Abstract
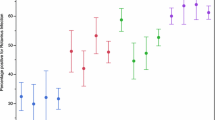
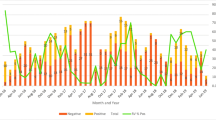
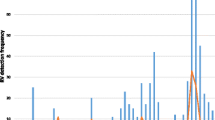
Rotavirus is the leading cause of acute gastroenteritis in worldwide young children. Effective vaccines to prevent rotavirus infection are currently available, although their clinical use is still limited, and rotavirus still causes many episodes of infantile gastroenteritis, mainly during the winter season. The aim of this study was to evaluate the prevalence of rotavirus infection in children aged <5-years-old who were hospitalised for gastroenteritis. One hundred and sixty-three stool samples from hospitalised children (<5-years-old) complicated with severe diarrhoea, in two hospitals in Jahrom City, Iran were collected from 2009 to 2010. Antigenic prevalence of rotavirus group A was distinguished by enzyme immunoassay. The antigen of group A rotavirus was diagnosed by EIA in 75 of 163 collected samples. The genotype of EIA-positive samples was determined by nested RT-PCR. The frequency of rotavirus genotypes G1, G2, G3, G4 and G9 was 17.33, 13.34, 2.67, 30.66 and 2.67 %, respectively. Also, the frequency of mixed and non-typable genotypes was detected in 2.67 and 30.66 %, respectively. G1/G8 mixed infection was the first of these rotavirus genotypes to be reported in Iran. Detection of high prevalence of group A rotavirus infection in hospitalised children with diarrhoea, and determination of circulating rotavirus genotypes in this region of Iran, provide useful data for formulating effective vaccines; especially for infants less than 5-years-old.


Similar content being viewed by others
References
Nguyen TV, Le Van Ph, Le Huy C, Weintraub A. Diarrhaea caused by rotavirus in children less than 5 years of age in Hanoi, Vietnam. J Clin Microbiol. 2004;42:5745–50.
Kim SY, Goldie SJ, Salomon JA. Cost-effectiveness of rotavirus vaccination in Vietnam. BMC Public Health. 2009;21(9):29.
Bioshop RF, Masendycz PJ, Bugg HC, Carlin JB, Baranes GL. Epidemiological patterns of rotavirus causing severe gastroenteritis in young children throughout Australia from 1993 to 1996. J Clin Microbiol. 2001;39:1085–91. doi:10.1128/JCM.39.3.1085-1091.2001.
Martinez-Laso J, Román A, Head J, Cervera I, Rodriguez M, Rodriguez-Avial I, Picazo JJ. Phylogeny of G9 rotavirus genotype: a possible explanation of its origin and evolution. J Clin Virol. 2009;44:52–7. doi:10.1016/j.jcv.2008.08.022.
Adlhoch C, Kaiser M, Hoehne M, Mas Marques A, Stefas I, Ceas F, Ellebrok H. Highly sensitive detection of the group A rotavirus using apoliporotein H-coated ELISA plates compared to quantitative real-time PCR. Virol J. 2011;8:63. doi:dio:10.1186/1743-422X-8-68.
Nakagomi T, Cuevas LE, Gurgel RG, Elrokhsi SH, Belkhir YA, Abugalia M, Dove W, Montenegro FM. Apparent extinction of non-G2 rotavirus strains from circulation in Recife, Brazil, after the introduction of rotavirus vaccine. Arch Virol. 2008;153:591–3.
Santos N, Hoshino Y. Global distribution rotavirus serotypes/genotypes and its implication for the development and implementation of an effective rotavirus vaccine. Rev Med Virol. 2005;15:29–56. doi:10.1002/rmv.448.
Rahman M, Matthijnssens J, Goegebuer T, De Leener K, Vanderwegen L, Van Hoovels L, De Vos S, Azim T, Van Ranst M. Predominance of rotavirus G9 genotype in children hospitalized for rotavirus gastroenteritis in Belgium during 1999–2003. J Clin Virol. 2005;33:1–6.
