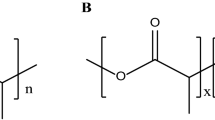
Abstract
In recent studies, we microencapsulated pancreatic β-cells using sodium alginate (SA) and poly-L-ornithine (PLO) and the bile acid, ursodeoxycholic acid (UDCA), and tested the morphology and cell viability post-microencapsulation. Cell viability was low probably due to limited strength of the microcapsules. This study aimed to assess a β-cell delivery system which consists of UDCA-based microcapsules incorporated with water-soluble gel matrix. The polyelectrolytes, water-soluble gel (WSG), polystyrenic sulphate (PSS), PLO and polyallylamine (PAA) at ratios 4:1:1:2.5 with or without 4 % UDCA, were incorporated into our microcapsules, and cell viability, metabolic profile, cell functionality, insulin production, levels of inflammation, microcapsule morphology, cellular distribution, UDCA partitioning, biocompatibility, thermal and chemical stabilities and the microencapsulation efficiency were examined. The incorporation of UDCA with PSS, PAA and WSG enhanced cell viability per microcapsule (p < 0.05), cellular metabolic profile (p < 0.01) and insulin production (p < 0.01); reduced the inflammatory release TNF-α (p < 0.01), INF-gamma (p < 0.01) and interleukin-6 (IL-6) (p < 0.01); and ceased the production of IL-1β. UDCA, PSS, PAA and WSG addition did not change the microencapsulation efficiency and resulted in biocompatible microcapsules. Our designed microcapsules showed good morphology and desirable insulin production, cell functionality and reduced inflammatory profile suggesting potential applications in diabetes.



Similar content being viewed by others
References
Chang TM, Johnson LJ, Ransome OJ. Semipermeable aqueous microcapsules. IV. nonthrombogenic microcapsules with heparin-complexed membranes. Can J Physiol Pharmacol. 1967;45(4):705–15.
Negrulj R, Mooranian A, Al-Salami H. Potentials and limitations of bile acids in type 2 diabetes mellitus: applications of microencapsulation as a novel oral delivery system. J Endocrinol Diabetes Mellitus. 2013;1(2):49–59.
Lim GJ, Zare S, Van DM, Atala A. Cell microencapsulation. Adv Exp Med Biol. 2010;670:126–36.
Chang TM. Therapeutic applications of polymeric artificial cells. Nat Rev Drug Discov. 2005;4(3):221–35.
Calafiore R, Basta G. Clinical application of microencapsulated islets: actual prospectives on progress and challenges. Adv Drug Deliv Rev. 2013.
Mooranian A, Negrulj R, Chen-Tan N, Fakhoury M, Arfuso F, Jones F, et al. Advanced bile acid-based multi-compartmental microencapsulated pancreatic beta-cells integrating a polyelectrolyte-bile acid formulation, for diabetes treatment. Artif Cells Nanomed Biotechnol. 2014:1–8
Mooranian A, Negrulj R, Arfuso F, Al-Salami H. Characterization of a novel bile acid-based delivery platform for microencapsulated pancreatic β-cells. Artif Cells Nanomed Biotechnol. 2014(0):1–7
Souza YE, Chaib E, Lacerda PG, Crescenzi A, Bernal-Filho A, D’Albuquerque LA. Islet transplantation in rodents. Do encapsulated islets really work? Arq Gastroenterol. 2011;48(2):146–52.
de Vos P, Lazarjani HA, Poncelet D, Faas MM. Polymers in cell encapsulation from an enveloped cell perspective. Adv Drug Deliv Rev. 2013.
Dominguez-Bendala JPA and Ricordi C. islet cell therapy and pancreatic stem cells. 2013;2:835–53.
Mineo D, Sageshima J, Burke GW, Ricordi C. Minimization and withdrawal of steroids in pancreas and islet transplantation. Transpl Int. 2009;22(1):20–37.
Weir GC. Islet encapsulation: advances and obstacles. Diabetologia. 2013;56(7):1458–61.
Beck J, Angus R, Madsen B, Britt D, Vernon B, Nguyen KT. Islet encapsulation: strategies to enhance islet cell functions. Tissue Eng. 2007;13(3):589–99.
Hamaguchi K, Gaskins HR, Leiter EH. NIT-1, a pancreatic beta-cell line established from a transgenic NOD/Lt mouse. Diabetes. 1991;40(7):842–9.
Mooranian A, Negrulj R, Arfuso F, Al-Salami H. Characterization of a novel bile acid-based delivery platform for microencapsulated pancreatic beta-cells. Artif Cells Nanomed Biotechnol. 2014:1–7.
Mooranian A, Negrulj R, Chen-Tan N, Al-Sallami HS, Fang Z, Mukkur T, et al. Novel artificial cell microencapsulation of a complex gliclazide-deoxycholic bile acid formulation: a characterization study. Drug Des Devel Ther. 2014;8:1003.
