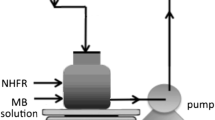
Abstract
Phenolic compounds are priority chemicals which, when discharged in wastewater, need to be removed. Photochemical treatment has been demonstrated to be very effective for this task. This paper reports a study to assess the potential of a nano-photo-Fenton-like reagent (NFLR), based on synthesized haematite (\(\upalpha \)-\(\hbox {Fe}_{2}\hbox {O}_{3})\) nano-crystalline powder, to remove phenol from wastewater. The use of haematite nano-structures as a source of \(\hbox {Fe}^{3+}\) in NFLR provides a novel and low-cost nano-catalysis treatment of phenol wastewater. The study evaluated the effect of the initial concentration of the phenol pollutant (200–1000 mg/L), and the photo-Fenton parameters such as the nano-haematite and \(\hbox {H}_{2}\hbox {O}_{2}\) concentrations (100–800 mg/L) and pH range from 2 to 8. NFLR was able to remove about 98% of phenol in about 20 min from a solution initially containing 400 ppm of phenol. Furthermore, a Box–Behnken factorial design was applied to optimize the operating NFLR parameters. The optimum conditions for phenol removal were shown to be 398 mg/L for \(\hbox {Fe}^{3+}\), 41 mg/L for \(\hbox {H}_{2}\hbox {O}_{2}\) and 3.25 for pH. The degradation obtained using a response surface methodology is compared with that recorded experimentally at the optimized values. The NFLR, based on nano-haematite, was found to be an effective treatment for the removal of phenol from wastewater.
Similar content being viewed by others
References
Gümüş, D.; Akbal, F.: Comparison of Fenton and electro-Fenton processes for oxidation of phenol. Process Saf. Environ. Prot. 103(Part A), 252–258 (2016)
Gad, N.S.; Saad, A.S.: Effect of environmental pollution by phenol on some physiological parameters of oreochromis niloticus. Glob. Vet. 2(6), 312–319 (2008)
Mohapatra, D.P.; Brar, S.K.; Tyagi, R.D.; Surampalli, R.Y.: Physico-chemical pre-treatment and biotransformation of wastewater and wastewater Sludge–Fate of bisphenol A. Chemosphere 78, 923–941 (2010)
Aksu, Z.: Application of biosorption for the removal of organic pollutants: a review. Process Biochem. 40, 997–1026 (2005)
Michałowicz, J.; Duda, W.: Phenols—sources and toxicity. Pol. J. Environ. Stud. 16(3), 347–362 (2007)
Vidic, R.D.; Suidan, M.T.; Brenner, R.C.: Oxidative coupling of phenols on activated carbon: impact on adsorption equilibrium. Environ. Sci. Technol. 27, 2079–2085 (2007)
Dabhade, M.A.; Saidutta, M.B.; Murthy, D.V.R.: Adsorption of phenol on granular activated carbon from nutrient medium: equilibrium and kinetic study. Int. J. Environ. Res. 3(4), 557–568 (2009)
Stavropoulos, G.G.; Samaras, P.; Sakellaropoulos, G.P.: Effect of activated carbons modification on porosity, surface structure and phenol adsorption. J. Hazard. Mater. 151, 414–421 (2008)
Arshad, S.H.; Aziz, A.A.; Ngadi, N.; Amin, N.S.: Phenol adsorption by activated carbon of different fiber size derived from empty fruit bunches. J. Oil Palm Res. 24, 1524–1532 (2012)
Babuponnusamim, A.; Muthukumar, K.: A review on Fenton and improvements to the Fenton process for wastewater treatment. J. Environ. Chem. Eng. 2, 557–572 (2014)
SAS Institute; Inc JMP 8: Statistical Discovery Software. Cary, NC (2009). http://www.jmp.com/
Montgomery, D.C.: Design and Analysis of Experiments. Wiley, New York (1991)
Benatti, C.T.; Tavares, C.R.G.; Guedes, T.A.: Optimization of Fenton’s oxidation of chemical laboratory wastewaters using the response surface methodology. J. Environ. Manag. 80, 66–74 (2006)
Tony, A.; Purcell, P.J.; Zhao, Y.Q.; Tayeb, A.M.; El-Sherbiny, M.F.: Photo-catalytic degradation of an oil–water emulsion using the photo-Fenton treatment process: effects and statistical optimization. J. Environ. Sci. Health A A44(2), 179–187 (2009)
Sathian, S.; Radha, G.; Shanmugapriya, V.; Rajasimman, M.; Karthikeyan, C.: Optimization and kinetic studies on treatment of textile dye wastewater using Pleurotus floridanus. Appl. Water Sci. 3(1), 41–48 (2013)
Suarez-Escobar, A.; Pataquiva-Mateus, A.; Lopez-Vasquez, A.: Electrocoagulation—photocatalytic process for the treatment of lithographic wastewater. Optimization using response surface methodology (RSM) and kinetic study. Catal. Today 266, 120–125 (2016)
