Abstract
Background
There have been no reports of human herpesvirus-6 (HHV-6) encephalitis treatment based on both HHV-6 DNA load and the antiviral agent’s concentration in the cerebrospinal fluid (CSF).
Patient
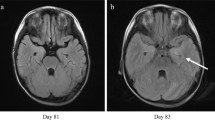
A 20-year-old male with a hematological malignancy developed HHV-6 encephalitis 15 days after unrelated cord blood transplantation (UCBT). He had fever, chest pain, memory impairment, and insomnia. His CSF showed no increased cell counts, but the amount of HHV-6 DNA was elevated to 2.0 × 106 copies/ìgDNA. Magnetic resonance imaging (MRI) of the head revealed abnormal high-intensity signals in the left limbic system on T2-weighted and diffusion-weighted images. Intravenous administration of ganciclovir (GCV) was initiated at 5 mg/kg every 12 h on day 18, and was continued until day 137. The amount of HHV-6 DNA in the plasma became undetectable on day 25. The HHV-6 load in the CSF decreased to 1.5 × 103 copies/ìgDNA on day 32, and reached undetectable levels on day 53. The mean concentration of GCV 1 h after an infusion of 5 mg/kg was 4.12 mg/mL in plasma and 0.7 mg/mL in CSF. The chest pain and insomnia disappeared on days 35 and 47, respectively. Memory defects recovered up to day 85.
Conclusion
Serial quantification of HHV-6 DNA in CSF may be useful for successful treatment with GCV in post-transplant HHV-6 encephalitis.



Similar content being viewed by others
References
Sakai R, Kanamori H, Motohashi K, Yamamoto W, Matsuura S, Fujita A, Ohshima R, Kuwabara H, Tanaka M, Fujita H, Maruta A, Ishigatsubo Y, Fujisawa S. Long-term outcome of human herpesvirus-6 encephalitis after allogeneic stem cell transplantation. Biol Blood Marrow Transplant. 2011;17:1389–94.
Zerr DM, Gupta D, Huang ML, Carter R, Corey L. Effect of antivirals on human herpesvirus 6 replication in hematopoietic stem cell transplant recipients. Clin Infect Dis. 2002;34:309–17.
Nakazawa Y, Suzuki T, Fukuyama T, Katsuyama Y, Tanaka M, Yanagisawa R, Sakashita K, Shiohara M, Koike K. Urinary excretion of ganciclovir contributes to improvement of adenovirus-associated hemorrhagic cystitis after allogeneic bone marrow transplantation. Pediatr Transplant. 2009;13:632–5.
Olli-Lähdesmäki T, Haataja L, Parkkola R, Waris M, Bleyzac N, Ruuskanen O. High-dose ganciclovir in HHV-6 encephalitis of an immunocompetent child. Pediatr Neurol. 2010;43:53–6.
Janoly-Duménil A, Galambrun C, Basset T, Mialou V, Bertrand Y, Bleyzac N. Human herpes virus-6 encephalitis in a paediatric bone marrow recipient: successful treatment with pharmacokinetic monitoring and high doses of ganciclovir. Bone Marrow Transplant. 2006;38:769–70.
Winston DJ, Ho WG, Bartoni K, Du Mond C, Ebeling DF, Buhles WC, Champlin RE. Ganciclovir prophylaxis of cytomegalovirus infection and disease in allogeneic bone marrow transplant recipients. Results of a placebo-controlled, double-blind trial. Ann Intern Med. 1993;118:179–84.
Long MC, Bidanset DJ, Williams SL, Kushner NL, Kern ER. Determination of antiviral efficacy against lymphotropic herpesviruses utilizing flow cytometry. Antiviral Res. 2003;58:149–57.
Manichanh C, Grenot P, Gautheret-Dejean A, Debré P, Huraux JM, Agut H. Susceptibility of human herpesvirus 6 to antiviral compounds by flow cytometry analysis. Cytometry. 2000;40:135–40.
Agut H, Aubin JT, Huraux JM. Homogeneous susceptibility of distinct human herpesvirus 6 strains to antivirals in vitro. J Infect Dis. 1991;163:1382–3.
Fletcher C, Sawchuk R, Chinnock B, de Miranda P, Balfour HH Jr. Human pharmacokinetics of the antiviral drug DHPG. Clin Pharmacol Ther. 1986;40:281–6.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirabayashi, K., Nakazawa, Y., Katsuyama, Y. et al. Successful ganciclovir therapy in a patient with human herpesvirus-6 encephalitis after unrelated cord blood transplantation: usefulness of longitudinal measurements of viral load in cerebrospinal fluid. Infection 41, 219–223 (2013). https://doi.org/10.1007/s15010-012-0329-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-012-0329-3