Abstract
Background:
Verapamil is used in the treatment of hypertension, angina pectoris, cardiac arrhythmia, hypertrophic scars, and keloids to block transmembrane calcium ion flux. Verapamil has antioxidant activity, which enhances the production of nitric oxide (NO). NO promotes the proliferation of fibroblasts, keratinocytes, endothelial cells, and epithelial cells during wound healing. In this study, we investigated the effect of verapamil and its antioxidant properties on the enhancement of acute wound healing via NO.
Methods:
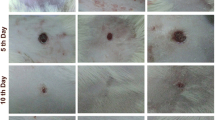
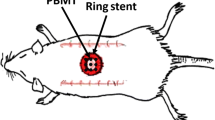
A full-thickness wound healing model was created on the rat dorsal with a silicone ring. The wound closure rate was estimated every 2 days for 14 days. A histological study was performed to evaluate wound healing. Immunofluorescence staining was analyzed for angiogenesis. The expressions of collagen type I (COL I), collagen type III (COL III), and vascular endothelial growth factor (VEGF) were assessed by Western blot. Real-time polymerase chain reaction (qRT-PCR) was performed to examine the expression of endothelial NO synthase and inducible NO synthase, which are related to antioxidant activity in the process of wound healing.
Results:
The wound closure rate was faster in the verapamil group compared to the control and silicone groups. Histologic analysis revealed capillaries and stratum basale in the verapamil group. Immunofluorescence staining was shown vessel formation in the verapamil group. Western blot and qRT-PCR analysis revealed high expression levels of COL I, VEGF, eNOS, and FGF in the verapamil.
Conclusion:
Verapamil’s antioxidant activity enhances NO production in acute wound healing. We suggest that verapamil can be used to promote acute wound healing.






Similar content being viewed by others
References
Elliott WJ, Ram CV. Calcium channel blockers. J Clin Hypertens (Greenwich). 2011;13:687–9.
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, et al. Seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension. 2003;42:1206–52.
Joshi N, Araque H. Blockade of calcium channels by electroacupuncture for hypertension treatment. Med Acupunct. 2010;22:243–7.
Doong H, Dissanayake S, Gowrishankar TR, LaBarbera MC, Lee RC. The 1996 Lindberg Award. Calcium antagonists alter cell shape and induce procollagenase synthesis in keloid and normal human dermal fibroblasts. J Burn Care Rehabil. 1996;17:497–514.
Lee RC, Doong H, Jellema AF. The response of burn scars to intralesional verapamil. Rep Five Cases Arch Surg. 1994;129:107–11.
Lee RC, Ping JA. Calcium antagonists retard extracellular matrix production in connective tissue equivalent. J Surg Res. 1990;49:463–6.
Srivastava S, Kumari H, Singh A. Comparison of fractional co2 laser, verapamil, and triamcinolone for the treatment of keloid. Adv Wound Care (New Rochelle). 2019;8:7–13.
Trisliana Perdanasari A, Lazzeri D, Su W, Xi W, Zheng Z, Ke L, et al. Recent developments in the use of intralesional injections keloid treatment. Arch Plast Surg. 2014;41:620–9.
Kim YO, Kim SM, Jo NH, Nam TS. The effect of simultaneous steroid and verapamil injection on scar formation in the incisional wound of rat. J Korean Soc Plast Reconstr Surg. 2004;31:375–84.
Mojiri-Forushani H. The role of calcium channel blockers in wound healing. Iran J Basic Med Sci. 2018;21:1198–9.
Bagheri M, Jahromi BM, Mirkhani H, Solhjou Z, Noorafshan A, Zamani A, et al. Azelnidipine, a new calcium channel blocker, promotes skin wound healing in diabetic rats. J Surg Res. 2011;169:e101–7.
Ashkani-Esfahani S, Hosseinabadi OK, Moezzi P, Moafpourian Y, Kardeh S, Rafiee S, et al. Verapamil, a calcium-channel blocker, improves the wound healing process in rats with excisional full-thickness skin wounds based on stereological parameters. Adv Skin Wound Care. 2016;29:271–4.
Hattori Y, Kasai K, So S, Hattori S, Banba N, Shimoda S. Effects of calcium channel antagonists on the induction of nitric oxide synthase in cultured cells by immunostimulants. Life Sci. 1995;57:1833–40.
