI users published before the Swedish study also reported a positive association albeit of lesser magnitude (OR 1.39, 1.19–1.64 and RR 1.43, 1.23–1.66) [46, 47]. Both meta-analyses consisted of the same three observational studies with numerous limitations.
Confounding is a major issue, particularly in relation to indication for PPI use. The Swedish study attempted to limit confounding by indication by analyzing subgroups by indication, but reasons for PPI therapy were unclear in 25% of subjects. Other potential confounders include unmeasured risk factors for gastric cancer including obesity, smoking, alcohol consumption, and family history. Furthermore, the degree of risk conferred by short duration of PPI therapy and the decrease in risk with duration of PPI use both strongly suggest an element of protopathic bias. Substantial overlap exists between early nonspecific symptoms of gastric cancer and benign conditions frequently treated with PPIs which could predispose to this phenomenon.
4.6 Fundic Gland Polyps
Fundic gland polyps (FGPs) are small mucosal lesions composed of cystically dilated fundic glands lined by parietal or chief cells arranged in a disordered fashion. Located in the fundus or body of the stomach, they can occur sporadically or as part of inherited polyposis syndromes. PPI use is associated with a pooled OR for developing FGPs of 2.46 (1.42–4.27; p = 0.001) and increases with duration of exposure, roughly doubling after one year [48]. Estimates of effect size are limited by heterogeneity in demographics, duration of PPI exposure, indication for endoscopy, and prevalence of HP infection between studies.
The pathogenesis of FGPs is unclear, which makes it difficult to speculate by what mechanism PPIs might affect their development. Among subjects without familial adenomatous polyposis, FGPs are reported in 1.9% of gastroscopies, and dysplasia is found in 1% [49, 50]. In this setting the clinical relevance of a twofold increase in prevalence with PPI use is questionable.
4.7 Microscopic Colitis
Microscopic colitis (MC) is an uncommon but increasingly diagnosed cause of chronic non-bloody diarrhea that predominantly affects older adults. A number of case reports and small-scale case control studies first identified a link between PPI use and development of MC [51,52,53,54]. This association was subsequently confirmed by a large Danish healthcare registry study that matched subjects with lymphocytic colitis (LC) or collagenous colitis (CC) with population-based controls between 2005 and 2011. A strong association was reported between redemption of prescriptions for PPIs and both CC (AOR 7.04, 6.55–7.56) and LC (AOR 3.37, 3.08–3.69) [55]. Data from the British Clinical Practice Research Datalink found that concomitant use of PPIs and NSAIDs was associated with a greater risk of MC than use of either agent alone (AOR 3.61, 2.46–5.29 for PPIs in combination with NSAIDs vs AOR 2.41, 1.98–2.92 for PPIs alone vs AOR 1.29, 0.90–1.86 for NSAIDs alone) [56]. There are no data demonstrating whether PPI discontinuation leads to resolution of MC.
The pathogenesis of MC is unclear but is believed to relate to altered epithelial barrier function leading to translocation of gut bacteria and antigens triggering an inflammatory response [57]. PPI exposure has been reported to induce intestinal dysbiosis in humans and could thus contribute to impaired barrier function [58]. Alternatively, a variant of the gastric H+K+-ATPase has been demonstrated in colonic tissue [59]. Binding of PPIs to this pump could theoretically also lead to impaired barrier function as a result of electrolyte disturbance, although the magnitude of the effect of PPIs on colonic proton pumps seems unlikely to be of clinical significance given their rapid absorption from the upper GI tract and targeted activation in the acidic gastric canaliculus. Impaired prostaglandin synthesis resulting from NSAID use can also increase gut permeability, potentially explaining the magnified risk of MC seen with co-administration of PPIs and NSAIDs.
Microscopic colitis should be considered and appropriately investigated among PPI-treated subjects presenting with watery diarrhea, particularly if being used together with NSAIDs. The natural history of MC is not well defined, and while there is no evidence demonstrating resolution of MC on PPI discontinuation, a trial off therapy is reasonable assuming a strong indication for ongoing acid suppression does not exist. Further research into pathogenic mechanisms is required before concluding a causal relationship.
5 Extraintestinal Complications of PPI Use
5.1 Pneumonia
The association between PPI use and pneumonia has been among the most widely studied purported complications of PPI use. A 2015 meta-analysis identified 26 studies including over six million participants, with 15 of 26 studies reporting a positive association between PPI use and development of community-acquired pneumonia. The pooled adjusted risk ratio of developing pneumonia among PPI users was 1.49 (95% CI 1.16–1.92) with significant heterogeneity between studies. Limiting the analysis to six studies using a stricter definition of CAP requiring radiographic confirmation reduced the strength of association (RR 1.22, 95% CI 0.99–1.52). The risk of pneumonia was not modified by either PPI dose or advanced age (> 65 years). The largest risk of pneumonia was seen in the first month of use and diminished thereafter, which raises the suspicion that protopathic bias may play a role for some degree of the association [60].
The lungs are constantly exposed to microbiota from inhaled air and from the upper respiratory tract [61]. Gastroesophageal reflux disease (GERD) may exacerbate this risk by facilitating the retrograde movement of gastric contents [62]. PPI use is thought to compound this risk by increasing the size of the inoculum introduced into the lungs as a result of hypochlorhydria-related gastric bacterial overgrowth, which in turn overwhelms host defenses.
As PPIs are most commonly used as treatment for GERD, and GERD itself may be a risk factor for pneumonia, it raises the possibility that GERD is a major confounder for the reported PPI-pneumonia association. Filion et al. tried to account for this confounder by evaluating the PPI-pneumonia association in a cohort of NSAID users presumably taking PPIs for peptic ulcer prophylaxis as opposed to GERD, and found no association between PPI use and pneumonia [63]. Similarly, no association between receipt of PPIs and development of pneumonia was demonstrated in a meta-analysis of 19 trials of PPI use among 2117 patients in the intensive care unit for stress ulcer prophylaxis (RR 1.12, 0.86–1.46) [64].
Given the concerns about protopathic bias and the lack of a clear mechanism that is independent of GERD, there is not enough evidence at this time to recommend avoidance of PPI use in persons with pulmonary disease, or those with a history of pneumonia.
5.2 Vitamin and Mineral Deficiency
5.2.1 Vitamin B12
By far the largest study addressing the question was conducted by Lam et al. within the Kaiser Permanente Northern California integrated healthcare system [65]. Subjects with a first diagnosis of vitamin B12 deficiency from 1997 to 2011 were compared with up to 10 matched controls. Cumulative prescription of PPIs for two or more years was associated with an OR of 1.65 (1.58–1.73) for development of vitamin B12 deficiency. Association was strongest amongst users of high doses for prolonged periods and diminished after PPI discontinuation. PPI use is also associated with lower mean serum Vitamin B12 levels [66].
Plausible mechanisms exist by which gastric acid suppression could lead to vitamin B12 malabsorption and ultimately deficiency. Gastric acid is produced by parietal cells, the same cells responsible for production of intrinsic factor required for absorption of vitamin B12. Furthermore, gastric acid is required to cleave vitamin B12 from ingested dietary protein to allow for its absorption. Therefore, there is reasonable evidence for there being a causal relationship between PPI use and low vitamin B12 levels. However, given that vitamin B12 deficiency is easy to reverse, this may not necessarily require the avoidance of PPIs in the presence of vitamin B12 deficiency.
5.2.2 Iron
The strongest evidence for an association between PPI use and iron deficiency again comes from a nested case control study from the Kaiser Permanente Northern California health system that compared rates of PPI exposure between 77,046 subjects with iron deficiency anemia and 389,314 matched controls. Cases were significantly more likely to have been dispensed a ≥ 2-year supply of PPIs (3.0% vs 0.9%) or H2RAs (1.4% vs 0.6%) prior to their diagnosis of iron-deficiency anemia than controls at the date of matching. Higher doses of PPIs (≥ 1.5 PPI pills per day: OR 2.61, 2.27–3.01; < 0.75 PPI pills per day: OR 2.01, 1.75–2.30; p = 0.004) and greater duration of use (OR 4.27, 2.53–7.21 for users of > 1.5 pills per day for ≥ 10 years) were associated with increased risk, as was younger age. Strength of association progressively decreased after PPI discontinuation [67].
