Abstract
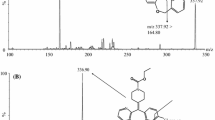
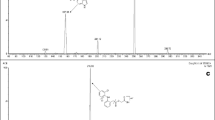
A sensitive LC–MS/MS method was developed and fully validated for the determination of spironolactone in human plasma using spironolactone-d6 as an internal standard (IS) after one-step liquid–liquid extraction with methyl tert-butyl ether: methylene chloride (MC) = 8:2 (v/v). Detection was performed using electrospray ionization in positive ion multiple reaction monitoring mode by monitoring the transitions: m/z 341.2 → 107.2 for spironolactone and m/z 347.1 → 107.2 for IS. Chromatographic separation was performed on a Cadenza CD-C18 column (3.0 × 100 mm i.d. 3 µm) with an isocratic mobile phase, which consisted of 0.1 % formic acid in water: methanol (30: 70, v/v), with a gradient flow rate as follow: 0–3.2 m, 320 µL/min; 3.2–3.5 m, 320–180 µL/min; 3.5–6.7 m, 180 µL/min; 6.7–7.0 m, 180–320 µL/min. The calibration curve was linear (correlation coefficients were >0.99) over the concentration range (0.5–150 ng/mL). The intraday and interday precisions ranged 0.89–6.00 and 1.20–10.511 %, respectively, and its accuracies ranged 96.90–105.08 and 97.99–104.13 %, respectively. The devised method was successfully applied in a bioequivalence study of two formulations of spironolactone, Spiracton tablet® and Aldactone tablet® in 50 healthy Korean male volunteers following single oral administration.





Similar content being viewed by others
References
BA calc 2007 software for windows (2007) KFDA. http://www.kfda.go.kr/
Brater DC (2000) Pharmacology of diuretics. Am J Med Sci 319(1):38–50
Dong H, Xu F, Zhang Z, Tian Y, Chen Y (2006) Simultaneous determination of spironolactone and its active metabolite carenone in human plasma by HPLC-APCI-MS. J Mass Spectr 41:477–486
Jankowski A, Storek-Jankowska A, Lamparczyk H (1996) Stimultanous determination of spironolactone and its metabolites in human plasma. J Pharm Biomed Anal 14:1359–1365
MFDS (2013) Guideline on bioanalytical method validation, bioequivalence division, Drug Evaluation Department, Ministry of Food and Drug Safety. Accessed http://www.mfds.go.kr/jsp/common/download.jsp?fileinfo=S*1*%BB%FD%C3%BC%BD%C3%B7%E1%20%BA%D0%BC%AE%B9%FD%20%B9%EB%B8%AE%B5%A5%C0%CC%BC%C7%20%B0%A1%C0%CC%B5%E5%B6%F3%C0%CE(%C0%CE%BC%E2).pdf*ad564b0ca9574150ff99975a65e8b796*pdf*/files/upload/1/TB_O_ANNOUNCE/7560/ad564b0ca9574150ff99975a65e8b796*2390643*2013:12:19%2017:21:33
Sica DA (2005) Pharmacokinetics and pharmacodynamics of mineralocorticoid blocking agents and their effects on potassium homeostasis. Heart Fail Rev 10:23–29
Sora DI, Udrescu S, Albu F, David V, Medvedovici A (2010) Analytical issues in HPLC/MS/MS simultaneous assay of furosemide, spironolactone and canrenone in human plasma samples. J Pharm Biomen Anal 52:734–740
US Department of Health & Human Service, Food and Drug Administration, Guidance for Industry, Bioanalytical Method Validation (2001) US FDA. http://www.fda.gov/downloads/regulatoryinformation/guidances/ucm195951.pdf
Valse L, Imre S, Muntean D, Achim M, Muntean DL (2011) Determination of Spironolactone and canrenone in human plasma by high-performance liquid chromatography with mass spectrometry detection. Croat Chem Acta 84(3):361–366
World Medical Association (2000) Declaration of Helsinki: ethical principles for medical research involving human subjects. As amended by the 52nd World Medical Assembly, Edinburgh
Xu D, Li H (2009) Bioequivalence and pharmacokinetics of spironolactone tablets in healthy volunteers. Chin J Clin Pharmacol 03:227–230
Xu FG, Zhang ZJ, Dong HJ, Tian Y, Liu Y, Chen Y (2008) Bioequivalence assessment of two formulations of spironolactone in chinese healthy Male Volunteers. Arzneimittelforschung 3:117–121
Acknowledgments
All authors (J.-H. Lee, T.-G. An, S. J. Kim, W.-S. Shim, K.-T. Lee) declare that they have no conflict of interest. This work was supported by Daewon pharm., Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jeong-Hun Lee and Tae-Gil have equal contribution in this work.
Rights and permissions
About this article
Cite this article
Lee, JH., An, TG., Kim, S.J. et al. Development of liquid chromatography tandem mass spectrometry method for determination of spironolactone in human plasma: application to a bioequivalence study of Daewon Spiracton tablet® (spironolactone 50 mg). Journal of Pharmaceutical Investigation 45, 601–609 (2015). https://doi.org/10.1007/s40005-015-0197-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40005-015-0197-9