Feng, H.; Le-cheng, L.: Degradation kinetics and mechanisms of phenol in photo-Fenton process. J. Zhejiang Univ. 5(2), 198–205 (2004)
Faisal, I.: Oxidation of phenolic wastewater by Fenton’s reagent. Iraqi J. Chem. Pet. Eng. 10(2), 35–42 (2009)
Assadi, A.; Eslami, A.: Comparison of phenol photodegradation by UV/\(\text{ H }_{2}\text{ O }_{2}\) and photo-Fenton processes. Environ. Eng. Manag. J. 9(6), 807–812 (2010)
Minella, M.; Marchetti, G.; De Laurentiisa, E.; Malandrinoa, M.; Maurinoa, V.; Mineroa, C.; Vionea, D.; Hanna, K.: Photo-Fenton oxidation of phenol with magnetite as iron source. Appl. Catal. B Environ. 154–155, 102–109 (2014)
Srodowiska, R.O.: Phenol oxidation in the photo-Fenton process catalyzed by clinoptylolite modified with co. Annu. Set Environ. Prot. 17, 113–124 (2015)
Jiang, C.; Pang, S.; Ouyang, F.; Ma, J.; Jiang, J.: A new insight into Fenton and Fenton-like processes for water treatment. J. Hazard. Mater. 174, 813–817 (2010)
Fang, X.; Chen, C.; Jin, M.; Kuang, Q.; Xie, Z.; Xie, S.; Rong- Huang, B.; Zheng, L.: Single-crystal-like hematite colloidal nanocrystal clusters: synthesis and applications in gas sensors, photocatalysis and water treatment. J. Mater. Chem. 19, 6154–6160 (2009)
Zeng, S.; Tang, K.; Li, T.; Liang, Z.; Wang, D.; Wang, Y.; Zhou, W.: Hematite hollow spindles and microspheres: selective synthesis, growth mechanisms, and application in lithium ion battery and water treatment. J. Phys. Chem. C 111(28), 10217–10225 (2007)
Lin, S.S.; Gurol, M.D.: Catalytic decomposition of hydrogen peroxide on iron oxide: kinetics, mechanism, and implications. Environ. Sci. Technol. 32(10), 1417–1423 (1998)
Lu, M.C.; Chen, J.N.; Huang, H.H.: Role of goethite dissolution in the oxidation of 2-chlorophenol with hydrogen peroxide. Chemosphere 46(1), 131–136 (2002)
Zhang, Y.; Wu, B.; Xu, H.; Liu, H.; Wang, M.; He, Y.; Pan, B.: Nanomaterials-enabled water and wastewater treatment. NanoImpact 3–4, 22–39 (2016)
Najjar, W.; Chirchi, L.; Santosb, E.; Ghorhel, A.: Kinetic study of 2-nitrophenol photodegradation on Al-pillared montmorillonite doped with copper. J. Environ. Monit. 3, 697–701 (2001)
He, F.; Lei, L.: Degradation kinetics and mechanisms of phenol in photo-Fenton process. J. Zhejiang Univ. 5(2), 198–205 (2004)
Wu, J.; Jia, R.; Liu, C.; Wang, H.: Study of degradation of phenol in wastewater sample via advanced oxidation technology. In: International Forum on Energy, Environmental Science and Material (IFEESM), pp. 584–587. Published by Atlantis Press (2015)
Radovic, M.D.; Mitrovic, J.Z.; Kostic, M.M.; Bojic, D.V.; Petrovic, M.M.; Najdanovic, S.M.; Bojicc, A.L.: Fenton and photo-Fenton processes for the decolorization of reactive dyes. Comparison of ultraviolet radiation/hydrogen peroxide. Hem. Ind. J. 69(6), 657–665 (2015)
Weichgrebe, D.; Vogelpohl, A.: A comparative study of wastewater treatment by chemical wet oxidation. Chem. Eng. Process 33(4), 199–203 (1994)
Utset, B.; Garcia, J.; Casado, J.; Domenech, X.; Peral, J.: Replacement of \(\text{ H }_{2}\text{ O }_{2}\) by \(\text{ O }_{2}\) in Fenton and photo-Fenton reactions. Chemosphere 41(8), 1187–1192 (2000)
Martínez, F.; Calleja, G.; Melero, J.A.; Molina, R.: Heterogeneous photo-Fenton degradation of phenolic aqueous solutions over iron-containing SBA-15 catalyst. Appl. Catal. B Environ. 60(3–4), 181–190 (2005)
Wang, N.; Zheng, T.; Zhang, G.; Wang, P.: A review on Fenton-like processes for organic wastewater treatment. J. Environ. Chem. Eng. 4(1), 762–787 (2016)
Sarria, V.; Kenfack, S.; Guillod, O.; Pulgarin, C.: An innovative coupled solar-biological system at field pilot scale for the treatment of biorecalcitrant pollutants. J. Photochem. Photobiol. A 159(1), 89–99 (2003)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tony, M.A., Mansour, S.A., Tayeb, A.M. et al. Use of a Fenton-Like Process Based on Nano-Haematite to Treat Synthetic Wastewater Contaminated by Phenol: Process Investigation and Statistical Optimization. Arab J Sci Eng 43, 2227–2235 (2018). https://doi.org/10.1007/s13369-017-2632-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-017-2632-x