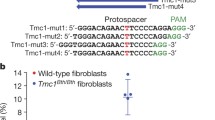
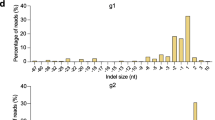
Abstract
Hereditary hearing loss (HHL) is a neurosensory disorder that affects every 1/500 newborns worldwide and nearly 1/3 people over the age of 65. Congenital deafness is inherited as monogenetic or polygenic disorder. The delicacy, tissue heterogeneity, deep location of the inner ear down the brainstem, and minute quantity of cells present in cochlea are the major challenges for current therapeutic approaches to cure deafness. Targeted genome editing is considered a suitable approach to treat HHL since it can target defective molecular components of auditory transduction to restore normal cochlear function. With the advent of CRISPR/Cas9 technique, targeted genome editing and biomedical research have been revolutionized. The robustness and simplicity of this technology lie in its design and delivery methods. It can directly deliver a complex of Cas9 endonuclease and single guide RNA (sgRNA) into zygote using either vector-mediated stable transfection or transient delivery of ribonucleoproteins complexes. This strategy induces DNA double strand breaks (DSBs) at target site followed by endogenous DNA repairing mechanisms of the cell. CRISPR/Cas9 has been successfully used in model animals to edit hearing genes like calcium and integrin-binding protein 2, myosin VIIA, Xin-actin binding repeat containing 2, leucine-zipper and sterile-alpha motif kinase Zak, epiphycan, transmembrane channel–like protein 1, and cadherin 23. This review discusses the utility of lipid-mediated transient delivery of Cas9/sgRNA complexes, an efficient way to restore hearing in humans, suffering from HHL. Notwithstanding, challenges like PAM requirement, HDR efficiency, off-target activity, and optimized delivery systems need to be addressed.




Similar content being viewed by others
References
Adachi N, So S, Iiizumi S et al (2006) The human pre-B cell line Nalm-6 is highly proficient in gene targeting by homologous recombination. DNA Cell Biol 25:19–24. https://doi.org/10.1089/dna.2006.25.19
Adams D, Gonzalez-Duarte A, O’Riordan WD et al (2018) Patisiran, an RNAi therapeutic, for hereditary transthyretin amyloidosis. N Engl J Med 379:11–21. https://doi.org/10.1056/NEJMoa1716153
Ahituv N, Avraham KB (2002) Mouse models for human deafness: current tools for new fashions. Trends Mol Med 8:447–451. https://doi.org/10.1016/S1471-4914(02)02388-2
Akil O, Seal RP, Burke K et al (2012) Restoration of hearing in the VGLUT3 knockout mouse using virally mediated gene therapy. Neuron 75:283–293. https://doi.org/10.1016/j.neuron.2012.05.019
Angeli S, Lin X, Liu XZ (2012) Genetics of hearing and deafness. Anat Rec (Hoboken) 295:1812–1829. https://doi.org/10.1002/ar.22579
Balciuniene J, Dahl N, Borg E et al (1998) Evidence for digenic inheritance of nonsyndromic hereditary hearing loss in a Swedish family. Am J Hum Genet 63:786–793. https://doi.org/10.1086/302012
Barrangou R, Fremaux C, Deveau H et al (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315:1709–1712. https://doi.org/10.1126/science.1138140
Beerli RR, Dreier B, Barbas CF (2000) Positive and negative regulation of endogenous genes by designed transcription factors. Proc Natl Acad Sci U S A 97:1495–1500. https://doi.org/10.1073/pnas.040552697