Gastric acid facilitates non-heme iron absorption by releasing iron from food and oxidizing it to the more absorbable ferric form [68]. By this mechanism, suppression of acid production through PPI or H2RA use could plausibly lead to iron malabsorption, suboptimal response to iron supplementation, and iron deficiency. This was supported by animal models [69], but early observational studies in humans were small and produced discordant results [70,71,72,73]. This association may also be confounded by the fact that many of the conditions for which PPIs are used can themselves lead to gastrointestinal blood loss and iron deficiency.
Similar to the situation with vitamin B12, as iron deficiency is itself easily remediable, PPI use should likely be continued in its presence so long as indications for PPI use are ongoing.
5.2.3 Magnesium
In contrast to iron and vitamin B12, where deficiency is hypothesized to result from the direct effect of acid suppression, the mechanism of PPI-induced hypomagnesemia is believed to derive from drug effects on transient receptor potential melastin (TRPM) 6 and 7 cation channels responsible for magnesium transport in the distal small intestine and large bowel [74].
Data linking PPI use with hypomagnesemia is less robust than that seen with other metabolic abnormalities. Elderly subjects in Ontario hospitalized with hypomagnesemia were more likely to be current PPI users than were controls matched on the basis of age, sex, kidney disease, and diuretic use (AOR 1.43, 1.06–1.93). Strength of association was greatest amongst PPI subject receiving concomitant diuretics (AOR 1.73, 1.11–2.70) [75]. In contrast, a study of health maintenance organization participants identified adult subjects receiving continuous PPI prescriptions between 2008 and 2013 who had serum magnesium measured on one or more occasions. 57 of 414 subjects (13.8%) receiving PPIs for more than six months had low serum magnesium on at least one occasion, but only eight subjects did not have another recognizable cause of hypomagnesemia [76]. There are a number of case reports linking PPI use to severe hypomagnesemia which resolves with drug withdrawal and recurs on rechallenge. It is presumed, although not proven, that this occurs in individuals with variant TRPM alleles making them particularly susceptible to the effects of PPIs [77].
A small subset of patients prone to development of severe hypomagnesemia with PPI use exists. Routine monitoring of magnesium levels is not indicated given the rarity with which this occurs, however quick recognition and discontinuation of PPIs in the setting of severe hypomagnesemia is warranted.
5.2.4 Calcium
The importance of gastric acid in facilitating intestinal calcium absorption is unclear. Achlorhydric individuals demonstrate impaired absorption of calcium carbonate supplements when fasting, but no difference in absorption when in the fed state [78]. Short-term studies of healthy young adults and postmenopausal women have not demonstrated significant PPI effect on fractional calcium absorption or urinary calcium excretion [79, 80]. Similarly, the SOPRAN and LOTUS studies, which compared the safety of long-term PPI use with open and laparoscopic anti-reflux surgeries, did not demonstrate any difference between calcium or vitamin D levels between PPI-treated and surgical groups [81].
5.3 Osteoporosis and Fractures
Association between long-term PPI use and risk of fracture was first reported in 2006 in independent studies by Vestergaard et al. [82] and Yang et al. [83]. More than 25 additional studies and several meta-analyses examining both fracture rates and effects on bone mineral density have followed and have reported widely divergent results.
The original paper by Yang and colleagues examined hip fractures among PPI users and non-users aged 50 and up identified from the UK General Practice Research Database. AOR for hip fracture after more than one year of PPI use was 1.44 (1.30–1.59), which increased to 2.65 (1.80–3.90) among long-term, high-dose users [83]. A similarly designed Canadian study investigating hip, wrist, and vertebral fractures did not identify an excess risk for fracture among users of PPIs for six or fewer years, but found that risk was increased among users for seven or more years (AOR 1.92 (1.16–3.18)] [84].
Meta-analysis of 18 studies (nine case control, nine prospective) showed a modestly increased risk of hip fracture (RR 1.26, 1.16–1.36) with significant heterogeneity between studies. Risk of vertebral fracture (RR 1.58, 1.38–1.82) and any site fracture (RR 1.33, 1.15–1.54) were also modestly increased [85].
The presence of a causal relationship between PPI use and development of fracture is not established, as there is no confirmed mechanism by which PPIs would weaken bone or otherwise increase the risk of fracture. Despite theoretical concerns that achlorhydria may impair intestinal calcium absorption, numerous studies have now shown that PPIs have no discernible effect on fractional calcium absorption or urinary calcium excretion. It has also been proposed that PPI use inhibits the vacuolar acid pump found in osteoclasts, thereby preventing osteoclasts from producing acid and inhibiting bone resorption, ultimately impairing bones’ ability to repair microfractures which may act as the leading edge of clinical fractures. However, this theory remains unproven.
Furthermore, studies evaluating BMD and 3D bone structure have demonstrated no significant difference in bone density or in bone structure among PPI users and non-users. Canadian subjects started on PPI therapy had lower baseline bone mineral density (BMD) than matched controls not started on PPIs but did not demonstrate accelerated loss of BMD over 10 years [86]. Similarly, no difference in aBMD, volumetric BMD, bone strength, or markers of bone metabolism was found between long-term (≥ 5 years) PPI users and subjects unexposed to PPIs in the preceding 5 years [87].
While there is a moderately consistent association between PPI use and the incidence of fracture, the lack of a plausible mechanism of action casts doubt as to its causality. Currently, we would not recommend screening persons who use PPIs for osteoporosis, nor would we recommend their discontinuation is persons with known osteoporosis or a history of fracture.
5.4 Myopathy
A 2005 search of the World Health Organization adverse drug reaction database identified 292 case reports where PPIs were associated with muscle weakness, myopathy, myositis, polymyositis, rhabdomyolysis, or creatine phosphokinase elevation. In 37% of cases the PPI was the only drug reported, while in 57% of cases where use of multiple drugs was reported, the PPI was the only suspected culprit medication. Thirty-five cases of rhabdomyolysis were documented, with 12 occurring in the setting of concomitant statin use. Sixteen cases reported time to onset of rhabdomyolysis after PPI initiation with nine cases occurring within the first week of PPI use [88]. A 2017 review article summarized 12 published case reports of myopathy occurring with PPI use [89]. In the majority of cases in both publications, symptoms resolved completely after PPI cessation.
There is no clear mechanism through which PPIs might induce myopathy, though as with all drugs, rare idiosyncratic side effects can never be ruled out entirely. The small number of cases reported over almost 30 years of use among millions of prescription and OTC users serves more to highlight the safety of the medications than to raise particular clinical concern.
5.5 Dementia
One of the more recent reported associations with PPI use has been development of incident dementia. This was first reported in a large German prospective observational longitudinal study using data from the largest German statutory health insurer [90]. This retrospective analysis included subjects 75 years or older and compared rates of incident dementia between regular PPI users and nonusers. Regular PPI use was associated with a significantly increased risk of incident dementia [HR 1.44 (95% CI 1.36–1.52), p < 0.001], an effect size similar to that seen with history of stroke. A subsequent study from Taiwan reported a similarly increased risk of dementia [91], while three studies have reported no association [92,93,94], and two have reported a protective effect to PPI use similar to that seen with statins or antihypertensives [95, 96].
The largest and most robust study to date compared PPI use among 70,718 Finnish subjects newly diagnosed with Alzheimer’s disease from 2005 to 2011 with PPI use among 282,858 age-, sex-, and region of residence-matched controls and reported no significant relationship between exposure and outcome, including among long-term and high-dose users (AOR for ≥ 3 years of PPI use = 0.99, 0.94–1.04) [93]. Importantly, data were analyzed using a three-year lag window between PPI exposure and development of Alzheimer’s dementia, a feature that studies reporting associations between PPI use and dementia did not [90, 91]. This is particularly relevant as dementia is often an insidious process that may go unnoticed for years, introducing bias into any retrospective study. There are also no data to demonstrate the association between PPI use and the rate of cognitive decline in persons with dementia.
There is no well-established mechanism by which PPI use might lead to dementia. Hypotheses include induction of vitamin B12 deficiency [65] and enhancement of amyloid-β plaque deposition in the brain due to altered activity of pH-dependent proteases involved in amyloid-β production [97].
Given the relatively modest association, the potential for significant other biases, and the lack of a plausible mechanism, the balance of evidence at this point would seem to suggest the lack of a causal association between PPI use and dementia. Thus, we would not recommend discontinuing PPIs in persons with dementia, and we would suggest using PPIs for appropriate indications in persons with established dementia.
