Abstract
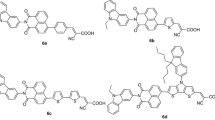
A series of novel push–pull-based D–π–A-containing organic dyes have been synthesized using triphenylamine and carbazole act as π-conjugated donor and cyanoacrylic acid acceptor as well as anchoring group for application of dye-sensitized solar cells. These organic dyes D1, D2 and D3 exhibited excellent photophysical, electrochemical properties, superior stoke shift, high thermal stabilities and sufficient HOMO–LUMO energy levels, which is facilitated to dye regeneration and effective injection of electron from the excited state of the dyes into the conduction band (TiO2). Our results suggested that carbazole π-bridge has an important role in the photovoltaic performance. The D1 and D2 dyes are planar linear structure, longer π-conjugated bridging units, when compared to the starburst structure of D3 organic dye. Moreover, the carbazole π-bridged conjugated D1 dye showed promising photovoltaic conversion efficiency (η) of 1.4%, Voc = 735 mV, Jsc = 2.7 mA/cm2, ff = 0.73 (fill factor), when compared with the conventional N719 and D5 organic dyes.
Graphical Abstract














Similar content being viewed by others

References
B.O. Regan, M. Gratzel, Nature 353, 737 (1991)
P. Wang, C. Klein, T.H. Baker, S.M. Zakeeruddin, M. Gratzel, J. Am. Chem. Soc. 127, 808 (2005)
K.A. Kakiage, Y. Yano, T. Oya, K. Fujisawa, J.I. Hanaya, J.J.H.M. Bearpark, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, Chem. Commun. 51, 15894 (2015)
R. Yan, X. Qian, Y. Jiang, Y. He, Y. Hang, L. Hou, (2017). https://doi.org/10.1016/j.dyepig.2017.02.011
J. Pan, H. Song, C. Lian, H. Liu, Y. Xie, Dyes and Pigments (2017). https://doi.org/10.1016/j.dyepig.2017.01.027
W.J. Fan, Y.Z. Chang, J.L. Zhao, Z.N. Xu, D.Z. Tan, Y.G. Chend, New J. Chem. 42, 20163 (2018)
Y. Mu, H. Wu, G. Dong, Z. Shen, S. Li, M. Zhang, J. Mater. Chem. A 6, 21493 (2018)
P. Naik, I.M. Abdellah, M.A. Shakour, M. Acharaya, N. Pilicode, A.E. Shafei, A. Vasudeva Adhikari, Chem. Sel. 3, 12297 (2018)
F. Bellaa, A. Verna, C. Gerbaldi, Materials Science Semiconductor Processing (2017). https://doi.org/10.1016/j.mssp.2017.07.030
A. Carella, R. Centore, F. Borbone, M. Toscanesi, M. Trifuoggi, F. Bella, C. Gerbaldi, S. Galliano, E. Schiavo, A. Massaro, A.B. Munoz-Garcia, M. Pavonea, Electrochimica Acta. https://doi.org/10.1016/j.electacta.2018.09.204
S. Galliano, F. Bella, G. Piana, G. Giacona, G. Viscardi, C. Gerbaldi, M. Grätzel, C. Barolo, Sol. Energy 163, 251 (2018)
A. Scalia, F. Bella, A. Lamberti, C. Gerbaldi, E. Tresso, Energy 166, 789 (2019)
F. Bella, J. Popovic, A. Lamberti, E. Tresso, C. Gerbaldi, J. Maier, A.C.S. Appl, Mater. Interfaces 9, 37797 (2017)
K.M. Manikandan, A. Yelilarasi, P. Senthamaraikannan, S.S. Saravanakumar, A. Khan, A.M. Asiri, J. Solid State Electrochem. 22, 3785 (2018)
V. Ganapathy, B. Karunagaran, S.W. Rhee, J. Power Sour. 195, 5138 (2010)
