Abstract
Background:
Secretome provides promising potential in replacing cell-based therapies in wound repair therapy. This study aimed to systematically review and conduct a meta-analysis on the effectiveness of secretome in promoting wound healing.
Methods:
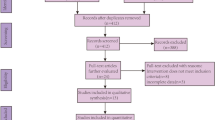
To ensure the rigor and transparency of our study, we followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, as registered in PROSPERO with ID: CRD42023412671. We conducted a comprehensive search on four electronic databases to identify studies evaluating the effect of secretome on various clinical parameters of wound repair. In addition, we evaluated the risk of bias for each study using the Jadad and Newcastle-Ottawa scale. To synthesize the data, we employed a fixed-effects model and calculated the mean difference or odds ratio (OR) with a 95% confidence interval (CI).
Results:
Based on six included articles, secretome is known to affect several clinical parameters in wound healing included the size and depth of ulcers during healing; the E´chelle d’évaluation clinique des cicatrices d’acne (ECCA) score, epidermal thickness, collagen fibers, abnormal elastic tissues, volume of atrophic acne scars, skin pore volume, and erythema during acne scar healing; and microcrust areas, erythema index, transepidermal water loss, volume of atrophic acne scars, erythema, and relative gene expression of procollagen type I, procollagen type III, and elastin were evaluated in wound healing after laser treatment. Meta-analysis studies showed that secretome reduced ulcer size (mean difference: 0.87, 95% CI of 0.37–1.38, p = 0.0007), decreased ulcer depth (mean difference: 0.18, 95% CI of 0.11–0.25, p < 0.00001), and provided patient satisfaction (odds ratio: 9.71, 95% CI of 3.47–21.17, p < 0.0001). However, secretome failed to reach significance in clinical improvement (OR 0.38, 95% CI 0.10, 1.53, p = 0.06).
Conclusion:
The secretome provides good effectiveness in accelerating wound healing through a mechanism that correlates with several clinical parameters of wound repair.
Graphical abstract






Similar content being viewed by others
References
Wound Assessment. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482198/. Accessed 31 Oct 2022.
Afonso AC, Oliveira D, Saavedra MJ, Borges A, Simões M. Biofilms in diabetic foot ulcers: impact, risk factors and control strategies. Int J Mol Sci. 2021;22:8278.
Wound Classification. Available online: https://www.ncbi.nlm.nih.gov/books/NBK554456/. Accessed 31 Oct 2022.
Larouche J, Sheoran S, Maruyama K, Martino MM. Immune regulation of skin wound healing: mechanisms and novel therapeutic targets. Adv Wound Care. 2018;7:209–31.
Raziyeva K, Kim Y, Zharkinbekov Z, Kassymbek K, Jimi S, Saparov A. Immunology of acute and chronic wound healing. Biomolecules. 2021;11:700.
Han G, Ceilley R. Chronic wound healing: a review of current management and treatments. Adv Ther. 2017;34:599–610.
Brumberg V, Astrelina T, Malivanova T, Samoilov A. Modern wound dressings: hydrogel dressings. Biomedicines. 2021;9:1235.
Monika P, Chandraprabha MN, Rangarajan A, Waiker PV, Murthy KNC. Challenges in healing wound: role of complementary and alternative medicine. Front Nutr. 2022;8:791899.
Sood A, Granick MS, Tomaselli NL. Wound dressings and comparative effectiveness data. Adv Wound Care. 2014;3:511–29.
Kanji S, Das H. Advances of stem cell therapeutics in cutaneous wound healing and regeneration. Mediat Inflamm. 2017;2017:5217967.
Hoang DM, Pham PT, Bach TQ, Ngo ATL, Nguyen QT, Phan TTK, et al. Stem cell-based therapy for human diseases. Signal Transduct Target Ther. 2022;7:272.
Oliveira PH, da Silva CL, Cabral JM. Concise review: genomic instability in human stem cells: current status and future challenges. Stem Cells. 2014;32:2824–32.
Ibrahim R, Mndlovu H, Kumar P, Adeyemi SA, Choonara YE. Cell secretome strategies for controlled drug delivery and wound-healing applications. Polymers. 2022;14:2929.
González-González A, García-Sánchez D, Dotta M, Rodríguez-Rey JC, Pérez-Campo FM. Mesenchymal stem cells secretome: the cornerstone of cell-free regenerative medicine. World J Stem Cells. 2020;12:1529–52.
Vizoso FJ, Eiro N, Cid S, Schneider J, Perez-Fernandez R. Mesenchymal stem cell secretome: toward cell-free therapeutic strategies in regenerative medicine. Int J Mol Sci. 2017;18:1852.
Ahangar P, Mills SJ, Cowin AJ. Mesenchymal stem cell secretome as an emerging cell-free alternative for improving wound repair. Int J Mol Sci. 2020;21:7038.
Pokrovskaya LA, Zubareva EV, Nadezhdin SV, Lysenko AS, Litovkina TL. Biological activity of mesenchymal stem cells secretome as a basis for cell-free therapeutic approach. Res Results Pharmacol. 2020;6:57–68.
Guo X, Schaudinn C, Blume-Peytavi U, Vogt A, Rancan F. Effects of adipose-derived stem cells and their conditioned medium in a human ex vivo wound model. Cells. 2022;11:1198.