5.6 Kidney Disease
5.6.1 Acute Interstitial Nephritis
A nested case-control study from New Zealand identified 46 cases of biopsy-confirmed acute interstitial nephritis (AIN) among current PPI users over a four-year period and reported an unadjusted OR of 5.16 (2.21–12.05) for current PPI use versus past use. No evidence of dose or duration effect was seen, and risk among recent users of PPIs was not elevated. Crude absolute risk of AIN was estimated at 11.98 cases per 100,000 person years among PPI users compared with 1.68 cases for former users, corresponding to a number needed to harm of approximately 10,000 [98]. A retrospective case review at two Australian teaching hospitals identified 18 cases of biopsy-proven, PPI-associated AIN. Mean duration of PPI use prior to presentation was 11 weeks and typical features of AIN were often not present, with only 10% of affected patients having the classic hypersensitivity triad of fever, rash, and eosinophilia [99].
The pathogenesis of acute interstitial nephritis (AIN) involves an idiosyncratic allergic reaction prompted by exposure to an offending agent. Historically, infection has been the most common cause, but modern series identify drugs as by far the most frequent culprits. A type IV (delayed-type) hypersensitivity response is often implicated, although how and why this response is elicited by a particular drug remains unclear [100].
It appears reasonable to discontinue PPI use in a patient who develops AIN, particularly if it occurs shortly after PPI initiation, once other causes of acute renal injury are ruled out and there are no other likely drug culprits. However, given the rarity of AIN, screening PPI users with serial assessments of serum creatinine or urinalysis is likely not warranted.
5.6.2 Acute Kidney Injury and Chronic Kidney Disease
Meta-analysis of seven studies examining the association between PPI use and development of acute kidney injury (AKI) found a pooled RR of 1.61 (1.16–2.22) with high heterogeneity between studies. This increased risk was limited to subjects aged < 60 years and those who were not using PPIs at baseline [101].
A second meta-analysis examined the association between PPI use and chronic kidney disease (CKD) and included five studies with over 500,000 subjects. Pooled RR of 1.22 (95% CI 1.14–1.30) existed for association between PPI use and incident CKD and 1.88 (95% CI 1.71–2.06) for developing end-stage renal disease (ESRD). The same meta-analysis also examined the association between H2RAs and CKD and found no significant association [102]. Risk of incident CKD and ESRD positively correlates with duration of PPI exposure over the first two years of use before declining somewhat thereafter [103], while twice-daily PPI dosing has also been reported to carry excess risk [104].
AIN offers a potential mechanism for development of AKI, but the mechanism by which PPI use might predispose to CKD is unclear. It has been suggested that CKD might be the result of incomplete recovery from AKI, but this is contradicted by analysis of Department of Veterans Affairs data showing increased risk of incident CKD even after censoring subjects who developed AKI prior to development of CKD (HR 1.26, 95% CI 1.20–1.33 for comparison between new PPI users and new H2RA users) [105]. Alternative hypotheses include development of CKD as a consequence of chronically low magnesium or high asymmetrical dimethylarginine levels, both of which have been associated with reduction in kidney function [106,107,108]. Changes in gut microbiota and decreased gut barrier integrity have also been described in CKD. It has been speculated that translocation of endotoxin into systemic circulation may contribute to uremic symptoms and CKD progression [109]. PPIs are also known to affect the gut microbiota, raising the possibility that drug-induced alterations in bacterial composition might produce deleterious effects on kidney function [110]. To date, these mechanisms remain largely theoretical.
Studies of renal complications among PPI users have generally been of high quality and have involved large cohorts with robust statistical methods. They are limited, however, by the potential for unrecorded medication use, particularly of over the counter NSAIDs and ASA which are frequently used together with PPIs and are themselves among the most common causes of AKI.
Given the morbidity and impact of AKI and CKD, the presence of a truly causal relationship between PPI use and these outcomes would be highly relevant. Persons with CKD have a high rate of PPI use as well as high rates of gastrointestinal bleeding, often related to ASA and anticoagulant use for cardiac comorbidities common among persons with CKD. The withdrawal of PPI use in this population would likely lead to a significant increase in gastrointestinal complications, most notably upper gastrointestinal bleeding. Any decision to discontinue PPI use in this population needs to be carefully considered.
6 Drug Interactions
6.1 Effect on Clopidogrel Metabolism
By far the most highly publicized purported risk associated with PPI use has been that of omeprazole on the metabolism of clopidogrel. Clopidogrel is a prodrug that is oxidized in two sequential steps by the cytochrome P450 system to form its active metabolite, with the CYP2C19 enzyme participating in both steps [111]. The CYP2C19 pathway is also involved in the activation and metabolism of several drugs, including the PPI omeprazole. Therefore, it is theorized that medications which require CYP2C19-mediated metabolism could reduce the activation of clopidogrel into its active metabolite, and thereby interfere with its desired therapeutic effect.
Ex vivo studies demonstrated lesser reduction in platelet reactivity in subjects starting omeprazole together with clopidogrel and aspirin following elective PCI [112] while observational studies reported higher rates of death or rehospitalization for cardiovascular disease among subjects prescribed clopidogrel in combination with a PPI following admission for ACS compared with those using clopidogrel alone [113, 114]. Ultimately the FDA issued a warning in 2009 against the combination of omeprazole with clopidogrel [115].
Despite this early concern, post-hoc analysis of the PRINCIPLE-TIMI 44 and TRITON-TIMI 38 trials demonstrated no difference in adverse clinical outcomes including myocardial infarction and stent thrombosis between PPI users and non-users [116]. French FAST-MI registry data from 3670 subjects with acute myocardial infarction found no effect of PPI use or CYP2C19 genotype on in-hospital events or one-year survival [117]. Finally, the Clopidogrel and the Optimization of Gastrointestinal Events (COGENT) trial randomized subjects requiring dual antiplatelet therapy to receive aspirin plus a fixed-dose combination of clopidogrel 75 mg and omeprazole 20 mg versus aspirin and clopidogrel alone. The trial was terminated early when the study sponsor filed for bankruptcy, but analysis of 3761 subjects enrolled prior to termination found no difference in cardiovascular endpoints at 180 days, while gastrointestinal events were significantly less frequent among omeprazole users [118]. Post-hoc analysis of COGENT looking at the highest-risk subjects for cardiovascular events (those with PCI within 14 days of randomization and those presenting with ACS) again identified no excess risk of cardiovascular events [119].
Despite the evidence of the safety of the combination of omeprazole and clopidogrel, the FDA reiterated in 2016 that the combination is to be avoided [120]. There is no such restriction with other PPIs as ex vivo studies of lansoprazole, esomeprazole, and pantoprazole have never demonstrated significant impact on platelet inhibition by clopidogrel [121, 122]. Given the large number of generally interchangeable PPIs on the market, the substitution of omeprazole for another agent amongst clopidogrel users is not overly onerous. It is somewhat unusual, however, that the agent with by far the most extensive evidence supporting its safety in this setting is the one drug to be avoided.
6.2 Drug Absorption
The absorption of orally administered drugs depends on the structural and functional integrity of the GI tract. Increasing gastric pH can alter the timing of drug release, affect solubility of co-administered molecules, and change the kinetics of pro-drugs.
The effect of acid suppression on oral absorption of small molecules has been most extensively studied in the oncology literature. Median progression free survival among metastatic renal cell cancer subjects receiving sunitinib was significantly shorter among subjects receiving concomitant acid suppression (40.9 weeks, 26.1–74.4 vs 62.4, 42.0–82.7 weeks; p = 0.02) [123]. Similarly, poor results were seen among advanced non-small cell lung cancer subjects receiving erlotinib [124]. PPI use together with capecitabine in colorectal cancer was not associated with decreased recurrence-free survival, although there was a trend in this direction and the total number of subjects was small [125]. Finally, post-hoc analysis of TRIO-013 subjects with metastatic gastroesophageal cancer subjects receiving capecitabine and oxaliplatin found that subjects using PPIs negatively affected both progression-free and overall survival [126].
The ledipasvir component of the hepatitis C treatment ledipasvir/sofosbuvir (Harvoni) is essentially insoluble at pH > 4. Package labeling recommends that no more than 20 mg of omeprazole (or equivalent) be taken concurrently with antiviral treatment in order to allow adequate absorption [127]. Real-world efficacy of ledipasvir/sofosbuvir treatment did not differ between PPI users and non-users; however, lower rates of sustained virologic response 12 weeks following treatment were seen among cirrhotic subjects using twice-daily PPI (aOR 0.11, 0.02–0.59; p = 0.005) that the study was not adequately powered to detect [128].