Bhaya D, Davison M, Barrangou R (2011) CRISPR-Cas systems in bacteria and archaea: versatile small RNAs for adaptive defense and regulation. Annu Rev Genet 45:273–297. https://doi.org/10.1146/annurev-genet-110410-132430
Bolotin A, Quinquis B, Sorokin A et al (2005) Clustered regularly interspaced short palindrome repeats (CRISPRs) have spacers of extrachromosomal origin. Microbiology 151:2551–2561. https://doi.org/10.1099/mic.0.28048-0
Brouns SJ, Jore MM, Lundgren M et al (2008) Small CRISPR RNAs guide antiviral defense in prokaryotes. Science 321:960–964. https://doi.org/10.1126/science.1159689
Capecchi MR (2005) Gene targeting in mice: functional analysis of the mammalian genome for the twenty-first century. Nat Rev Genet 6:507–512. https://doi.org/10.1038/nrg1619
Carroll D (2011) Genome engineering with zinc-finger nucleases. Genetics 188:773–782. https://doi.org/10.1534/genetics.111.131433
Chandrasegaran S, Carroll D (2016) Origins of programmable nucleases for genome engineering. J Mol Biol 428:963–989. https://doi.org/10.1016/j.jmb.2015.10.014
Chen H, Choi J, Bailey S (2014a) Cut site selection by the two nuclease domains of the Cas9 RNA-guided endonuclease. J Biol Chem 289:13284–13294. https://doi.org/10.1074/jbc.M113.539726
Chen J, Chen J, Zhu Y et al (2014b) Deafness induced by connexin 26 (GJB2) deficiency is not determined by endocochlear potential (EP) reduction but is associated with cochlear developmental disorders. Biochem Biophys Res Commun 448:28–32. https://doi.org/10.1016/j.bbrc.2014.04.016
Chen J, Hong F, Zhang C et al (2018) Differentiation and transplantation of human induced pluripotent stem cell-derived otic epithelial progenitors in mouse cochlea. Stem Cell Res Ther 9:230. https://doi.org/10.1186/s13287-018-0967-1
Cheng Y-C, Kuo W-W, Wu H-C et al (2009) ZAK induces MMP-2 activity via JNK/p38 signals and reduces MMP-9 activity by increasing TIMP-1/2 expression in H9c2 cardiomyoblast cells. Mol Cell Biochem 325:69–77. https://doi.org/10.1007/s11010-008-0021-1
Cho SW, Kim S, Kim JM et al (2013) Targeted genome engineering in human cells with the Cas9 RNA-guided endonuclease. Nat Biotechnol 31:230–232. https://doi.org/10.1038/nbt.2507
Chu C, Qu K, Zhong FL et al (2011) Genomic maps of long noncoding RNA occupancy reveal principles of RNA-chromatin interactions. Mol Cell 44:667–678. https://doi.org/10.1016/j.molcel.2011.08.027
Clarke DL, Johansson CB, Wilbertz J et al (2000) Generalized potential of adult neural stem cells. Science 288:1660–1663. https://doi.org/10.1126/science.288.5471.1660
Cong L, Zhou R, Y-c K et al (2012) Comprehensive interrogation of natural TALE DNA-binding modules and transcriptional repressor domains. Nat Commun 3:968. https://doi.org/10.1038/ncomms1962
Cong L, Ran FA, Cox D et al (2013) Multiplex genome engineering using CRISPR/Cas systems. Science. 339:819–823. https://doi.org/10.1126/science.1231143
Deltcheva E, Chylinski K, Sharma CM et al (2011) CRISPR RNA maturation by trans-encoded small RNA and host factor RNase III. Nature 471:602–607. https://doi.org/10.1038/nature09886
DeWitt MA, Corn JE, Carroll D (2017) Genome editing via delivery of Cas9 ribonucleoprotein. Methods 121:9–15. https://doi.org/10.1016/j.ymeth.2017.04.003
Di Domenico M, Ricciardi C, Martone T et al (2011) Towards gene therapy for deafness. J Cell Physiol 226:2494–2499. https://doi.org/10.1002/jcp.22617