Bartaula-Brevik S, Bolstad A, Mustafa K, Pedersen TO. Secretome of mesenchymal stem cells grown in hypoxia accelerates wound healing and vessel formation in vitro. Int J Stem Cell Res Ther. 2017;4. https://doi.org/10.23937/2469-570X/1410045
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg. 2021;88:105906.
Olivo SA, Macedo LG, Gadotti IC, Fuentes J, Stanton T, Magee DJ. Scales to assess the quality of randomized controlled trials: a systematic review. Phys Ther. 2008;88:156–75.
Lo CK, Mertz D, Loeb M. Newcastle-Ottawa scale: comparing reviewers’ to authors’ assessments. BMC Med Res Methodol. 2014;14:45.
Oremus M, Wolfson C, Perrault A, Demers L, Momoli F, Moride Y. Interrater reliability of the modified Jadad quality scale for systematic reviews of Alzheimer’s disease drug trials. Dement Geriatr Cogn Disord. 2001;12:232–6.
Margulis AV, Pladevall M, Riera-Guardia N, Varas-Lorenzo C, Hazell L, Berkman N, et al. Quality assessment of observational studies in a drug-safety systematic review, comparison of two tools: the Newcastle-Ottawa scale and the RTI item bank. Clin Epidemiol. 2014;6:359–68.
The Nordic Cochrane Centre. Review Manager, Version 5.4; The Cochrane Collaboration: Copenhagen, Denmark, 2020.
Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane handbook for systematic reviews of interventions. Wiley; 2019.
Alinda MD, Christopher PM, Listiawan MY, Endaryanto A, Suroto H, Rantam FA, et al. Comparative efficacy of topical adipocyte-derived mesenchymal stem cells-conditioned medium (Admsc-cm) and amniotic membrane mesenchymal stem cells-conditioned medium (amsc-cm) on chronic plantar ulcers in leprosy: a randomized controlled trial. Bali Med J. 2021;10:958–63.
Kim J, Kim B, Kim S, Lee YI, Kim J, Lee JH. The effect of human umbilical cord blood-derived mesenchymal stem cell media containing serum on recovery after laser treatment: a double-blinded, randomized, split-face controlled study. J Cosmet Dermatol. 2020;19:651–6.
Abdel-Maguid EM, Awad SM, Hassan YS, El-Mokhtar MA, El-Deek HE, Mekkawy MM. Efficacy of stem cell-conditioned medium vs. platelet-rich plasma as an adjuvant to ablative fractional CO2 laser resurfacing for atrophic post-acne scars: a split-face clinical trial. J Dermatol Treat. 2021;32:242–9.
El-Domyati M, Moftah NH, Nasif GA, Ragaie MH, Ibrahim MR, Ameen SW. Amniotic fluid-derived mesenchymal stem cell products combined with microneedling for acne scars: a split-face clinical, histological, and histometric study. J Cosmet Dermatol. 2019;18:1300–6
Park CS, Park JH, Kim CR, Lee JH. Objective analysis of volume restoration in atrophic acne scars and skin pores: a split study using human stem cell-conditioned media. J Dermatol Treat. 2021;32:73–7.
Zhou BR, Xu Y, Guo SL, Xu Y, Wang Y, Zhu F, et al. The effect of conditioned media of adipose-derived stem cells on wound healing after ablative fractional carbon dioxide laser resurfacing. Biomed Res Int. 2013;2013:519126.
Fife D. Practical evaluation and management of atrophic acne scars: tips for the general dermatologist. J Clin Aesthet Dermatol. 2011;4:50–7.
Gozali MV, Zhou B. Effective treatments of atrophic acne scars. J Clin Aesthet Dermatol. 2015;8:33–40.
Marshall CD, Hu MS, Leavitt T, Barnes LA, Lorenz HP, Longaker MT. Cutaneous scarring: basic science, current treatments, and future directions. Adv Wound Care. 2018;7:29–45.
Chilicka K, Rusztowicz M, Szyguła R, Nowicka D. Methods for the Improvement of acne scars used in dermatology and cosmetology: a review. J Clin Med. 2022;11:2744.
Fan H, Zhang JW, Liu DJ, Liu FB. Transepidermal water loss of scar skin in three types of scar patients and its correlation with scar severity. Zhonghua Shao Shang Za Zhi. 2021;37:629–34.
Acknowledgements
The authors would like to express their gratitude to the Rector of Universitas Padjadjaran for providing funding for this study. Additionally, they would like to extend their appreciation to the Deanship of Scientific Research at Northern Border University in Arar, Kingdom of Saudi Arabia, for generously funding this research through project number “NBU-FFR-2023-0065.”
Author information
Authors and Affiliations
Contributions
CS, NW, AFAM and AER participated in performing research and data analysis, CS initially written the original draft, CS, NW, AFAM and AER also participated in reviewing and editing final manuscript, GW participated in supervising and taking final decision of issues.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest in this manuscript.
Ethical statement
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suhandi, C., Mohammed, A.F.A., Wilar, G. et al. Effectiveness of Mesenchymal Stem Cell Secretome on Wound Healing: A Systematic Review and Meta-analysis. Tissue Eng Regen Med 20, 1053–1062 (2023). https://doi.org/10.1007/s13770-023-00570-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-023-00570-9