Azole antifungals are particularly sensitive to the effects of gastric pH. Acid suppression with omeprazole reduces the relative bioavailability of fluconazole by over 80% [129], while combining esomeprazole with posaconazole produces a 32% decrease [130, 131] Bioavailability of the HIV medication atazanavir is similarly reduced when given together with lansoprazole [131].
In contrast, PPI co-administration with the serotonin selective reuptake inhibitors (SSRIs) citalopram, escitalopram, and sertraline can increase serum SSRI concentrations by more than 90%, though the clinical impact of this effect is unclear [132]. Nifedipine and digoxin exhibit modestly greater absorption [133,134,135,136], while coadministration of dexlansoprazole with warfarin, diazepam, and phenytoin demonstrates no significant effect [137, 138].
Cefpodoxime, enoxacin [139, 140], and dipyridamole [141] have all exhibited decreased bioavailability when coadministered with H2RAs, while alendronate [142] is more readily absorbed. Similar effects would presumably be seen with PPIs.
Aside from the oncology literature, studies of drug absorption with acid suppression have, for the most part, been conducted in young, healthy subjects and have looked at only two drugs in combination. It is impossible to predict what the effects might be in elderly subject receiving multiple drugs. Further study is needed in this area. Until then, physicians should exercise caution, particularly with regard to chemotherapeutics, antimicrobials, and drugs whose absorption or release is known to be pH dependent. As always, the risks of discontinuing PPI therapy need to be put in the appropriate context of the benefits of optimally administering the desired therapy.
7 Overall Mortality
As PPI use is associated with several highly morbid conditions, it would be reasonable to believe that this association would extend to overall mortality. Several small cohort studies have examined whether an association exists between PPI use and mortality risk and have reported divergent results [143,144,145,146,147,148,149]. By far the largest study to address the issue used US Department of Veterans Affairs administrative data to identify a cohort of 257,977 new PPI users followed for a median of 5.71 years. PPI use was associated with an increased risk of death compared with a propensity score adjusted cohort of H2RA users (HR 1.16, CI 1.13–1.18]. A similar magnitude of risk was seen when the analysis was repeated among PPI users with and without a documented GI indication for acid suppression and among PPI users with and without established cardiovascular disease. There was a graded association between duration of exposure and risk of death up to 720 days, but notably prior PPI exposure (i.e. users who had discontinued PPIs for > 90 days) was associated with a greater risk of death than was current use (HR 1.53, CI 1.50–1.57 for prior use, HR 1.23, 95% 1.21–1.26 for current use). This is of particular interest, as multiple unmeasured confounders existed, including obesity, smoking, and use of anticoagulants, antiplatelet agents, and NSAIDs [150].
The biologic plausibility of the link between PPI use and mortality is contingent on there being a causal relationship between PPI use and conditions which are highly prevalent and highly morbid. As the associations between PPI use and the most clinically impactful of its associated adverse effects (renal injury, cardiac disease, dementia) are highly speculative, it remains difficult to make a causal link between PPI use and overall mortality.
8 Discussion
The volume of literature published on PPI-related adverse effects has reached daunting proportions. The vast majority of data reviewed stems from retrospective analyses of large-scale epidemiologic databases that are prone to residual confounding despite the best efforts of researchers. This is underscored by the frequent disagreement between studies using similar methodologies. PPI users are almost invariably in poorer overall health than non-users which makes them more likely to develop adverse clinical outcomes irrespective of the effect of PPIs. This confounding by indication is exceedingly difficult to adjust for and represents a limitation of research methodology as opposed to an adverse drug effect. Moreover, physiologic mechanisms that would provide a causal explanation for most of these associations are incompletely characterized or highly speculative or have only been demonstrated in animal models or in vitro.
The evidence for the majority of PPI-related adverse events reviewed here is weak and should not impact the decision to prescribe PPIs when indicated. Few, if any, of the studies reporting on adverse events have attempted to balance the benefits of PPI use with its purported harms. Even when ignoring the possibility of confounding influencing results, the majority of reported associations are of small magnitude and of questionable clinical relevance. Given the uncertainty about the causality of these relationships and leaving open the possibility that some or all of them may eventually prove to be legitimate, it is important for prescribers and care providers to be circumspect in their use of PPIs.
Adverse effects are not unique to PPIs. Their widespread use, however, magnifies the potential for harm. A number of studies have found a substantial proportion of PPI users to be on therapy for no appreciable reason, and PPI use may often be continued for empiric indication or syndrome where the evidence for the efficacy of long-term therapy is poor or nonexistent. As an example, PPI therapy is frequently initiated for non-specific upper gastrointestinal symptoms by primary care providers. In a review of outpatient PPI prescribing, a documented indication for therapy was not documented in one-third of PPI users, and in over half of instances the need for continuing PPI therapy was never re-addressed [150].
It is incumbent on all health care professionals involved in drug prescribing to be vigilant for patients who are using PPIs without clear indications, and to identify these candidates for deprescription. Medication list reviews and proactive questions raised by patients offer opportunities to discuss PPI safety and indications for continued PPI use. Deprescribing should be offered when a clear indication is absent, both to mitigate the remote possibility of PPI-related complications and to reduce pill burden and its associated economic costs. Where PPI therapy is indicated, there are consequences for its inappropriate discontinuation, which range from increased gastrointestinal symptomatology and its negative impact on quality of life, to highly morbid complications like gastrointestinal bleeding and perforation. For these patients, it is thus important for clinicians to emphasize both the magnitude and certainty of the benefits and to contrast this with the substantial disagreement and questionable relevance of purported harms.
References
IMS Institute for Healthcare Informatics. Declining medicine use and costs: For better or worse? A review of the use of medicines in the United States in 2012. Burlington, MA.
Schumock GT, Li EC, Suda KJ, Wiest MD, Stubbings J, Matusiak LM, et al. National trends in prescription drug expenditures and projections for 2015. AJHP. 2015;72(9):717–36.
Jaisser F, Beggah AT. The nongastric H+-K+-ATPases: molecular and functional properties. Am J Physiol. 1999;276(6):F812–24.
Novak I, Wang J, Henriksen KL, Haanes KA, Krabbe S, Nitschke R, et al. Pancreatic bicarbonate secretion involves two proton pumps. J Biol Chem. 2011;286(1):280–9.
Wang J, Barbuskaite D, Tozzi M, Giannuzzo A, Sorensen CE, Novak I. Proton pump inhibitors inhibit pancreatic secretion: role of gastric and non-gastric H+/K+-ATPases. PLoS One. 2015;10(5):e0126432.
Koop H, Klein M, Arnold R. Serum gastrin levels during long-term omeprazole treatment. Aliment Pharmacol Ther. 1990;4(2):131–8.
Lundell L, Vieth M, Gibson F, Nagy P, Kahrilas PJ. Systematic review: the effects of long-term proton pump inhibitor use on serum gastrin levels and gastric histology. Aliment Pharmacol Ther. 2015;42(6):649–63.
Sanduleanu S, Jonkers D, De Bruine A, Hameeteman W, Stockbrugger RW. Non-Helicobacter pylori bacterial flora during acid-suppressive therapy: differential findings in gastric juice and gastric mucosa. Aliment Pharmacol Ther. 2001;15(3):379–88.
Sharma BK, Santana IA, Wood EC, Walt RP, Pereira M, Noone P, et al. Intragastric bacterial activity and nitrosation before, during, and after treatment with omeprazole. BMJ. 1984;289(6447):717–9.
Del Piano M, Anderloni A, Balzarini M, Ballare M, Carmagnola S, Montino F, et al. The innovative potential of Lactobacillus rhamnosus LR06, Lactobacillus pentosus LPS01, Lactobacillus plantarum LP01, and Lactobacillus delbrueckii Subsp. delbrueckii LDD01 to restore the “gastric barrier effect” in patients chronically treated with PPI: a pilot study. J Clin Gastroenterol. 2012;46(Suppl):S18–26.
Vesper BJ, Jawdi A, Altman KW, Haines GK 3rd, Tao L, Radosevich JA. The effect of proton pump inhibitors on the human microbiota. Curr Drug Metab. 2009;10(1):84–9.
Altman KW, Chhaya V, Hammer ND, Pavlova S, Vesper BJ, Tao L, et al. Effect of proton pump inhibitor pantoprazole on growth and morphology of oral Lactobacillus strains. Laryngoscope. 2008;118(4):599–604.