Engreitz JM, Pandya-Jones A, McDonel P et al (2013) The Xist lncRNA exploits three-dimensional genome architecture to spread across the X chromosome. Science 341:1237973. https://doi.org/10.1126/science.1237973
Francis SP, Katz J, Fanning KD et al (2013) A novel role of cytosolic protein synthesis inhibition in aminoglycoside ototoxicity. J Neurosci 33:3079–3093. https://doi.org/10.1523/JNEUROSCI.3430-12.2013
Francis SP, Krey JF, Krystofiak ES et al (2015) A short splice form of Xin-actin binding repeat containing 2 (XIRP2) lacking the Xin repeats is required for maintenance of stereocilia morphology and hearing function. J Neurosci 35:1999–2014. https://doi.org/10.1523/JNEUROSCI.3449-14.2015
Friedman TB, Griffith AJ (2003) Human nonsyndromic sensorineural deafness. Annu Rev Genomics Hum Genet 4:341–402. https://doi.org/10.1146/annurev.genom.4.070802.110347
Fu Y, Foden JA, Khayter C et al (2013) High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nat Biotechnol 31:822–826. https://doi.org/10.1038/nbt.2623
Fukushima K, Kasai N, Ueki Y et al (1999) A gene for fluctuating, progressive autosomal dominant nonsyndromic hearing loss, DFNA16, maps to chromosome 2q23-24.3. Am J Hum Genet 65:141–150. https://doi.org/10.1086/302461
Gao X, Tao Y, Lamas V et al (2018) Treatment of autosomal dominant hearing loss by in vivo delivery of genome editing agents. Nature 553:217–221. https://doi.org/10.1038/nature25164
Garneau JE, Dupuis M-È, Villion M et al (2010) The CRISPR/Cas bacterial immune system cleaves bacteriophage and plasmid DNA. Nature 468:67–71. https://doi.org/10.1038/nature09523
Gasiunas G, Barrangou R, Horvath P et al (2012) Cas9–crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proc Natl Acad Sci U S A 109:E2579–E2586. https://doi.org/10.1073/pnas.1208507109
Gentry HR, Singer AU, Betts L et al (2005) Structural and biochemical characterization of CIB1 delineates a new family of EF-hand-containing proteins. J Biol Chem 280:8407–8415. https://doi.org/10.1074/jbc.M411515200
Gonzalez JM, Morgani SM, Bone RA et al (2016) Embryonic stem cell culture conditions support distinct states associated with different developmental stages and potency. Stem Cell Reports 7:177–191. https://doi.org/10.1016/j.stemcr.2016.07.009
Gorlin RJ, Toriello HV, Cohen MM (1995) Hereditary hearing loss and its syndromes, vol 28. Oxford University Press, USA
Grissa I, Vergnaud G, Pourcel C (2007) CRISPRFinder: a web tool to identify clustered regularly interspaced short palindromic repeats. Nucleic Acids Res 35:W52–W57. https://doi.org/10.1093/nar/gkm360
Gross EA, Callow MG, Waldbaum L et al (2002) MRK, a mixed lineage kinase-related molecule that plays a role in γ-radiation-induced cell cycle arrest. J Biol Chem 277:13873–13882. https://doi.org/10.1074/jbc.M111994200
Hale CR, Majumdar S, Elmore J et al (2012) Essential features and rational design of CRISPR RNAs that function with the Cas RAMP module complex to cleave RNAs. Mol Cell 45:292–302. https://doi.org/10.1016/j.molcel.2011.10.023
Hanada Y, Nakamura Y, Ishida Y et al (2017) Epiphycan is specifically expressed in cochlear supporting cells and is necessary for normal hearing. Biochem Biophys Res Commun 492:379–385. https://doi.org/10.1016/j.bbrc.2017.08.092