A. Hagfeldt, G. Boschloo, L. Sun, L. Kloo, H. Pettersson, Chem. Rev. 110, 6595 (2010)
V. Karthikeyan, S. Maniarasu, V. Manjunath, E. Ramasamy, G. Veerappan, Sol. Energy 147, 202 (2017)
G. Veerappan, D.W. Jung, J. Kwon, J.M. Choi, N. Heo, G.R. Yi, J.H. Park, Langmuir 30, 3010 (2014)
R. Sivakumar, R. Recabarren, S. Ramkumar, A. Manivel, J.A. Morales, D. Contreras, P. Manidurai, New J. Chem. 41, 5605 (2017)
X. Li, J. Gui, H. Yang, W. Wu, F. Li, H. Tian, C. Huang, Inorg. Chim. Acta 361, 2835 (2008)
H. Tian, X. Yang, R. Chen, Y. Pan, L. Li, A. Hagfeldt, L. Sun, Chem. Commun. 36, 3741 (2007)
Y.S. Yen, H.H. Chou, Y.C. Chen, C.Y. Hsu, J.T. Lin, J. Mater. Chem. 22, 8734 (2012)
H. Masui, M.M. Maitani, S. Fuse, A. Yamamura, Y. Ogomi, S. Hayase, T. Kaiho, H. Tanaka, Y. Wada, T. Takahashi, Asian J. Org. Chem. 7, 458 (2018)
T.Y. Wu, M.H. Tsao, F.L. Chen, S.G. Su, C.W. Chang, H.P. Wang, Y.C. Lin, I.W. Sun, J. Iran. Chem. Soc. 7, 707 (2010)
F. Zanjanchi, J. Beheshtian, J. Iran. Chem. Soc. (2018). https://doi.org/10.1007/s13738-018-1561-2
K. Sayama, K. Hara, N. Mori, M. Satsuki, S. Suga, S. Tsukagoshi, Y. Abe, H. Sugihara, H. Arakawa, Chem. Commun. 0, 1173 (2000)
C. Chen, J.Y. Liao, Z. Chi, B. Xu, X. Zhang, D.B. Kuang, Y. Zhang, S. Liu, J. Xu, J. Mater. Chem. 22, 8994 (2012)
W. Li, Y. Wu, X. Li, Y. Xe, W. Zhu, Energy Environ. Sci. 4, 1830 (2011)
D.Y. Chen, Y.Y. Hsu, H.C. Hsu, B.S. Chen, Y.T. Lee, H. Fu, M.W. Chung, S.H. Liu, H.C. Chen, Y. Chi, P.T. Chou, Chem. Commun. 46, 5256 (2010)
H. Zhou, P. Xue, Y. Zhang, X. Zhao, J. Jia, X. Zhang, X. Lu, R. Lu, Tetrahedron 67, 8477 (2011)
B. Nagarajan, S. Kushwaha, R. Elumalai, S. Mandal, K. Ramanujam, D. Raghavachari, J. Mater. Chem. A. 5, 10289 (2017)
K.B. Fadadu, S.S. Soni, Electrochim. Acta 88, 270 (2013)
X. Ma, J. Hua, W. Wu, Y. Jin, F. Meng, W. Zhan, H. Tian, Tetrahedron 64, 345 (2008)
K.D. Seo, I.T. Choi, Y.G. Park, S. Kang, J.Y. Lee, H.K. Kim, Dyes Pigm. 94, 469 (2012)
K. Hara, Z.S. Wang, T. Sato, A. Furube, R. Katoh, H. Sugihara, Y.D. Oh, C. Kasada, A. Shinpo, S. Suga, J. Phys. Chem. B. 109, 15476 (2005)
Z. Shen, J. Chen, X. Li, X. Li, Y. Zhou, Y. Yu, H. Ding, J. Li, L. Zhu, J. Hua, ACS Sustinable Chem. Eng. 4, 3518 (2016)
X. Liu, Z. Cao, H. Huang, X. Liu, Y. Tan, H. Chen, Y. Pei, S. Tan, J. Power Sour. 248, 400 (2014)
X.F. Wang, H. Tamiaki, O. Kitao, T. Ikeuchi, S.I. Sasaki, J. Power, Sources 242, 860 (2013)
J. Chung, C.P. Lee, D. Kumar, P.W. Chen, L.Y. Lin, K.R.J. Thomas, K.C. Ho, J. Power Sour. 240, 779 (2013)
D. Pugliese, A. Lamberti, F. Bella, A. Sacco, S. Bianco, E. Tresso, Org. Electron. 15, 3715 (2014)
T. Horiuchi, H. Miura, K. Sumioka, S. Uchida, J. Am. Chem. Soc. 126, 12218 (2004)
V.A. Chiykowski, B. Lam, C. Du, C.P. Berlinguette, Chem. Commun. 53, 2367 (2017)
G. Zhang, H. Bala, Y. Cheng, D. Shi, X. Lv, Q. Yu, P. Wang, Chem. Commun. 16, 2198 (2009)
T.B. Raju, J.V. Vaghasiya, M.A. Afroz, S.S. Soni, P. Krishnan Iyer, Phys. Chem. Chem. Phys. 18, 28485 (2016)