Parsons BN, Ijaz UZ, D’Amore R, Burkitt MD, Eccles R, Lenzi L, et al. Comparison of the human gastric microbiota in hypochlorhydric states arising as a result of Helicobacter pylori-induced atrophic gastritis, autoimmune atrophic gastritis and proton pump inhibitor use. PLoS Pathog. 2017;13(11):e1006653.
Husebye E, Skar V, Hoverstad T, Melby K. Fasting hypochlorhydria with gram positive gastric flora is highly prevalent in healthy old people. Gut. 1992;33(10):1331–7.
Stotzer PO, Bjornsson ES, Abrahamsson H. Interdigestive and postprandial motility in small-intestinal bacterial overgrowth. Scand J Gastroenterol. 1996;31(9):875–80.
Castiglione F, Del Vecchio Blanco G, Rispo A, Petrelli G, Amalfi G, Cozzolino A, et al. Orocecal transit time and bacterial overgrowth in patients with Crohn’s disease. J Clin Gastroenterol. 2000;31(1):63–6.
McLoughlin GA, Hede JE, Temple JG, Bradley J, Chapman DM, McFarland J. The role of IgA in the prevention of bacterial colonization of the jejunum in the vagotomized subject. Br J Surg. 1978;65(6):435–7.
Lo WK, Chan WW. Proton pump inhibitor use and the risk of small intestinal bacterial overgrowth: a meta-analysis. Clin Gastroenterol Hepatol. 2013;11(5):483–90.
Su T, Lai S, Lee A, He X, Chen S. Meta-analysis: proton pump inhibitors moderately increase the risk of small intestinal bacterial overgrowth. J Gastroenterol. 2018;53(1):27–36.
Magill SS, Edwards JR, Bamberg W, Beldavs ZG, Dumyati G, Kainer MA, et al. Multistate point-prevalence survey of health care-associated infections. N Engl J Med. 2014;370(13):1198–208.
Rao A, Jump RL, Pultz NJ, Pultz MJ, Donskey CJ. In vitro killing of nosocomial pathogens by acid and acidified nitrite. Antimicrob Agents Chemother. 2006;50(11):3901–4.
Clooney AG, Bernstein CN, Leslie WD, Vagianos K, Sargent M, Laserna-Mendieta EJ, et al. A comparison of the gut microbiome between long-term users and non-users of proton pump inhibitors. Aliment Pharmacol Ther. 2016;43(9):974–84.
De La Cochetiere MF, Durand T, Lepage P, Bourreille A, Galmiche JP, Dore J. Resilience of the dominant human fecal microbiota upon short-course antibiotic challenge. J Clin Microbiol. 2005;43(11):5588–92.
Freedberg DE, Toussaint NC, Chen SP, Ratner AJ, Whittier S, Wang TC, et al. Proton pump inhibitors alter specific taxa in the human gastrointestinal microbiome: a crossover trial. Gastroenterology. 2015;149(4):883 e9–885 e9.
Loo VG, Bourgault AM, Poirier L, Lamothe F, Michaud S, Turgeon N, et al. Host and pathogen factors for Clostridium difficile infection and colonization. N Engl J Med. 2011;365(18):1693–703.
Kwok CS, Arthur AK, Anibueze CI, Singh S, Cavallazzi R, Loke YK. Risk of Clostridium difficile infection with acid suppressing drugs and antibiotics: meta-analysis. Am J Gastroenterol. 2012;107(7):1011–9.
Tariq R, Singh S, Gupta A, Pardi DS, Khanna S. Association of gastric acid suppression with recurrent Clostridium difficile infection: a systematic review and meta-analysis. JAMA Intern Med. 2017;177(6):784–91.
Leonard J, Marshall JK, Moayyedi P. Systematic review of the risk of enteric infection in patients taking acid suppression. Am J Gastroenterol. 2007;102(9):2047–56 (quiz 57).
Hassing RJ, Verbon A, de Visser H, Hofman A, Stricker BH. Proton pump inhibitors and gastroenteritis. Eur J Epidemiol. 2016;31(10):1057–63.
Xu HB, Wang HD, Li CH, Ye S, Dong MS, Xia QJ, et al. Proton pump inhibitor use and risk of spontaneous bacterial peritonitis in cirrhotic patients: a systematic review and meta-analysis. GMR. 2015;14(3):7490–501.
Dam G, Vilstrup H, Watson H, Jepsen P. Proton pump inhibitors as a risk factor for hepatic encephalopathy and spontaneous bacterial peritonitis in patients with cirrhosis with ascites. Hepatology. 2016;64(4):1265–72.
Terg R, Casciato P, Garbe C, Cartier M, Stieben T, Mendizabal M, et al. Proton pump inhibitor therapy does not increase the incidence of spontaneous bacterial peritonitis in cirrhosis: a multicenter prospective study. J Hepatol. 2015;62(5):1056–60.
Tergast TL, Wranke A, Laser H, Gerbel S, Manns MP, Cornberg M, et al. Dose-dependent impact of proton pump inhibitors on the clinical course of spontaneous bacterial peritonitis. Liver Int. 2018;38(9):1602–13.
Song H, Ekheden IG, Zheng Z, Ericsson J, Nyren O, Ye W. Incidence of gastric cancer among patients with gastric precancerous lesions: observational cohort study in a low risk Western population. BMJ. 2015;351:h3867.
Mauch F, Bode G, Malfertheiner P. Identification and characterization of an ATPase system of Helicobacter pylori and the effect of proton pump inhibitors. Am J Gastroenterol. 1993;88(10):1801–2.
Stoschus B, Dominguez-Munoz JE, Kalhori N, Sauerbruch T, Malfertheiner P. Effect of omeprazole on Helicobacter pylori urease activity in vivo. Eur J Gastroenterol Hepatol. 1996;8(8):811–3.
Logan RP, Walker MM, Misiewicz JJ, Gummett PA, Karim QN, Baron JH. Changes in the intragastric distribution of Helicobacter pylori during treatment with omeprazole. Gut. 1995;36(1):12–6.
Kuipers EJ, Uyterlinde AM, Pena AS, Hazenberg HJ, Bloemena E, Lindeman J, et al. Increase of Helicobacter pylori-associated corpus gastritis during acid suppressive therapy: implications for long-term safety. Am J Gastroenterol. 1995;90(9):1401–6.
Stolte M, Meining A, Schmitz JM, Alexandridis T, Seifert E. Changes in Helicobacter pylori-induced gastritis in the antrum and corpus during 12 months of treatment with omeprazole and lansoprazole in patients with gastro-oesophageal reflux disease. Aliment Pharmacol Ther. 1998;12(3):247–53.
Solcia E, Fiocca R, Havu N, Dalvag A, Carlsson R. Gastric endocrine cells and gastritis in patients receiving long-term omeprazole treatment. Digestion. 1992;51(Suppl 1):82–92.
Creutzfeldt W, Lamberts R. Inter-relationship between serum gastrin levels, gastric mucosal histology and gastric endocrine cell growth. Digestion. 1992;51(Suppl 1):76–81.
Eissele R, Brunner G, Simon B, Solcia E, Arnold R. Gastric mucosa during treatment with lansoprazole: Helicobacter pylori is a risk factor for argyrophil cell hyperplasia. Gastroenterology. 1997;112(3):707–17.
Moayyedi P, Wason C, Peacock R, Walan A, Bardhan K, Axon AT, et al. Changing patterns of Helicobacter pylori gastritis in long-standing acid suppression. Helicobacter. 2000;5(4):206–14.
Kuipers EJ, Lundell L, Klinkenberg-Knol EC, Havu N, Festen HP, Liedman B, et al. Atrophic gastritis and Helicobacter pylori infection in patients with reflux esophagitis treated with omeprazole or fundoplication. N Engl J Med. 1996;334(16):1018–22.
Brusselaers N, Wahlin K, Engstrand L, Lagergren J. Maintenance therapy with proton pump inhibitors and risk of gastric cancer: a nationwide population-based cohort study in Sweden. BMJ Open. 2017;7(10):e017739.
Ahn JS, Eom CS, Jeon CY, Park SM. Acid suppressive drugs and gastric cancer: a meta-analysis of observational studies. World J Gastroenterol. 2013;19(16):2560–8.
Tran-Duy A, Spaetgens B, Hoes AW, de Wit NJ, Stehouwer CD. Use of proton pump inhibitors and risks of fundic gland polyps and gastric cancer: systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2016;14(12):1706 e5–1719 e5.