Ma J, Kishida S, Wang GQ, Meguro K, Imuta H, Oonuma H, et al. Comparative effects of azelnidipine and other Ca2+-channel blockers on the induction of inducible nitric oxide synthase in vascular smooth muscle cells. J Cardiovasc Pharmacol. 2006;47:314–21.
Nour S, Baheiraei N, Imani R, Khodaei M, Alizadeh A, Rabiee N, et al. A review of accelerated wound healing approaches: biomaterial-assisted tissue remodeling. J Mater Sci Mater Med. 2019;30:120.
Li J, Chen J, Kirsner R. Pathophysiology of acute wound healing. Clin Dermatol. 2007;25:9–18.
Schwentker A, Vodovotz Y, Weller R, Billiar TR. Nitric oxide and wound repair: role of cytokines? Nitric Oxide. 2002;7:1–10.
Frank S, Kampfer H, Wetzler C, Pfeilschifter J. Nitric oxide drives skin repair: novel functions of an established mediator. Kidney Int. 2002;61:882–8.
Yan X, Zeng B, Chai Y, Luo C, Li X. Improvement of blood flow, expression of nitric oxide, and vascular endothelial growth factor by low-energy shockwave therapy in random-pattern skin flap model. Ann Plast Surg. 2008;61:646–53.
Schäffer MR, Efron PA, Thornton FJ, Klingel K, Gross SS, Barbul A. Nitric oxide, an autocrine regulator of wound fibroblast synthetic function. J Immunol. 1997;158:2375–81.
Lee PC, Salyapongse AN, Bragdon GA, Shears LL 2nd, Watkins SC, Edington HD, et al. Impaired wound healing and angiogenesis in eNOS-deficient mice. Am J Physiol. 1999;277:H1600–8.
Guo S, Dipietro LA. Factors affecting wound healing. J Dent Res. 2010;89:219–29.
Rha EY, Kim YH, Kim TJ, Yoo G, Rhie JW, Kim HJ, et al. Topical application of a silicone gel sheet with verapamil microparticles in a rabbit model of hypertrophic scar. Plast Reconstr Surg. 2016;137:144–51.
Kedziora-Kornatowska K, Szram S, Kornatowski T, Szadujkis-Szadurski L, Kedziora J, Bartosz G. The effect of verapamil on the antioxidant defence system in diabetic kidney. Clin Chim Acta. 2002;322:105–12.
Witte MB, Barbul A. Role of nitric oxide in wound repair. Am J Surg. 2002;183:406–12.
Luo JD, Wang YY, Fu WL, Wu J, Chen AF. Gene therapy of endothelial nitric oxide synthase and manganese superoxide dismutase restores delayed wound healing in type 1 diabetic mice. Circulation. 2004;110:2484–93.
Clugston PA, Vistnes MD, Perry LC, Maxwell GP, Fisher J. Evaluation of silicone-gel sheeting on early wound healing of linear incisions. Ann Plast Surg. 1995;34:12–5.
Docheva D, Müller SA, Majewski M, Evans CH. Biologics for tendon repair. Adv Drug Deliv Rev. 2015;84:222–39.
Schäfer M, Werner S. Oxidative stress in normal and impaired wound repair. Pharmacol Res. 2008;58:165–71.
Gay S, Vijanto J, Raekallio J, Penttinen R. Collagen types in early phases of wound healing in children. Acta Chir Scand. 1978;144:205–11.
Barrientos S, Stojadinovic O, Golinko MS, Brem H, Tomic-Canic M. Growth factors and cytokines in wound healing. Wound Repair Regen. 2008;16:585–601.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (2017M3A9E2060428) and by a Ministry of Trade Industry and Energy of Korea (10062127). This study was supported by Genewel, Seongnam-si, Korea.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest.
Ethical statement
The animal studies were performed after receiving approval of the Institutional Animal Care and Use Committee (IACUC) in Catholic University (IACUC approval No. CUMC-2018–0067-02).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Han, Y.N., Lee, Y.J., Kim, K.J. et al. Nitric Oxide Produced by the Antioxidant Activity of Verapamil Improves the Acute Wound Healing Process. Tissue Eng Regen Med 18, 179–186 (2021). https://doi.org/10.1007/s13770-020-00308-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-020-00308-x