Harper JW, Elledge SJ (2007) The DNA damage response: ten years after. Mol Cell 28:739–745. https://doi.org/10.1016/j.molcel.2007.11.015
Hasson T, Gillespie PG, Garcia JA et al (1997) Unconventional myosins in inner-ear sensory epithelia. J Cell Biol 137:1287–1307. https://doi.org/10.1083/jcb.137.6.1287
Hilgert N, Smith RJ, Van Camp G (2009) Forty-six genes causing nonsyndromic hearing impairment: which ones should be analyzed in DNA diagnostics? Mutat Res 681:189–196. https://doi.org/10.1016/j.mrrev.2008.08.002
Hsu PD, Scott DA, Weinstein JA et al (2013) DNA targeting specificity of RNA-guided Cas9 nucleases. Nat Biotechnol 31:827–832. https://doi.org/10.1038/nbt.2647
Hu Z, Ulfendahl M (2013) The potential of stem cells for the restoration of auditory function in humans. Regen Med 8:309–318. https://doi.org/10.2217/rme.13.32
Ishino Y, Shinagawa H, Makino K et al (1987) Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J Bacteriol 169:5429–5433. https://doi.org/10.1128/jb.169.12.5429-5433.1987
Jansen R, van Embden JD, Gaastra W et al (2002) Identification of a novel family of sequence repeats among prokaryotes. OMICS. 6:23–33. https://doi.org/10.1089/15362310252780816
Jinek M, Chylinski K, Fonfara I et al (2012) A programmable dual-RNA–guided DNA endonuclease in adaptive bacterial immunity. Science 337:816–821. https://doi.org/10.1126/science.1225829
Kalatzis V, Petit C (1998) The fundamental and medical impacts of recent progress in research on hereditary hearing loss. Hum Mol Genet 7:1589–1597. https://doi.org/10.1093/hmg/7.10.1589
Kang X, He W, Huang Y et al (2016) Introducing precise genetic modifications into human 3PN embryos by CRISPR/Cas-mediated genome editing. J Assist Reprod Genet 33:581–588. https://doi.org/10.1007/s10815-016-0710-8
Kawamoto K, Yagi M, Stöver T et al (2003) Hearing and hair cells are protected by adenoviral gene therapy with TGF-β1 and GDNF. Mol Ther 7:484–492. https://doi.org/10.1016/s1525-0016(03)00058-3
Kim VN, Han J, Siomi MC (2009) Biogenesis of small RNAs in animals. Nat Rev Mol Cell Biol 10:126–139. https://doi.org/10.1038/nrm2632
Kim S, Kim D, Cho SW et al (2014) Highly efficient RNA-guided genome editing in human cells via delivery of purified Cas9 ribonucleoproteins. Genome Res 24:1012–1019. https://doi.org/10.1101/gr.171322.113
Kleinstiver BP, Prew MS, Tsai SQ et al (2015) Engineered CRISPR-Cas9 nucleases with altered PAM specificities. Nature 523:481–485. https://doi.org/10.1038/nature14592
Koehler KR, Mikosz AM, Molosh AI et al (2013) Generation of inner ear sensory epithelia from pluripotent stem cells in 3D culture. Nature 500:217–221. https://doi.org/10.1038/nature12298
Koehler KR, Nie J, Longworth-Mills E et al (2017) Generation of inner ear organoids containing functional hair cells from human pluripotent stem cells. Nat Biotechnol 35:583–589. https://doi.org/10.1038/nbt.3840
Kono M, Belyantseva IA, Skoura A et al (2007) Deafness and stria vascularis defects in S1P2 receptor-null mice. J Biol Chem 282:10690–10696. https://doi.org/10.1074/jbc.M700370200
Kraft K, Geuer S, Will AJ et al (2015) Deletions, inversions, duplications: engineering of structural variants using CRISPR/Cas in mice. Cell Rep 10:833–839. https://doi.org/10.1016/j.celrep.2015.01.016
Kral A, O’donoghue GM (2010) Profound deafness in childhood. N Engl J Med 363:1438–1450. https://doi.org/10.1056/NEJMra0911225