Martin FC, Chenevix-Trench G, Yeomans ND. Systematic review with meta-analysis: fundic gland polyps and proton pump inhibitors. Aliment Pharmacol Ther. 2016;44(9):915–25.
Kinoshita Y, Tojo M, Yano T, Kitajima N, Itoh T, Nishiyama K, et al. Incidence of fundic gland polyps in patients without familial adenomatous polyposis. Gastrointest Endosc. 1993;39(2):161–3.
Wu TT, Kornacki S, Rashid A, Yardley JH, Hamilton SR. Dysplasia and dysregulation of proliferation in foveolar and surface epithelia of fundic gland polyps from patients with familial adenomatous polyposis. Am J Surg Pathol. 1998;22(3):293–8.
Keszthelyi D, Jansen SV, Schouten GA, de Kort S, Scholtes B, Engels LG, et al. Proton pump inhibitor use is associated with an increased risk for microscopic colitis: a case-control study. Aliment Pharmacol Ther. 2010;32(9):1124–8.
Pascua MF, Kedia P, Weiner MG, Holmes J, Ellenberg J, Lewis JD. Microscopic colitis and medication use. Clin Med Insights Gastroenterol. 2010;2010(3):11–9.
Fernandez-Banares F, de Sousa MR, Salas A, Beltran B, Piqueras M, Iglesias E, et al. Epidemiological risk factors in microscopic colitis: a prospective case-control study. Inflamm Bowel Dis. 2013;19(2):411–7.
Masclee GM, Coloma PM, Kuipers EJ, Sturkenboom MC. Increased risk of microscopic colitis with use of proton pump inhibitors and non-steroidal anti-inflammatory drugs. Am J Gastroenterol. 2015;110(5):749–59.
Bonderup OK, Fenger-Gron M, Wigh T, Pedersen L, Nielsen GL. Drug exposure and risk of microscopic colitis: a nationwide Danish case-control study with 5751 cases. Inflamm Bowel Dis. 2014;20(10):1702–7.
Verhaegh BP, de Vries F, Masclee AA, Keshavarzian A, de Boer A, Souverein PC, et al. High risk of drug-induced microscopic colitis with concomitant use of NSAIDs and proton pump inhibitors. Aliment Pharmacol Ther. 2016;43(9):1004–13.
Andersen T, Andersen JR, Tvede M, Franzmann MB. Collagenous colitis: are bacterial cytotoxins responsible? Am J Gastroenterol. 1993;88(3):375–7.
Imhann F, Vich Vila A, Bonder MJ, Lopez Manosalva AG, Koonen DPY, Fu J, et al. The influence of proton pump inhibitors and other commonly used medication on the gut microbiota. Gut microbes. 2017;8(4):351–8.
Crowson MS, Shull GE. Isolation and characterization of a cDNA encoding the putative distal colon H+, K(+)-ATPase. Similarity of deduced amino acid sequence to gastric H+, K(+)-ATPase and Na+, K(+)-ATPase and mRNA expression in distal colon, kidney, and uterus. J Biol Chem. 1992;267(19):13740–8.
Lambert AA, Lam JO, Paik JJ, Ugarte-Gil C, Drummond MB, Crowell TA. Risk of community-acquired pneumonia with outpatient proton-pump inhibitor therapy: a systematic review and meta-analysis. PLoS One. 2015;10(6):e0128004.
Dickson RP, Erb-Downward JR, Martinez FJ, Huffnagle GB. The microbiome and the respiratory tract. Annu Rev Physiol. 2016;78:481–504.
El-Serag HB, Sonnenberg A. Comorbid occurrence of laryngeal or pulmonary disease with esophagitis in United States military veterans. Gastroenterology. 1997;113(3):755–60.
Filion KB, Chateau D, Targownik LE, Gershon A, Durand M, Tamim H, et al. Proton pump inhibitors and the risk of hospitalisation for community-acquired pneumonia: replicated cohort studies with meta-analysis. Gut. 2014;63(4):552–8.
Alshamsi F, Belley-Cote E, Cook D, Almenawer SA, Alqahtani Z, Perri D, et al. Efficacy and safety of proton pump inhibitors for stress ulcer prophylaxis in critically ill patients: a systematic review and meta-analysis of randomized trials. Crit Care. 2016;20(1):120.
Lam JR, Schneider JL, Zhao W, Corley DA. Proton pump inhibitor and histamine 2 receptor antagonist use and vitamin B12 deficiency. JAMA. 2013;310(22):2435–42.
Qorraj-Bytyqi H, Hoxha R, Sadiku S, Bajraktari IH, Sopjani M, Thaci K, et al. Proton pump inhibitors intake and iron and vitamin B12 status: a prospective comparative study with a follow up of 12 months. Open Access Maced. J Med Sci. 2018;6(3):442–6.
Lam JR, Schneider JL, Quesenberry CP, Corley DA. Proton pump inhibitor and histamine-2 receptor antagonist use and iron deficiency. Gastroenterology. 2017;152(4):821 e1–829 e1.
Schade SG, Cohen RJ, Conrad ME. Effect of hydrochloric acid on iron absorption. N Engl J Med. 1968;279(13):672–4.
Golubov J, Flanagan P, Adams P. Inhibition of iron absorption by omeprazole in rat model. Dig Dis Sci. 1991;36(4):405–8.
Koop H, Bachem MG. Serum iron, ferritin, and vitamin B12 during prolonged omeprazole therapy. J Clin Gastroenterol. 1992;14(4):288–92.
Stewart CA, Termanini B, Sutliff VE, Serrano J, Yu F, Gibril F, et al. Iron absorption in patients with Zollinger–Ellison syndrome treated with long-term gastric acid antisecretory therapy. Aliment Pharmacol Ther. 1998;12(1):83–98.
Hutchinson C, Geissler CA, Powell JJ, Bomford A. Proton pump inhibitors suppress absorption of dietary non-haem iron in hereditary haemochromatosis. Gut. 2007;56(9):1291–5.
Sarzynski E, Puttarajappa C, Xie Y, Grover M, Laird-Fick H. Association between proton pump inhibitor use and anemia: a retrospective cohort study. Dig Dis Sci. 2011;56(8):2349–53.
William JH, Danziger J. Proton-pump inhibitor-induced hypomagnesemia: current research and proposed mechanisms. World J Nephrol. 2016;5(2):152–7.
Zipursky J, Macdonald EM, Hollands S, Gomes T, Mamdani MM, Paterson JM, et al. Proton pump inhibitors and hospitalization with hypomagnesemia: a population-based case–control study. PLoS Med. 2014;11(9):e1001736.
Sharara AI, Chalhoub JM, Hammoud N, Harb AH, Sarkis FS, Hamadeh G. Low prevalence of hypomagnesemia in long-term recipients of proton pump inhibitors in a managed care cohort. Clin Gastroenterol Hepatol. 2016;14(2):317–21.
Hess MW, Hoenderop JG, Bindels RJ, Drenth JP. Systematic review: hypomagnesaemia induced by proton pump inhibition. Aliment Pharmacol Ther. 2012;36(5):405–13.
Recker RR. Calcium absorption and achlorhydria. N Engl J Med. 1985;313(2):70–3.
Wright MJ, Sullivan RR, Gaffney-Stomberg E, Caseria DM, O’Brien KO, Proctor DD, et al. Inhibiting gastric acid production does not affect intestinal calcium absorption in young, healthy individuals: a randomized, crossover, controlled clinical trial. J Bone Miner Res. 2010;25(10):2205–11.
Hansen KE, Jones AN, Lindstrom MJ, Davis LA, Ziegler TE, Penniston KL, et al. Do proton pump inhibitors decrease calcium absorption? J Bone Miner Res. 2010;25(12):2786–95.
Attwood SE, Ell C, Galmiche JP, Fiocca R, Hatlebakk JG, Hasselgren B, et al. Long-term safety of proton pump inhibitor therapy assessed under controlled, randomised clinical trial conditions: data from the SOPRAN and LOTUS studies. Aliment Pharmacol Ther. 2015;41(11):1162–74.
Vestergaard P, Rejnmark L, Mosekilde L. Proton pump inhibitors, histamine H2 receptor antagonists, and other antacid medications and the risk of fracture. Calcif Tissue Int. 2006;79(2):76–83.
Yang YX, Lewis JD, Epstein S, Metz DC. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296(24):2947–53.
Targownik LE, Lix LM, Metge CJ, Prior HJ, Leung S, Leslie WD. Use of proton pump inhibitors and risk of osteoporosis-related fractures. CMAJ. 2008;179(4):319–26.
