Abstract
Background:
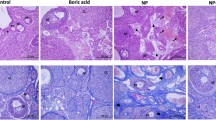
Cryopreservation can cause mechanical and chemical stress, ultimately leading to the formation of reactive oxygen species (ROS) and oxidative stress. ROS inhibits the expression of antioxidant enzymes in cells, resulting in increased DNA fragmentation and apoptosis. In this paper, we used a vitrification method that has the advantage of producing less ice crystal formation, cost-effectiveness, and time efficiency during cryopreservation. The objective of this paper is to evaluate the degree of protection of ovarian tissue against oxidative stress when N-acetylcysteine (NAC) and Klotho proteins are treated in the vitrification process of ovarian tissue.
Methods:
The control group and the cryopreservation groups were randomly assigned, and treated NAC, Klotho, or the combination (NAC + Klotho). The cell morphological change, DNA damage, senescence, and apoptosis of each group after the freeze–thaw process were compared using transmission electron microscopy, immunohistochemistry, and western blot analysis.
Results:
Both NAC and Klotho were found to be more effective at protecting against DNA damage than the control; however, DNA damage was greater in the NAC + Klotho group than in the group treated with NAC and Klotho, respectively. DNA damage and cellular senescence were also reduced during the vitrification process when cells were treated with NAC, Klotho, or the combination (NAC + Klotho). NAC increased apoptosis during cryopreservation, whereas Klotho inhibited apoptosis and NAC-induced apoptosis.
Conclusion:
This study highlights Klotho's benefits in inhibiting DNA damage, cell senescence, and apoptosis, including NAC-induced apoptosis, despite its unclear role in vitrification.





Similar content being viewed by others
References
Adolescents and Young Adults (AYAs) with Cancer - National Cancer Institute. 2015. https://www.cancer.gov/types/aya. Accessed 24 March 2022.
Infertility. - World Health Organization. https://www.who.int/health-topics/infertility#tab=tab_1. Accessed 24 March 2022.
Leonel ECR, Lucci CM, Amorim CA. Cryopreservation of human ovarian tissue: a review. Transfus Med Hemother. 2019;46:173–81.
Sun TC, Liu XC, Yang SH, Song LL, Zhou SJ, Deng SL et al. Melatonin inhibits oxidative stress and apoptosis in cryopreserved ovarian tissues via Nrf2/HO-1 Signaling pathway. Front Mol Biosci. 2020;7:163.
Kerksick C, Willoughby D. The antioxidant role of glutathione and N-Acetyl- Cysteine supplements and exercise-Induced oxidative stress. J Int Soc Sports Nutr. 2005;2:38–44.
Kuro-o M. The Klotho proteins in health and disease. Nat Rev Nephrol. 2019;15:27–44.
Kim JH, Hwang KH, Park KS, Kong ID, Cha SK. Biological role of anti-aging protein Klotho. J Lifestyle Med. 2015;5:1–6.
Lee S, Ryu KJ, Kim B, Kang D, Kim YY, Kim T. Comparison between slow freezing and vitrification for human ovarian tissue cryopreservation and xenotransplantation. Int J Mol Sci. 2019;20:3346.
Brockbank KGM, Chen Z, Greene ED, Campbell LH. Ice-free cryopreservation by vitrification. MOJ Cell Sci Rep. 2014;1:00007.
Ock SA, Rho GJ. Effect of dimethyl sulfoxide (DMSO) on cryopreservation of porcine mesenchymal stem cells (pMSCs). Cell Transplant. 2011;20:1231–9.
Rogers SC, Dosier LB, McMahon TJ, Zhu H, Timm D, Zhang H, et al. Red blood cell phenotype fidelity following glycerol cryopreservation optimized for research purposes. PLoS One. 2018;13:e0209201.
Xiang H, Yang X, Ke L, Hu Y. The properties, biotechnologies, and applications of antifreeze proteins. Int J Biol Macromol. 2020;153:661–75.
Toker MB, Alcay S, Gokce E, Ustuner B. Cryopreservation of ram semen with antioxidant supplemented soybean lecithin-based extenders and impacts on incubation resilience. Cryobiology. 2016;72:205–9.
Ren F, Feng T, Dai G, Wang Y, Zhu H, Hu J. Lycopene and alpha-lipoic acid improve semen antioxidant enzymes activity and cashmere goat sperm function after cryopreservation. Cryobiology. 2018;84:27–32.