Liang X, Potter J, Kumar S et al (2015) Rapid and highly efficient mammalian cell engineering via Cas9 protein transfection. J Biotechnol 208:44–53. https://doi.org/10.1016/j.jbiotec.2015.04.024
Lintner NG, Kerou M, Brumfield SK et al (2011) Structural and functional characterization of an archaeal clustered regularly interspaced short palindromic repeat (CRISPR)-associated complex for antiviral defense (CASCADE). J Biol Chem 286:21643–21656. https://doi.org/10.1074/jbc.M111.238485
Liu X, Yan D (2007) Ageing and hearing loss. J Pathol 211:188–197. https://doi.org/10.1002/path.2102
Liu S, Li S, Zhu H et al (2012) A mutation in the cdh23 gene causes age-related hearing loss in Cdh23 nmf308/nmf308 mice. Gene 499:309–317. https://doi.org/10.1016/j.gene.2012.01.084
Low BE, Krebs MP, Joung JK et al (2014) Correction of the Crb1rd8 allele and retinal phenotype in C57BL/6N mice via TALEN-mediated homology-directed repair. Invest Ophthalmol Vis Sci 55:387–395. https://doi.org/10.1167/iovs.13-13278
Lupiáñez DG, Kraft K, Heinrich V et al (2015) Disruptions of topological chromatin domains cause pathogenic rewiring of gene-enhancer interactions. Cell 161:1012–1025. https://doi.org/10.1016/j.cell.2015.04.004
Maeda Y, Fukushima K, Nishizaki K et al (2005) In vitro and in vivo suppression of GJB2 expression by RNA interference. Hum Mol Genet 14:1641–1650. https://doi.org/10.1093/hmg/ddi172
Maeda Y, Sheffield AM, Smith RJ (2009) Therapeutic regulation of gene expression in the inner ear using RNA interference. In: Gene therapy of cochlear deafness, vol 66. Karger Publishers, pp 13-36
Mali P, Yang L, Esvelt KM et al (2013) RNA-guided human genome engineering via Cas9. Science 339:823–826. https://doi.org/10.1126/science.1232033
Mao Z, Bozzella M, Seluanov A et al (2008) Comparison of nonhomologous end joining and homologous recombination in human cells. DNA Repair (Amst) 7:1765–1771. https://doi.org/10.1016/j.dnarep.2008.06.018
Marraffini LA, Sontheimer EJ (2010) Self versus non-self discrimination during CRISPR RNA-directed immunity. Nature 463:568–571. https://doi.org/10.1038/nature08703
Mashiko D, Fujihara Y, Satouh Y et al (2013) Generation of mutant mice by pronuclear injection of circular plasmid expressing Cas9 and single guided RNA. Sci Rep 3:3355. https://doi.org/10.1038/srep03355
Mashiko D, Young SA, Muto M et al (2014) Feasibility for a large scale mouse mutagenesis by injecting CRISPR/Cas plasmid into zygotes. Develop Growth Differ 56:122–129. https://doi.org/10.1111/dgd.12113
Matsuoka AJ, Morrissey ZD, Zhang C et al (2017) Directed differentiation of human embryonic stem cells toward placode-derived spiral ganglion-like sensory neurons. Stem Cells Transl Med 6:923–936. https://doi.org/10.1002/sctm.16-0032
McManus MT, Sharp PA (2002) Gene silencing in mammals by small interfering RNAs. Nat Rev Genet. 3:737–747. https://doi.org/10.1038/nrg908
Mianné J, Chessum L, Kumar S et al (2016) Correction of the auditory phenotype in C57BL/6N mice via CRISPR/Cas9-mediated homology directed repair. Genome Med 8:16. https://doi.org/10.1186/s13073-016-0273-4
Morton CC, Nance WE (2006) Newborn hearing screening—a silent revolution. N Engl J Med 354:2151–2164. https://doi.org/10.1056/NEJMra050700
Nishimasu H, Ran FA, Hsu PD et al (2014) Crystal structure of Cas9 in complex with guide RNA and target DNA. Cell 156:935–949. https://doi.org/10.1016/j.cell.2014.02.001