Zhou B, Huang Y, Li H, Sun W, Liu J. Proton-pump inhibitors and risk of fractures: an update meta-analysis. Osteoporos Int. 2016;27(1):339–47.
Targownik LE, Leslie WD, Davison KS, Goltzman D, Jamal SA, Kreiger N, et al. The relationship between proton pump inhibitor use and longitudinal change in bone mineral density: a population-based study [corrected] from the Canadian Multicentre Osteoporosis Study (CaMos). Am J Gastroenterol. 2012;107(9):1361–9.
Targownik LE, Goertzen AL, Luo Y, Leslie WD. Long-term proton pump inhibitor use is not associated with changes in bone strength and structure. Am J Gastroenterol. 2017;112(1):95–101.
Clark DW, Strandell J. Myopathy including polymyositis: a likely class adverse effect of proton pump inhibitors? Eur J Clin Pharmacol. 2006;62(6):473–9.
Colmenares EW, Pappas AL. Proton pump inhibitors: risk for myopathy? Ann Pharmacother. 2017;51(1):66–71.
Gomm W, von Holt K, Thome F, Broich K, Maier W, Fink A, et al. Association of proton pump inhibitors with risk of dementia: a pharmacoepidemiological claims data analysis. JAMA Neurol. 2016;73(4):410–6.
Tai SY, Chien CY, Wu DC, Lin KD, Ho BL, Chang YH, et al. Risk of dementia from proton pump inhibitor use in Asian population: a nationwide cohort study in Taiwan. PLoS One. 2017;12(2):e0171006.
Lochhead P, Hagan K, Joshi AD, Khalili H, Nguyen LH, Grodstein F, et al. Association between proton pump inhibitor use and cognitive function in women. Gastroenterology. 2017;153(4):971 e4–979 e4.
Taipale H, Tolppanen AM, Tiihonen M, Tanskanen A, Tiihonen J, Hartikainen S. No association between proton pump inhibitor use and risk of Alzheimer’s disease. Am J Gastroenterol. 2017;112(12):1802–8.
Wod M, Hallas J, Andersen K, Garcia Rodriguez LA, Christensen K, Gaist D. Lack of association between proton pump inhibitor use and cognitive decline. Clin Gastroenterol Hepatol. 2018;16(5):681–9.
Booker A, Jacob LE, Rapp M, Bohlken J, Kostev K. Risk factors for dementia diagnosis in German primary care practices. Int Psychogeriatr. 2016;28(7):1059–65.
Goldstein FC, Steenland K, Zhao L, Wharton W, Levey AI, Hajjar I. Proton pump inhibitors and risk of mild cognitive impairment and dementia. J Am Geriatr Soc. 2017;65(9):1969–74.
Badiola N, Alcalde V, Pujol A, Munter LM, Multhaup G, Lleo A, et al. The proton-pump inhibitor lansoprazole enhances amyloid beta production. PLoS One. 2013;8(3):e58837.
Blank ML, Parkin L, Paul C, Herbison P. A nationwide nested case–control study indicates an increased risk of acute interstitial nephritis with proton pump inhibitor use. Kidney Int. 2014;86(4):837–44.
Geevasinga N, Coleman PL, Webster AC, Roger SD. Proton pump inhibitors and acute interstitial nephritis. Clin Gastroenterol Hepatol. 2006;4(5):597–604.
Coca SG, Perazella MA. Early steroid treatment for drug-induced acute interstitial nephritis. Nat Clin Pract Nephrol. 2008;4(6):298–9.
Yang Y, George KC, Shang WF, Zeng R, Ge SW, Xu G. Proton-pump inhibitors use, and risk of acute kidney injury: a meta-analysis of observational studies. Drug Des Dev Ther. 2017;11:1291–9.
Wijarnpreecha K, Thongprayoon C, Chesdachai S, Panjawatanana P, Ungprasert P, Cheungpasitporn W. Associations of proton-pump inhibitors and H2 receptor antagonists with chronic kidney disease: a meta-analysis. Dig Dis Sci. 2017;62(10):2821–7.
Xie Y, Bowe B, Li T, Xian H, Balasubramanian S, Al-Aly Z. Proton pump inhibitors and risk of incident CKD and progression to ESRD. JASN. 2016;27(10):3153–63.
Lazarus B, Chen Y, Wilson FP, Sang Y, Chang AR, Coresh J, et al. Proton pump inhibitor use and the risk of chronic kidney disease. JAMA Intern Med. 2016;176(2):238–46.
Xie Y, Bowe B, Li T, Xian H, Yan Y, Al-Aly Z. Long-term kidney outcomes among users of proton pump inhibitors without intervening acute kidney injury. Kidney Int. 2017;91(6):1482–94.
Tin A, Grams ME, Maruthur NM, Astor BC, Couper D, Mosley TH, et al. Results from the atherosclerosis risk in communities study suggest that low serum magnesium is associated with incident kidney disease. Kidney Int. 2015;87(4):820–7.
Ghebremariam YT, LePendu P, Lee JC, Erlanson DA, Slaviero A, Shah NH, et al. Unexpected effect of proton pump inhibitors: elevation of the cardiovascular risk factor asymmetric dimethylarginine. Circulation. 2013;128(8):845–53.
Eiselt J, Rajdl D, Racek J, Vostry M, Rulcova K, Wirth J. Asymmetric dimethylarginine and progression of chronic kidney disease: a one-year follow-up study. Kidney Blood Pressure Res. 2014;39(1):50–7.
Ramezani A, Raj DS. The gut microbiome, kidney disease, and targeted interventions. JASN. 2014;25(4):657–70.
Klatte DCF, Gasparini A, Xu H, de Deco P, Trevisan M, Johansson ALV, et al. Association between proton pump inhibitor use and risk of progression of chronic kidney disease. Gastroenterology. 2017;153(3):702–10.
Kazui M, Nishiya Y, Ishizuka T, Hagihara K, Farid NA, Okazaki O, et al. Identification of the human cytochrome P450 enzymes involved in the two oxidative steps in the bioactivation of clopidogrel to its pharmacologically active metabolite. Drug Metab Dispos Biol Fate Chem. 2010;38(1):92–9.
Gilard M, Arnaud B, Cornily JC, Le Gal G, Lacut K, Le Calvez G, et al. Influence of omeprazole on the antiplatelet action of clopidogrel associated with aspirin: the randomized, double-blind OCLA (Omeprazole CLopidogrel Aspirin) study. J Am Coll Cardiol. 2008;51(3):256–60.
Ho PM, Maddox TM, Wang L, Fihn SD, Jesse RL, Peterson ED, et al. Risk of adverse outcomes associated with concomitant use of clopidogrel and proton pump inhibitors following acute coronary syndrome. JAMA. 2009;301(9):937–44.
Juurlink DN, Gomes T, Ko DT, Szmitko PE, Austin PC, Tu JV, et al. A population-based study of the drug interaction between proton pump inhibitors and clopidogrel. CMAJ. 2009;180(7):713–8.
prilosec IfhputtlocbmaPtahpaadiwomapa. United States Food and Drug Administration. https://wayback.archive-it.org/7993/20170406044850/. https://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/DrugSafetyInformationforHeathcareProfessionals/ucm190787.htm.
O’Donoghue ML, Braunwald E, Antman EM, Murphy SA, Bates ER, Rozenman Y, et al. Pharmacodynamic effect and clinical efficacy of clopidogrel and prasugrel with or without a proton-pump inhibitor: an analysis of two randomised trials. Lancet. 2009;374(9694):989–97.
Simon T, Steg PG, Gilard M, Blanchard D, Bonello L, Hanssen M, et al. Clinical events as a function of proton pump inhibitor use, clopidogrel use, and cytochrome P450 2C19 genotype in a large nationwide cohort of acute myocardial infarction: results from the French Registry of Acute ST-Elevation and non-ST-Elevation Myocardial Infarction (FAST-MI) registry. Circulation. 2011;123(5):474–82.
Bhatt DL, Cryer BL, Contant CF, Cohen M, Lanas A, Schnitzer TJ, et al. Clopidogrel with or without omeprazole in coronary artery disease. N Engl J Med. 2010;363(20):1909–17.
Vaduganathan M, Cannon CP, Cryer BL, Liu Y, Hsieh WH, Doros G, et al. Efficacy and safety of proton-pump inhibitors in high-risk cardiovascular subsets of the COGENT trial. Am J Med. 2016;129(9):1002–5.
