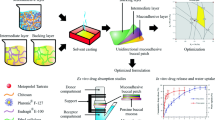
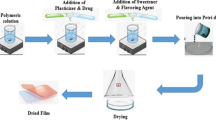
Abstract
The oral bioavailability of felodipine (FEL) is very low, i.e., about 15%. This could be due to low water solubility and hepatic first-pass effect. The objective of the present study was to develop FEL microemulsion-based gel, to bypass the first pass effect, for buccal delivery. The optimized FEL microemulsion (OPT-MEF) was used to prepare buccoadhesive gels, with varying concentrations of hydroxypropyl methylcellulose (HPMC) E4M and polycarbophil (PCP), and evaluated. The cross-linking of the PCP gelling agent was done by adjusting the pH with a neutralizing agent, triethanolamine (TEA). The formulations, namely drug suspension, OPT-MEF, microemulsion-based buccal gel containing 1% w/v (MEF-E4M1), 2% w/v (MEF-E4M2), and 3% w/v (MEF-E4M3) of HPMC K4M and 1% w/v (MEF-PCP1), 2% w/v (MEF-PCP2), and 3% w/v (MEF-PCP3) of PCP were prepared and optimized on the basis of ex vivo permeation study, mucoadhesion force, and viscosity. The optimized buccal gel (MEF-PCP1) showed significantly higher (p < 0.01) permeation flux (J = 0.44 ± 0.16 mg/cm2/h), when compared with the drug suspension (J = 0.17 ± 0.14 mg/cm2/h). The permeation enhancement ratio of MEF-PCP1 was found to be 2.59 times higher than that of the aqueous suspension of the drug. The texture profile analysis of MEF-PCP1 was performed which showed spreadability (3.2 mJ), extrudability (151.8 mJ), hardness (13.8 g), and adhesiveness (41.0 g), and results indicated good spreadability and adhesiveness. The rheological study revealed the pseudoplastic flow behavior of MEF-PCP1 buccal gel. The Cmax value 9.21 ± 2.88 μg/ml of MEF-PCP1 gel was found to be significantly higher (P < 0.01) compared to the same dose administered by oral route (Cmax value 3.51 ± 1.74 μg/ml). The relative bioavailability (Fr) of the optimized MEF-PCP1 buccal gel was about 397.39% higher than that of oral route. In conclusion, consistent and effective buccal gel containing optimized FEL-loaded microemulsion, with improved buccal permeation and pharmacokinetic parameters was developed successfully to improve the bioavailability of FEL.



Similar content being viewed by others
References
Ikram M, Gilhotra N, Gilhotra RM. Formulation and optimization of mucoadhesive buccal patches of losartan potassium by using response surface methodology. Adv Biomed Res. 2015;4:239.
Adhikari SN, Nayak BS, Nayak AK, Mohanty B. Formulation and evaluation of buccal patches for delivery of atenolol. AAPS Pharm Sci Tech. 2010;11(3):1038–44. https://doi.org/10.1208/s12249-010-9459-z.
Boyapally H, Nukala RK, Bhujbal P, Douroumis D. Controlled release from directly compressible theophylline buccal tablets. Colloids Surf B: Biointerfaces. 2010;77(2):227–33. https://doi.org/10.1016/j.colsurfb.2010.01.031.
Singh M, Kanoujia J, Singh P, Parashar P, Arya M, Tripathi CB, et al. Development of an α-linolenic acid containing a soft nanocarrier for oral delivery-part II: buccoadhesive gel. RSC Adv. 2016;6(103):101602–12. https://doi.org/10.1039/C6RA20896G.
Bruschi ML, de Freitas O. Oral bioadhesive drug delivery systems. Drug Dev Ind Pharm. 2005;31(3):293–310. https://doi.org/10.1081/DDC-52073.
Madhav NS, Shakya AK, Shakya P, Singh K. Orotransmucosal drug delivery systems: a review. J Control Release. 2009;140(1):2–11. https://doi.org/10.1016/j.jconrel.2009.07.016.
Mizrahi B, Domb AJ. Mucoadhesive polymers for delivery of drugs to the oral cavity. Recent Pat Drug Deliv Formul. 2008;2(2):108–19. https://doi.org/10.2174/187221108784534126.
Jain N, Jain GK, Javed S, Iqbal Z, Talegaonkar S, Ahmad FJ, et al. Recent approaches for the treatment of periodontitis. Drug Discov Today. 2008;13(21):932–43. https://doi.org/10.1016/j.drudis.2008.07.010.