O’Connell MR, Oakes BL, Sternberg SH et al (2014) Programmable RNA recognition and cleavage by CRISPR/Cas9. Nature 516:263–266. https://doi.org/10.1038/nature13769
Oshima K, Shin K, Diensthuber M et al (2010) Mechanosensitive hair cell-like cells from embryonic and induced pluripotent stem cells. Cell 141:704–716. https://doi.org/10.1016/j.cell.2010.03.035
Pan B, Géléoc GS, Asai Y et al (2013) TMC1 and TMC2 are components of the mechanotransduction channel in hair cells of the mammalian inner ear. Neuron 79:504–515. https://doi.org/10.1016/j.neuron.2013.06.019
Patel K, Giese AP, Grossheim J et al (2015) A novel C-terminal CIB2 (calcium and integrin binding protein 2) mutation associated with non-syndromic hearing loss in a Hispanic family. PLoS One 10:e0133082. https://doi.org/10.1371/journal.pone.0133082
Peng T, Dong Y, Zhu G et al (2014) Induced pluripotent stem cells: landscape for studying and treating hereditary hearing loss. J Otol 9:151–155. https://doi.org/10.1016/j.joto.2015.02.001
Perny M, Ting C-C, Kleinlogel S et al (2017) Generation of otic sensory neurons from mouse embryonic stem cells in 3D culture. Front Cell Neurosci 11:409. https://doi.org/10.3389/fncel.2017.00409
Pulleyn L, Jackson A, Roberts E et al (2000) A new locus for autosomal recessive non-syndromal sensorineural hearing impairment (DFNB27) on chromosome 2q23–q31. Eur J Hum Genet 8:991–993. https://doi.org/10.1038/sj.ejhg.5200567
Qin W, Dion SL, Kutny PM et al (2015) Efficient CRISPR/Cas9-mediated genome editing in mice by zygote electroporation of nuclease. Genetics 200:423–430. https://doi.org/10.1534/genetics.115.176594
Ran FA, Hsu PD, Lin C-Y et al (2013) Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154:1380–1389. https://doi.org/10.1016/j.cell.2013.08.021
Ran FA, Cong L, Yan WX et al (2015) In vivo genome editing using Staphylococcus aureus Cas9. Nature 520:186–191. https://doi.org/10.1038/nature14299
Riazuddin S, Belyantseva IA, Giese AP et al (2012) Alterations of the CIB2 calcium-and integrin-binding protein cause Usher syndrome type 1J and nonsyndromic deafness DFNB48. Nat Genet 44:1265–1271. https://doi.org/10.1038/ng.2426
Richardson C, Jasin M (2000) Frequent chromosomal translocations induced by DNA double-strand breaks. Nature 405:697–700. https://doi.org/10.1038/35015097
Richardson CD, Ray GJ, DeWitt MA et al (2016) Enhancing homology-directed genome editing by catalytically active and inactive CRISPR-Cas9 using asymmetric donor DNA. Nat Biotechnol 34:339–344. https://doi.org/10.1038/nbt.3481
Rong YS, Golic KG (2000) Gene targeting by homologous recombination in Drosophila. Science 288:2013–2018. https://doi.org/10.1126/science.288.5473.2013
Ruf S, Symmons O, Uslu VV et al (2011) Large-scale analysis of the regulatory architecture of the mouse genome with a transposon-associated sensor. Nat Genet 43:379–386. https://doi.org/10.1038/ng.790
Sakuma T, Nishikawa A, Kume S et al (2014) Multiplex genome engineering in human cells using all-in-one CRISPR/Cas9 vector system. Sci Rep 4:5400. https://doi.org/10.1038/srep05400
Sashital DG, Wiedenheft B, Doudna JA (2012) Mechanism of foreign DNA selection in a bacterial adaptive immune system. Mol Cell 46:606–615. https://doi.org/10.1016/j.molcel.2012.03.020