A. Karuppasamy, K. Stalindurai, J. Peng, K.C. Ho, C. Ramalingan, Phys. Chem. Chem. Phys. 18, 30105 (2016)
D. Kumar, K.T. Wong, Mater. Today Energy 5, 243 (2017)
Y. Ooyama, N. Yamaguchi, I. Imae, K. Komaguchi, J. Ohshita, Y. Harima, Chem. Commun. 49, 2548 (2013)
A. Abbotto, N. Manfredi, C. Marinzi, F.D. Angelis, E. Mosconi, J. Yum, Z. Xianxi, M.K. Nazeeruddin, M. Gratzel, Energy Environ. Sci. 2, 1094 (2009)
J. Wang, S. Liu, Z. Chai, K. Chang, M. Fang, M. Han, Y. Wang, S. Li, H. Han, Q. Li, Z. Li, J. Mater. Chem. A 6, 22256 (2018)
G. Prakash, K. Subramanian, New J. Chem. 42, 17939 (2018)
D.P. Hagberg, T. Edvinsson, T. Marinado, G. Boschloo, A. Hagfeldtb, L. Sun, Chem. Commun. 0, 2245 (2006)
M.A. Mamun, Q. Qiao, B.A. Logue, RSC Adv. 8, 31943 (2018)
A. Karuppasamy, K. Stalindurai, J.D. Peng, K.C. Ho, C. Ramalingan, Phys. Chem. Chem. Phys. 18, 30105 (2016)
F. Meng, Y. Liu, X. Yu, W. Lin, New J. Chem. 40, 7399 (2016)
G. Li, Y.F. Zhou, X.B. Cao, P. Bao, K.J. Jiang, Y. Lin, L.M. Yang, Chem. Commun. 0, 2201 (2009)
Z. Ning, Q. Zhang, W. Wu, H. Pei, B. Liu, H. Tian, J. Org. Chem. 73, 3791 (2008)
R. Balasaravanan, V. Sadhasivam, G. Sivaraman, A. Siva, Asian J. Org. Chem. 5, 399 (2016)
R.Y.Y. Lin, H.W. Lin, Y.S. Yen, C.H. Chang, H.H. Chou, P.W. Chen, C.Y. Hsu, Y.C. Chen, J.T. Lin, K.C. Ho, Energy Environ. Sci. 6, 2477 (2013)
Y. Ooyama, K. Furue, T. Enoki, M. Kanda, Y. Adachi, J. Ohshita, Phys. Chem. Chem. Phys. 18, 30662 (2016)
A. Jankovic, Z.V. Saponjic, M.I. Comor, J.M. Nedeljkovic, J. Phys. Chem. C 113, 12645 (2009)
J.V. Vaghasiya, K.K. Sonigara, J. Prasad, T. Beuvier, A. Gibaud, S.S. Soni, J. Mater. Chem. A. 5, 5373 (2017)
B.G. Kim, K.M. Chung, J.S. Kim, Chem. Eur. J. 19, 5220 (2013)
Y.A. Sadiki, S.M. Bouzzine, L. Bejjit, G.S. Moran, M. Hamidi, M. Bouachrine, F. Serein-Spirau, J.P. Lere Porte, J. Marc Sotiropoulos, L.G. Candia, D.G. Mitnik, J. Iran. Chem. Soc. 13, 37 (2016)
A.N. Krol, B. Filmmel, M. Son, D. Kim, F. Warthner, Faraday Discuss. 185, 507 (2015)
P. Arul, S.A. John, Electrochim. Acta 235, 680 (2017)
Acknowledgements
The authors acknowledge the financial support from the Department of Science and Technology, SERB, Extramural Major Research Project (Grant No. EMR/2015/000969), the Department of Science and Technology, CERI, New Delhi, India (Grant No. DST/TM/CERI/C130(G), and the University Grants Commission, New Delhi, India (Grant No. UGC No.41-215/2012 (SR). We acknowledge the DST-FIST, DST-PURSE, UPE programme for providing instrumental support.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Velu, S., Muniyasamy, H., Ayyanar, S. et al. Design, synthesis of organic sensitizers containing carbazole and triphenylamine π-bridged moiety for dye-sensitized solar cells. J IRAN CHEM SOC 16, 1923–1937 (2019). https://doi.org/10.1007/s13738-019-01663-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-019-01663-w