Khor SP, Yeow LC, Poobathy R, Zakaria R, Chew BL, Subramaniam S. Droplet-vitrification of Aranda Broga blue orchid: role of ascorbic acid on the antioxidant system and genetic fidelity assessments via RAPD and SCoT markers. Biotechnol Rep. 2020;26:e00448.
Banihani SA, Alawneh RF. Human semen samples with high antioxidant reservoir may exhibit lower post-cryopreservation recovery of sperm motility. Biomolecules. 2019;9:E111.
Yang Q, Hergenhahn M, Weninger A, Bartsch H. Cigarette smoke induces direct DNA damage in the human B-lymphoid cell line Raji. Carcinogenesis. 1999;20:1769–75.
Yedjou CG, Tchounwou CK, Haile S, Edwards F, Tchounwou PB. N-Acetyl-cysteine protects against DNA damage associated with lead toxicity in HepG2 Cells. Ethn Dis. 2010;20:S1-101-3.
Pollman MJ, Hall JL, Gibbons GH. Determinants of vascular smooth muscle cell apoptosis after balloon angioplasty injury. Circ Res. 1999;84:113–21.
Arav A. Cryopreservation by directional freezing and vitrification focusing on large tissues and organs. Cells. 2022;11:1072.
Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35:495–516.
Lee S, Cho H-W, Kim B, Lee JK, Kim T. The effectiveness of anti-apoptotic agents to preserve primordial follicles and prevent tissue damage during ovarian tissue cryopreservation and xenotransplantation. Int J Mol Sci. 2021;22:2534.
Erkkila K, Hirvonen V, Wuokko E, Parvinen M, Dunkel L. N-Acetyl-L-cysteine inhibits apoptosis in human male germ cells in vitro. J Clin Endocrinol Metab. 1998;83:2523–31.
Liu Y, Liu K, Wang N, Zhang H. N-acetylcysteine induces apoptosis via the mitochondria-dependent pathway but not via endoplasmic reticulum stress in H9c2 cells. Mol Med Rep. 2017;16:6626–33.
Kim B, Ryu KJ, Lee S, Kim T. Changes in telomere length and senescence markers during human ovarian tissue cryopreservation. Sci Rep. 2021;11:2238.
Xiao N, Zhang Y, Zheng Q, Gu J. Klotho is a serum factor related to human aging. Chin Med J. 2004;117:742–7.
Arking DE, Atzmon G, Arking A, Barzilai N, Dietz HC. Association between a functional variant of the Klotho gene and high-density lipoprotein cholesterol, blood pressure, stroke, and longevity. Circ Res. 2005;96:412–8.
Arking DE, Becker DM, Yanek LR, Fallin D, Judge DP, Moy TF, et al. Klotho allele status and the risk of early-onset occult coronary artery disease. Am J Hum Genet. 2003;72:1154–61.
Ullah M, Sun Z. klotho deficiency accelerates stem cells aging by impairing telomerase activity. J Gerontol Series A. 2019;74:1396–407.
Ravikumar P, Ye J, Zhang J, Pinch SN, Hu MC, Kuro-o M, et al. α-Klotho protects against oxidative damage in pulmonary epithelia. Am J Physiol Lung Cell Mol Physiol. 2014;307:L566–75.
Kim B, Yoon H, Kim T, Lee S. Role of Klotho as a modulator of oxidative stress associated with ovarian tissue cryopreservation. Int J Mol Sci. 2021;22:13547.
Acknowledgements
This research was funded by the National Research Foundation of Korea Grant by the Korean Government, grant number NRF-2016R1C1B3015250 (date of approval: 1 March 2016).
Author information
Authors and Affiliations
Contributions
BK designed and performed the experiments. S-ML designed the study and analyzed data with analytic tools. S-JP designed and analyzed data. SL supervised the project, the main conceptual ideas and proof outline, and analyzed data. All authors wrote and revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical statement
The study protocol was approved by the institutional review board of Korea University (Korea-2020-0136). Informed consent was confirmed by the IRB.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, B., Lee, SM., Park, SJ. et al. Role of Klotho and N-acetylcysteine in Oxidative Stress Associated with the Vitrification of Ovarian Tissue Cytoprotective Function of Klotho in Cryopreservation. Tissue Eng Regen Med 20, 637–646 (2023). https://doi.org/10.1007/s13770-023-00556-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-023-00556-7