Seco CZ, Giese AP, Shafique S et al (2016) Novel and recurrent CIB2 variants, associated with nonsyndromic deafness, do not affect calcium buffering and localization in hair cells. Eur J Hum Genet 24:542–549. https://doi.org/10.1038/ejhg.2015.157
Semenova E, Jore MM, Datsenko KA et al (2011) Interference by clustered regularly interspaced short palindromic repeat (CRISPR) RNA is governed by a seed sequence. Proc Natl Acad Sci U S A 108:10098–10103. https://doi.org/10.1073/pnas.1104144108
Shang H, Yan D, Tayebi N et al (2018) Targeted next-generation sequencing of a deafness gene panel (MiamiOtoGenes) analysis in families unsuitable for linkage analysis. Biomed Res Int 2018:3103986. https://doi.org/10.1155/2018/3103986
Shibata SB, Ranum PT, Moteki H et al (2016) RNA interference prevents autosomal-dominant hearing loss. Am J Hum Genet 98:1101–1113. https://doi.org/10.1016/j.ajhg.2016.03.028
Simon MD, Wang CI, Kharchenko PV et al (2011) The genomic binding sites of a noncoding RNA. Proc Natl Acad Sci U S A 108:20497–20502. https://doi.org/10.1073/pnas.1113536108
Snoeckx RL, Huygen PL, Feldmann D et al (2005) GJB2 mutations and degree of hearing loss: a multicenter study. Am J Hum Genet 77:945–957. https://doi.org/10.1086/497996
Spielmann M, Kakar N, Tayebi N et al (2016) Exome sequencing and CRISPR/Cas genome editing identify mutations of ZAK as a cause of limb defects in humans and mice. Genome Res 26:183–191. https://doi.org/10.1101/gr.199430.115
Staecker H, Gabaizadeh R, Federoff H et al (1998) Brain-derived neurotrophic factor gene therapy prevents spiral ganglion degeneration after hair cell loss. Otolaryngol Head Neck Surg 119:7–13. https://doi.org/10.1016/S0194-5998(98)70194-9
Sternberg SH, Redding S, Jinek M et al (2014) DNA interrogation by the CRISPR RNA-guided endonuclease Cas9. Nature 507:62–67. https://doi.org/10.1038/nature13011
Sugai K, Fukuzawa R, Shofuda T et al (2016) Pathological classification of human iPSC-derived neural stem/progenitor cells towards safety assessment of transplantation therapy for CNS diseases. Mol Brain 9:85. https://doi.org/10.1186/s13041-016-0265-8
Suzuki M, Yagi M, Brown J et al (2000) Effect of transgenic GDNF expression on gentamicin-induced cochlear and vestibular toxicity. Gene Ther 7:1046–1054. https://doi.org/10.1038/sj.gt.3301180
Terns MP, Terns RM (2011) CRISPR-based adaptive immune systems. Curr Opin Microbiol 14:321–327. https://doi.org/10.1016/j.mib.2011.03.005
Urnov FD, Rebar EJ, Holmes MC et al (2010) Genome editing with engineered zinc finger nucleases. Nat Rev Genet. 11:636–646. https://doi.org/10.1038/nrg2842
Van Camp G, Smith RJ (2006) Hereditary hearing loss homepage. https://hereditaryhearingloss.org/
Volarevic V, Markovic BS, Gazdic M et al (2018) Ethical and safety issues of stem cell-based therapy. Int J Med Sci 15:36–45. https://doi.org/10.7150/ijms.21666
Wang Q, Lin JL-C, Wu K-H et al (2012) Xin proteins and intercalated disc maturation, signaling and diseases. Front Biosci (Landmark Ed) 17:2566–2593. https://doi.org/10.2741/4072
Wang Q, Lin JL-C, Chan SY et al (2013) The Xin repeat-containing protein, mXinβ, initiates the maturation of the intercalated discs during postnatal heart development. Dev Biol 374:264–280. https://doi.org/10.1016/j.ydbio.2012.12.007
