Abstract
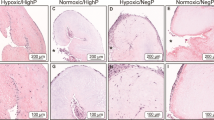
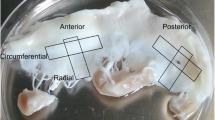
The goal of this study was to test the hypothesis that stem cells, as a response to valve-specific extracellular matrix “niches” and mechanical stimuli, would differentiate into valvular interstitial cells (VICs). Porcine aortic root scaffolds were prepared by decellularization. After verifying that roots exhibited adequate hemodynamics in vitro, we seeded human adipose-derived stem cells (hADSCs) within the interstitium of the cusps and subjected the valves to in vitro pulsatile bioreactor testing in pulmonary pressures and flow conditions. As controls we incubated cell-seeded valves in a rotator device which allowed fluid to flow through the valves ensuring gas and nutrient exchange without subjecting the cusps to significant stress. After 24 days of conditioning, valves were analyzed for cell phenotype using immunohistochemistry for vimentin, alpha-smooth muscle cell actin (SMA) and prolyl-hydroxylase (PHA). Fresh native valves were used as immunohistochemistry controls. Analysis of bioreactor-conditioned valves showed that almost all seeded cells had died and large islands of cell debris were found within each cusp. Remnants of cells were positive for vimentin. Cell seeded controls, which were only rotated slowly to ensure gas and nutrient exchange, maintained about 50% of cells alive; these cells were positive for vimentin and negative for alpha-SMA and PHA, similar to native VICs. These results highlight for the first time the extreme vulnerability of hADSCs to valve-specific mechanical forces and also suggest that careful, progressive mechanical adaptation to valve-specific forces might encourage stem cell differentiation towards the VIC phenotype.
Similar content being viewed by others
References
Wirrig EE, Yutzey KE. Conserved transcriptional regulatory mechanisms in aortic valve development and disease. Arterioscler Thromb Vasc Biol 2014;34:737–741.
Mathieu P, Boulanger MC. Basic mechanisms of calcific aortic valve disease. Can J Cardiol 2014;30:982–993.
Weiss RM, Miller JD, Heistad DD. Fibrocalcific aortic valve disease:opportunity to understand disease mechanisms using mouse models. Circ Res 2013;113:209–222.
Towler DA. Molecular and cellular aspects of calcific aortic valve disease. Circ Res 2013;113:198–208.
Akerström F, Barderas MG, Rodríguez-Padial L. Aortic stenosis:a general overview of clinical, pathophysiological and therapeutic aspects. Expert Rev Cardiovasc Ther 2013;11:239–250.
Alexopoulos A, Kaoukis A, Papadaki H, Pyrgakis V. Pathophysiologic mechanisms of calcific aortic stenosis. Ther Adv Cardiovasc Dis 2012;6:71–80.
Ladich E, Nakano M, Carter-Monroe N, Virmani R. Pathology of calcific aortic stenosis. Future Cardiol 2011;7:629–642.
Chester AH, El-Hamamsy I, Butcher JT, Latif N, Bertazzo S, Yacoub MH. The living aortic valve:from molecules to function. Glob Cardiol Sci Pract 2014;2014:52–77.
Simionescu DT, Chen J, Jaeggli M, Wang B, Liao J. Form follows function:advances in trilayered structure replication for aortic heart valve tissue engineering. J Healthc Eng 2012;3:179–202.
Tedder ME, Simionescu A, Chen J, Liao J, Simionescu DT. Assembly and testing of stem cell-seeded layered collagen constructs for heart valve tissue engineering. Tissue Eng Part A 2011;17:25–36.
Schoen FJ. Heart valve tissue engineering:quo vadis? Curr Opin Biotechnol 2011;22:698–705.
Voges I, Bräsen JH, Entenmann A, Scheid M, Scheewe J, Fischer G, et al. Adverse results of a decellularized tissue-engineered pulmonary valve in humans assessed with magnetic resonance imaging. Eur J Cardiothorac Surg 2013;44:e272–e279.
Isenburg JC, Simionescu DT, Starcher BC, Vyavahare NR. Elastin stabilization for treatment of abdominal aortic aneurysms. Circulation 2007;115:1729–1737.
Chow JP, Simionescu DT, Warner H, Wang B, Patnaik SS, Liao J, et al. Mitigation of diabetes-related complications in implanted collagen and elastin scaffolds using matrix-binding polyphenol. Biomaterials 2013;34:685–695.
Pennel T, Fercana G, Bezuidenhout D, Simionescu A, Chuang TH, Zilla P, et al. The performance of cross-linked acellular arterial scaffolds as vascular grafts;pre-clinical testing in direct and isolation loop circulatory models. Biomaterials 2014;35:6311–6322.
Colazzo F, Alrashed F, Saratchandra P, Carubelli I, Chester AH, Yacoub MH, et al. Shear stress and VEGF enhance endothelial differentiation of human adipose-derived stem cells. Growth Factors 2014;32:139–149.
Sierad LN, Simionescu A, Albers C, Chen J, Maivelett J, Tedder ME, et al. Design and testing of a pulsatile conditioning system for dynamic endothelialization of polyphenol-stabilized tissue engineered heart valves. Cardiovasc Eng Technol 2010;1:138–153.
Sierad LN, Shaw EL, Launius R, McBride S, Storholt C, Poole R, et al. Toward an endothelial-cell covered mechanical valve;surface re-engineering and bioreactor testing of mechanical heart valves. Chall Regen Med 2014;1:22–34.
Londono R, Badylak SF. Biologic scaffolds for regenerative medicine:mechanisms of in vivo remodeling. Ann Biomed Eng 2015;43:577–592.
Keane TJ, Badylak SF. The host response to allogeneic and xenogeneic biological scaffold materials. J Tissue Eng Regen Med 2015;9:504–511.
Badylak SF. Decellularized allogeneic and xenogeneic tissue as a bioscaffold for regenerative medicine:factors that influence the host response. Ann Biomed Eng 2014;42:1517–1527.
Tedder ME, Liao J, Weed B, Stabler C, Zhang H, Simionescu A, et al. Stabilized collagen scaffolds for heart valve tissue engineering. Tissue Eng Part A 2009;15:1257–1268.
Chuang TH, Stabler C, Simionescu A, Simionescu DT. Polyphenol-stabilized tubular elastin scaffolds for tissue engineered vascular grafts. Tissue Eng Part A 2009;15:2837–2851.
Colazzo F, Chester AH, Taylor PM, Yacoub MH. Induction of mesenchymal to endothelial transformation of adipose-derived stem cells. J Heart Valve Dis 2010;19:736–744.
Schoen FJ, Hobson CE. Anatomic analysis of removed prosthetic heart valves:causes of failure of 33 mechanical valves and 58 bioprostheses, 1980 to 1983. Hum Pathol 1985;16:549–559.
Schoen FJ. Pathologic findings in explanted clinical bioprosthetic valves fabricated from photooxidized bovine pericardium. J Heart Valve Dis 1998;7:174–179.
Rajamannan NM. The role of Lrp5/6 in cardiac valve disease:LDL-density-pressure theory. J Cell Biochem 2011;112:2222–2229.
Rajamannan NM. Bicuspid aortic valve disease:the role of oxidative stress in Lrp5 bone formation. Cardiovasc Pathol 2011;20:168–176.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kennamer, A., Sierad, L., Pascal, R. et al. Bioreactor conditioning of valve scaffolds seeded internally with adult stem cells. Tissue Eng Regen Med 13, 507–515 (2016). https://doi.org/10.1007/s13770-016-9114-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-016-9114-1