Morales JO, McConville JT. Manufacture and characterization of mucoadhesive buccal films. Eur J Pharm Biopharm. 2011;77(2):187–99. https://doi.org/10.1016/j.ejpb.2010.11.023.
Saltiel E, Ellrodt AG, Monk JP, Langley MS. Felodipine. Drugs. 1988;36(4):387–428. https://doi.org/10.2165/00003495-198836040-00002.
Patil PR, Biradar SV, Paradkar AR. Extended-release felodipine self-nanoemulsifying system. AAPS PharmSciTech. 2009;10(2):515–23. https://doi.org/10.1208/s12249-009-9235-0.
Singh M, Kanoujia J, Singh P, Parashar P, Arya M, Tripathi CB, et al. Augmented bioavailability of felodipine through a α-linolenic acid based microemulsion. Drug Deliv Transl Res. 2018;8(1):204–25. https://doi.org/10.1007/s13346-017-0453-9.
Blychert E, Edgar B, Elmfeldt D, Hedner T. A population study of the pharmacokinetics of felodipine. Br J Clin Pharmacol. 1991;31(1):15–24. https://doi.org/10.1111/j.1365-2125.1991.tb03852.x.
Kim EJ, Chun MK, Jang JS, Lee IH, Lee KR, Choi HK. Preparation of a solid dispersion of felodipine using a solvent wetting method. Eur J Pharm Biopharm. 2006;64(2):200–5. https://doi.org/10.1016/j.ejpb.2006.04.001.
Ko J, Park HJ, Hwang S, Park J, Lee J. Preparation and characterization of chitosan microparticles intended for controlled drug delivery. Int J Pharm. 2002;249(1):165–74. https://doi.org/10.1016/S0378-5173(02)00487-8.
Acharya G, Shin CS, McDermott M, Mishra H, Park H, Kwon IC, et al. The hydrogel template method for fabrication of homogeneous nano/microparticles. J Control Release. 2010;141(3):314–9. https://doi.org/10.1016/j.jconrel.2009.09.032.
Sharma VK. Preparation of micron-size pharmaceutical particles by microfluidization. Google Patents; 2003.
Won DH, Kim MS, Lee S, Park JS, Hwang SJ. Improved physicochemical characteristics of felodipine solid dispersion particles by supercritical anti-solvent precipitation process. Int J Pharm. 2005;301(1):199–208. https://doi.org/10.1016/j.ijpharm.2005.05.017.
Karavas E, Georgarakis E, Bikiaris D. Felodipine nanodispersions as active core for predictable pulsatile chronotherapeutics using PVP/HPMC blends as coating layer. Int J Pharm. 2006;313(1):189–97. https://doi.org/10.1016/j.ijpharm.2006.01.015.
Jing B, Wang Z, Yang R, Zheng X, Zhao J, Tang S, et al. Enhanced oral bioavailability of felodipine by novel solid self-microemulsifying tablets. Drug Dev Ind Pharm. 2016;42(3):506–12. https://doi.org/10.3109/03639045.2015.1058816.
Singh M, Kanoujia J, Singh P, Tripathi CB, Arya M, Parashar P, et al. Development of an α-linolenic acid containing soft nanocarrier for oral delivery: in vitro and in vivo evaluation. RSC Adv. 2016;6(81):77590–602. https://doi.org/10.1039/C6RA15166C.
Rai VK, Yadav NP, Sinha P, Mishra N, Luqman S, Dwivedi H, et al. Development of cellulosic polymer based gel of novel ternary mixture of miconazole nitrate for buccal delivery. Carbohydr Polym. 2014;103:126–33. https://doi.org/10.1016/j.carbpol.2013.12.019.
Hixson A, Crowell J. Dependence of reaction velocity upon surface and agitation. Ind Eng Chem. 1931;23(10):1160–8. https://doi.org/10.1021/ie50262a025.
Higuchi T. Rate of release of medicaments from ointment bases containing drugs in suspension. J Pharm Sci. 1961;50(10):874–5. https://doi.org/10.1002/jps.2600501018.
Korsmeyer R, Gurny R, Doelker E, Buri P, Peppas N. Mechanisms of potassium chloride release from compressed, hydrophilic, polymeric matrices: effect of entrapped air. J Pharm Sci. 1983;72(10):1189–91. https://doi.org/10.1002/jps.2600721021.
Brookfield C. Texture Analyzer Operating Instructions Manual No: M/08-371A0708; 2011 Contract No.: Document Number|.
Bandyopadhyay S, Katare O, Singh B. Optimized self nano-emulsifying systems of ezetimibe with enhanced bioavailability potential using long chain and medium chain triglycerides. Colloids Surf B: Biointerfaces. 2012;100:50–61. https://doi.org/10.1016/j.colsurfb.2012.05.019.