Wang H, Wang X, Liao A et al (2017) Hypomethylation agent decitabine restores drug sensitivity by depressing P-glycoprotein activity through MAPK signaling pathway. Mol Cell Biochem 433:141–148. https://doi.org/10.1007/s11010-017-3022-0
Westra ER, van Erp PB, Künne T et al (2012) CRISPR immunity relies on the consecutive binding and degradation of negatively supercoiled invader DNA by Cascade and Cas3. Mol Cell 46:595–605. https://doi.org/10.1016/j.molcel.2012.03.018
Wiedenheft B, van Duijn E, Bultema JB et al (2011) RNA-guided complex from a bacterial immune system enhances target recognition through seed sequence interactions. Proc Natl Acad Sci U S A 108:10092–10097. https://doi.org/10.1073/pnas.1102716108
Wiedenheft B, Sternberg SH, Doudna JA (2012) RNA-guided genetic silencing systems in bacteria and archaea. Nature 482:331–338. https://doi.org/10.1038/nature10886
Yan D, Liu XZ (2008) Cochlear molecules and hereditary deafness. Front Biosci 13:4972–4983. https://doi.org/10.2741/3056
Yan D, Liu XZ (2010) Genetics and pathological mechanisms of Usher syndrome. J Hum Genet 55:327–335. https://doi.org/10.1038/jhg.2010.29
Yang J-J (2002) Mixed lineage kinase ZAK utilizing MKK7 and not MKK4 to activate the c-Jun N-terminal kinase and playing a role in the cell arrest. Biochemical and Biophysical Research Communications 297 (1):105–110. https://doi.org/10.1016/s0006-291x(02)02123-x
Yin H, Song C-Q, Dorkin JR et al (2016) Therapeutic genome editing by combined viral and non-viral delivery of CRISPR system components in vivo. Nat Biotechnol 34:328–333. https://doi.org/10.1038/nbt.3471
Yoshimura H, Shibata SB, Ranum PT et al (2019) Targeted allele suppression prevents progressive hearing loss in the mature murine model of human TMC1 deafness. Mol Ther 27:681–690. https://doi.org/10.1016/j.ymthe.2018.12.014
Zhang J, Rouillon C, Kerou M et al (2012) Structure and mechanism of the CMR complex for CRISPR-mediated antiviral immunity. Mol Cell 45:303–313. https://doi.org/10.1016/j.molcel.2011.12.013
Zhang Q, Peng C, Song J et al (2017) Germline mutations in CDH23, encoding cadherin-related 23, are associated with both familial and sporadic pituitary adenomas. Am J Hum Genet 100:817–823. https://doi.org/10.1016/j.ajhg.2017.03.011
Zhao Y, Wang D, Zong L et al (2014) A novel DFNA36 mutation in TMC1 orthologous to the Beethoven (Bth) mouse associated with autosomal dominant hearing loss in a Chinese family. PLoS One 9:e97064. https://doi.org/10.1371/journal.pone.0097064
Zine A, de Ribaupierre F (1998) Replacement of mammalian auditory hair cells. Neuroreport. 9:263–268. https://doi.org/10.1097/00001756-199801260-00016
Zou B, Mittal R, Grati M et al (2015) The application of genome editing in studying hearing loss. Hear Res 327:102–108. https://doi.org/10.1016/j.heares.2015.04.016
Zuris JA, Thompson DB, Shu Y et al (2015) Cationic lipid-mediated delivery of proteins enables efficient protein-based genome editing in vitro and in vivo. Nat Biotechnol 33:73–80. https://doi.org/10.1038/nbt.3081
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by: Michal Witt
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Farooq, R., Hussain, K., Tariq, M. et al. CRISPR/Cas9: targeted genome editing for the treatment of hereditary hearing loss. J Appl Genetics 61, 51–65 (2020). https://doi.org/10.1007/s13353-019-00535-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-019-00535-6