Mooranian A, Negrulj R, Mathavan S, Martinez J, Sciarretta J, Chen-Tan N, et al. An advanced microencapsulated system: a platform for optimized oral delivery of antidiabetic drug-bile acid formulations. Pharm Dev Technol. 2015;20(6):702–9.
Mooranian A, Negrulj R, Chen-Tan N, Watts GF, Arfuso F, Al-Salami H. An optimized probucol microencapsulated formulation integrating a secondary bile acid (deoxycholic acid) as a permeation enhancer. Drug Des Devel Ther. 2014;8:1673–83.
Mooranian A, Negrulj R, Chen-Tan N, Al-Sallami HS, Fang Z, Mukkur T, et al. Microencapsulation as a novel delivery method for the potential antidiabetic drug. Probucol Drug Des Devel Ther. 2014;8:1221–30.
Uludag H, Sefton MV. Colorimetric assay for cellular activity in microcapsules. Biomaterials. 1990;11(9):708–12.
Mooranian A, Negrulj R, Mikov M, Golocorbin-Kon S, Arfuso F, Al-Salami H. Novel chenodeoxycholic acid-sodium alginate matrix in the microencapsulation of the potential antidiabetic drug, probucol. An in vitro study. J Microencapsul. 2015;32(6):589–97.
Donath MY, Böni-Schnetzler M, Ellingsgaard H, Ehses JA. Islet inflammation impairs the pancreatic β-cell in type 2 diabetes. Physiology (Bethesda, Md). 2009;24:325–31.
Perez MJ, Briz O. Bile-acid-induced cell injury and protection. World J Gastroenterol. 2009;15(14):1677–89.
Stepanov V, Stankov K, Mikov M. The bile acid membrane receptor TGR5: a novel pharmacological target in metabolic, inflammatory and neoplastic disorders. J Recept Signal Transduct Res. 2013;33(4):213–23.
Brand Martin D, Nicholls DG. Assessing mitochondrial dysfunction in cells. Biochem J. 2011;435(Pt 2):297–312.
Wu M, Neilson A, Swift AL, Moran R, Tamagnine J, Parslow D, et al. Multiparameter metabolic analysis reveals a close link between attenuated mitochondrial bioenergetic function and enhanced glycolysis dependency in human tumor cells. Am J Physiol Cell Physiol. 2007;292(1):C125–36.
Arai T, Wilson DL, Kasai N, Freddi G, Hayasaka S, Tsukada M. Preparation of silk fibroin and polyallylamine composites. J Appl Polym Sci. 2002;84(11):1963–70.
Gill P, Moghadam TT, Ranjbar B. Differential scanning calorimetry techniques: applications in biology and nanoscience. J Biomol Tech. 2010;21(4):167.
Lamcharfi L, Meyer C, Lutton C. Rationalization of the relative hydrophobicity of some common bile acids by infrared and Raman spectroscopy. Biospectroscopy. 1997;3(5):393–401.
Darrabie MD, Kendall Jr WF, Opara EC. Characteristics of poly-L-ornithine-coated alginate microcapsules. Biomaterials. 2005;26(34):6846–52.
Tam SK, Dusseault J, Polizu S, Ménard M, Hallé J-P, Yahia LH. Physicochemical model of alginate–poly-L-lysine microcapsules defined at the micrometric/nanometric scale using ATR-FTIR, XPS, and ToF-SIMS. Biomaterials. 2005;26(34):6950–61.
Tam S, Bilodeau S, Dusseault J, Langlois G, Hallé J-P, Yahia L. Biocompatibility and physicochemical characteristics of alginate–polycation microcapsules. Acta Biomater. 2011;7(4):1683–92.
Bhatia SR, Khattak SF, Roberts SC. Polyelectrolytes for cell encapsulation. Curr Opin Colloid Interface Sci. 2005;10(1):45–51.
de Vos P, Faas MM, Strand B, Calafiore R. Alginate-based microcapsules for immunoisolation of pancreatic islets. Biomaterials. 2006;27(32):5603–17.
Acknowledgments
The authors acknowledge Australian Postgraduate Award (APA) and Curtin Research Scholarship (CRS) and acknowledge the use of laboratory equipment, scientific and technical assistance of the Curtin University, Electron Microscope Facility, which has been partially funded by the University, State and Commonwealth Governments. The authors acknowledge the Pharmaceutical Technology Laboratory (Curtin School of Pharmacy) and acknowledge the ARC Centre of Excellence in Plant Energy Biology (UWA) for the access to equipment. The authors acknowledge the generous donation of NIT-1 cells from Professor Grant Morahan at the University of Western Australia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mooranian, A., Negrulj, R. & Al-Salami, H. The incorporation of water-soluble gel matrix into bile acid-based microcapsules for the delivery of viable β-cells of the pancreas, in diabetes treatment: biocompatibility and functionality studies. Drug Deliv. and Transl. Res. 6, 17–23 (2016). https://doi.org/10.1007/s13346-015-0268-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-015-0268-5