Kumria R, Nair AB, Goomber G, Gupta S. Buccal films of prednisolone with enhanced bioavailability. Drug Deliv. 2016;23(2):471–8. https://doi.org/10.3109/10717544.2014.920058.
Sahu BP, Das MK. Preparation and in vitro/in vivo evaluation of felodipine nanosuspension. Eur J Drug Metab Pharmacokinet. 2014;39(3):183–93. https://doi.org/10.1007/s13318-013-0158-5.
Sudhakar Y, Kuotsu K, Bandyopadhyay A. Buccal bioadhesive drug delivery—a promising option for orally less efficient drugs. J Control Release. 2006;114(1):15–40. https://doi.org/10.1016/j.jconrel.2006.04.012.
Perioli L, Pagano C, Mazzitelli S, Rossi C, Nastruzzi C. Rheological and functional characterization of new antiinflammatory delivery systems designed for buccal administration. Int J Pharm. 2008;356(1):19–28. https://doi.org/10.1016/j.ijpharm.2007.12.027.
Cid YP, Pedrazzi V, de Sousa VP, Pierre MBR. In vitro characterization of chitosan gels for buccal delivery of celecoxib: influence of a penetration enhancer. AAPS PharmSciTech. 2012;13(1):101–11. https://doi.org/10.1208/s12249-011-9725-8.
Carvalho FC, Bruschi ML, Evangelista RC, Gremião MPD. Mucoadhesive drug delivery systems. Braz J Pharm Sci. 2010;46(1):1–17. https://doi.org/10.1590/S1984-82502010000100002.
Salamat-Miller N, Chittchang M, Johnston TP. The use of mucoadhesive polymers in buccal drug delivery. Adv Drug Deliv Rev. 2005;57(11):1666–91.
Reddy CP, Chaitanya K, Rao MY. A review on bioadhesive buccal drug delivery systems: current status of formulation and evaluation methods. Daru. 2011;19(6):385–403.
Robinson JR, Mlynek GM. Bioadhesive and phase-change polymers for ocular drug delivery. Adv Drug Deliv Rev. 1995;16(1):45–50. https://doi.org/10.1016/0169-409X(95)00013-W.
Attia M, El-Gibaly I, Shaltout S, Fetih G. Transbuccal permeation, anti-inflammatory activity and clinical efficacy of piroxicam formulated in different gels. Int J Pharm. 2004;276(1):11–28.
Kim H, Fassihi R. A new ternary polymeric matrix system for controlled drug delivery of highly soluble drugs: I. Diltiazem hydrochloride. Pharm Res. 1997;14(10):1415–21. https://doi.org/10.1023/A:1012124806316.
Ritger PL, Peppas NA. A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J Control Release. 1987;5(1):37–42. https://doi.org/10.1016/0168-3659(87)90035-6.
Korsmeyer RW, Gurny R, Doelker E, Buri P, Peppas NA. Mechanisms of solute release from porous hydrophilic polymers. Int J Pharm. 1983;15(1):25–35. https://doi.org/10.1016/0378-5173(83)90064-9.
Gaspar L, Campos PM. Rheological behavior and the SPF of sunscreens. Int J Pharm. 2003;250(1):35–44. https://doi.org/10.1016/S0378-5173(02)00462-3.
Inoue Y, Furuya K, Matumoto M, Murata I, Kimura M, Kanamoto I. A comparison of the physicochemical properties and a sensory test of acyclovir creams. Int J Pharm. 2012;436(1):265–71. https://doi.org/10.1016/j.ijpharm.2012.06.023.
Patel VF, Liu F, Brown MB. Advances in oral transmucosal drug delivery. J Control Release. 2011;153(2):106–16. https://doi.org/10.1016/j.jconrel.2011.01.027.
Smart JD. Buccal drug delivery. Expert Opin Drug Deliv. 2005;2(3):507–17. https://doi.org/10.1517/17425247.2.3.507.
Acknowledgements
The authors acknowledge the kind support of IPCA Pharmaceuticals, Pvt. Ltd., Mumbai, for providing the gift sample of felodipine and also acknowledge the UIPS department, Panjab University, Chandigarh, for providing some evaluation facilities for this research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Singh, M., Kanoujia, J., Parashar, P. et al. Assessment of improved buccal permeation and bioavailability of felodipine microemulsion-based cross-linked polycarbophil gel. Drug Deliv. and Transl. Res. 8, 591–601 (2018). https://doi.org/10.1007/s13346-018-0489-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-018-0489-5