Shell WE, DeWood MA, Peter T, Mickle D, Prause JA, Forrester JS, et al. Comparison of clinical signs and hemodynamic state in the early hours of transmural myocardial infarction. Am Heart J. 1982;104(3):521–8.
Small DS, Farid NA, Payne CD, Weerakkody GJ, Li YG, Brandt JT, et al. Effects of the proton pump inhibitor lansoprazole on the pharmacokinetics and pharmacodynamics of prasugrel and clopidogrel. J Clin Pharmacol. 2008;48(4):475–84.
Siller-Matula JM, Spiel AO, Lang IM, Kreiner G, Christ G, Jilma B. Effects of pantoprazole and esomeprazole on platelet inhibition by clopidogrel. Am Heart J. 2009;157(1):148 e1–5.
Ha VH, Ngo M, Chu MP, Ghosh S, Sawyer MB, Chambers CR. Does gastric acid suppression affect sunitinib efficacy in patients with advanced or metastatic renal cell cancer? J Oncol Pharm Pract. 2015;21(3):194–200.
Chu MP, Ghosh S, Chambers CR, Basappa N, Butts CA, Chu Q, et al. Gastric acid suppression is associated with decreased erlotinib efficacy in non-small-cell lung cancer. Clin Lung Cancer. 2015;16(1):33–9.
Sun J, Ilich AI, Kim CA, Chu MP, Wong GG, Ghosh S, et al. Concomitant administration of proton pump inhibitors and capecitabine is associated with increased recurrence risk in early stage colorectal cancer patients. Clin Colorectal Cancer. 2016;15(3):257–63.
Chu MP, Hecht JR, Slamon D, Wainberg ZA, Bang YJ, Hoff PM, et al. Association of proton pump inhibitors and capecitabine efficacy in advanced gastroesophageal cancer: secondary analysis of the TRIO-013/logic randomized clinical trial. JAMA Oncol. 2017;3(6):767–73.
Davies MJ, Thomas A. Thrombosis and acute coronary-artery lesions in sudden cardiac ischemic death. N Engl J Med. 1984;310(18):1137–40.
Tapper EB, Bacon BR, Curry MP, Dieterich DT, Flamm SL, Guest LE, et al. Evaluation of proton pump inhibitor use on treatment outcomes with ledipasvir and sofosbuvir in a real-world cohort study. Hepatology. 2016;64(6):1893–9.
Chin TW, Loeb M, Fong IW. Effects of an acidic beverage (Coca–Cola) on absorption of ketoconazole. Antimicrob Agents Chemother. 1995;39(8):1671–5.
Krishna G, Moton A, Ma L, Medlock MM, McLeod J. Pharmacokinetics and absorption of posaconazole oral suspension under various gastric conditions in healthy volunteers. Antimicrob Agents Chemother. 2009;53(3):958–66.
Tomilo DL, Smith PF, Ogundele AB, Difrancesco R, Berenson CS, Eberhardt E, et al. Inhibition of atazanavir oral absorption by lansoprazole gastric acid suppression in healthy volunteers. Pharmacotherapy. 2006;26(3):341–6.
Gjestad C, Westin AA, Skogvoll E, Spigset O. Effect of proton pump inhibitors on the serum concentrations of the selective serotonin reuptake inhibitors citalopram, escitalopram, and sertraline. Ther Drug Monit. 2015;37(1):90–7.
Soons PA, van den Berg G, Danhof M, van Brummelen P, Jansen JB, Lamers CB, et al. Influence of single- and multiple-dose omeprazole treatment on nifedipine pharmacokinetics and effects in healthy subjects. Eur J Clin Pharmacol. 1992;42(3):319–24.
Bliesath H, Huber R, Steinijans VW, Koch HJ, Kunz K, Wurst W. Pantoprazole does not interact with nifedipine in man under steady-state conditions. Int J Clin Pharmacol Ther. 1996;34(1 Suppl):S81–5.
Oosterhuis B, Jonkman JH, Andersson T, Zuiderwijk PB, Jedema JN. Minor effect of multiple dose omeprazole on the pharmacokinetics of digoxin after a single oral dose. Br J Clin Pharmacol. 1991;32(5):569–72.
Cohen AF, Kroon R, Schoemaker R, Hoogkamer H, van Vliet A. Influence of gastric acidity on the bioavailability of digoxin. Ann Intern Med. 1991;115(7):540–5.
Hartmann M, Huber R, Bliesath H, Steinijans VW, Koch HJ, Wurst W, et al. Lack of interaction between pantoprazole and digoxin at therapeutic doses in man. Int J Clin Pharmacol Ther. 1996;34(1 Suppl):S67–71.
Vakily M, Lee RD, Wu J, Gunawardhana L, Mulford D. Drug interaction studies with dexlansoprazole modified release (TAK-390MR), a proton pump inhibitor with a dual delayed-release formulation: results of four randomized, double-blind, crossover, placebo-controlled, single-centre studies. Clin Drug Investig. 2009;29(1):35–50.
Hughes GS, Heald DL, Barker KB, Patel RK, Spillers CR, Watts KC, et al. The effects of gastric pH and food on the pharmacokinetics of a new oral cephalosporin, cefpodoxime proxetil. Clin Pharmacol Ther. 1989;46(6):674–85.
Grasela TH Jr, Schentag JJ, Sedman AJ, Wilton JH, Thomas DJ, Schultz RW, et al. Inhibition of enoxacin absorption by antacids or ranitidine. Antimicrob Agents Chemother. 1989;33(5):615–7.
Derendorf H, VanderMaelen CP, Brickl RS, MacGregor TR, Eisert W. Dipyridamole bioavailability in subjects with reduced gastric acidity. J Clin Pharmacol. 2005;45(7):845–50.
Gertz BJ, Holland SD, Kline WF, Matuszewski BK, Freeman A, Quan H, et al. Studies of the oral bioavailability of alendronate. Clin Pharmacol Ther. 1995;58(3):288–98.
Bell JS, Strandberg TE, Teramura-Gronblad M, Laurila JV, Tilvis RS, Pitkala KH. Use of proton pump inhibitors and mortality among institutionalized older people. Arch Intern Med. 2010;170(17):1604–5.
Wilson N, Gnjidic D, March L, Sambrook P, Hilmer SN. Use of PPIs are not associated with mortality in institutionalized older people. Arch Intern Med. 2011;171(9):866 (author reply-7).
Teramura-Gronblad M, Bell JS, Poysti MM, Strandberg TE, Laurila JV, Tilvis RS, et al. Risk of death associated with use of PPIs in three cohorts of institutionalized older people in Finland. J Am Med Direct Assoc. 2012;13(5):488 e9–513 e9.
Maggio M, Corsonello A, Ceda GP, Cattabiani C, Lauretani F, Butto V, et al. Proton pump inhibitors and risk of 1-year mortality and rehospitalization in older patients discharged from acute care hospitals. JAMA Intern Med. 2013;173(7):518–23.
de Francisco ALM, Varas J, Ramos R, Merello JI, Canaud B, Stuard S, et al. Proton pump inhibitor usage and the risk of mortality in hemodialysis patients. Kidney Int Rep. 2018;3(2):374–84.
Pegoli MA, Dedigama M, Mangoni AA, Russell PT, Grzeskowiak LE, Thynne T. Proton pump inhibitors and risk of readmission and mortality in older patients discharged from a tertiary hospital to residential aged care facilities. Ther Adv Drug Saf. 2017;8(4):137–9.
Pello Lazaro AM, Cristobal C, Franco-Pelaez JA, Tarin N, Acena A, Carda R, et al. Use of proton-pump inhibitors predicts heart failure and death in patients with coronary artery disease. PLoS One. 2017;12(1):e0169826.
Heidelbaugh JJ, Goldberg KL, Inadomi JM. Magnitude and economic effect of overuse of antisecretory therapy in the ambulatory care setting. Am J Manag Care. 2010;16(9):e228–34.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for the preparation of this manuscript
Conflicts of Interest
Evan Elias has no conflicts of interest to declare. Laura E. Targownik has no conflicts of interest to declare that directly relate to this manuscript.
Rights and permissions
About this article
Cite this article
Elias, E., Targownik, L.E. The Clinician’s Guide to Proton Pump Inhibitor Related Adverse Events. Drugs 79, 715–731 (2019). https://doi.org/10.1007/s40265-019-01110-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-019-01110-3