Romany S, Banerjee I, Gladstone B, Sarkar R, Selvapandian D, Le Fever AM, Jaffar Sh, Iturriza-Gomara M, Gray JJ, Estes MK. Geographic information systems and genotyping in identification of rotavirus G12 infections in residents of an urban slum with subsequent detection in hospitalized children: emergence of G12 genotype in South India. J Clin Virol. 2007;45:432–7. doi:10.1128/JCM.01710-06.
Samarbafzadeh A, Tehrani E, Makvandi M, Taremi M. Epidemiological aspects of rotavirus infection in Ahwaz, Iran. J Health Popul Nurt. 2005;23:245–9.
Gouvea V, Glass RI, Wood P, Taniguchi K, Clark HF, Forrester B, Fang ZH. Polymerase chain reaction amplification and typing of rotavirus nucleic acid from stool specimens. J Clin Virol. 1990;28:276–82.
Gouvea V, Santos N, Timenetsky MC. Identification of bovine and porcine rotavirus G types by PCR. J Clin Virol. 1994;32:1338–40.
Bok K, Castagnaro N, Borsa A, Nates S, Espul C, Fay O, Fabri A, Grinstein S, Miceli I, Matson DO, Gomez JA. Surveillance for rotavirus in Argentina. J Med Virol. 2001;65:190–8. doi:10.1002/jmv.2020.
Staat MA, Azimi PH, Berke T, Roberts N, Bernstein DI, Ward RI, Pickering LK, Matson DO. Clinical presentations of rotavirus infection among hospitalized children. Pediatr Infect Dis J. 2002;21:221–7.
Kazemi A, Tabatabaie F, Agha-Ghazvini MR, Kelishadi R. The role of rotavirus in acute paediatric diarrhea in Isfahan, Iran. Pak J Med Sci. 2006;22:282–5.
Jin Y, Ye H, Fang ZY, Li YN, Yang XM, Dong QL, Huang X. Molecular epidemic feature and variation of rotavirus among children with diarrhaea in Lanzhou, China, 2001–2006. World J Pediatr. 2008;4:197–201. doi:10.1007/s12519-008-0036-4.
López-de-Andreś A, Jiménez-García R, Carrasco-Garrido P, Alvaro-Meca A, Galarza PG, de Miguel AG. Hospitalizations associated with rotavirus gastroenteritis in Spain, 2001–2005. BMC Public Health. 2008;8:109. doi:10.1186/1471-2458-8-109.
Kargar M, Zaree-Mahmood-abadi B, Tabatabaei H. Genotyping of VP7 protein with Nested RT-PCR in children hospitalized in Tehran. Iranian J Infect Dis Trop Med. 2008; 12:11–17 (Persian).
Suzuki H, Sakai T, Tanabe N, Okabe N. Peak rotavirus activity shifted from winter to early spring in Japan. Pediatr Infect Dis J. 2005;24:257–60.
Intusoma U, Sornsrivichai V, Jiraphongsa C, Varavithaya W. Epidemiology, clinical presentations and burden of rotavirus diarrhaea in children under five see at Ramathibodi hospital, Thailand. J Med Assoc Thai. 2008;91:1350–5.
Zhang LJ, Zhang Q. Burden of hospitalizations attributable to rotavirus diarrhaea in the People, s Republic of China, August 2001–July 2003. J Infect Dis. 2005;192((Suppl1)):S94–9.
Iturriza-Gomara M, Kang G, Gray J. Rotavirus genotyping: keeping up with an evolving population of human rotaviruses. J Clin Virol. 2004;31:259–65. doi:10.1016/j.jcv.2004.04.009.
Desselberger U, Wolleswinkel-Van den Bosch J, Mrukowicz J, Mrukowicz J, Rodrigo C, Giaquinto C, Vesikari T. Rotavirus types in Europe and their significance for vaccination. Pediatr Infect Dis J. 2006;25:S30–41.
Khalili B, Cuevas LE, Reisi N, Dove W, Cunliffe NA, Hart CA. Epidemiology of rotavirus diarrhoea in Iranian children. J Med Virol. 2004;73:309–12. doi:10.1002/jmv.20092.
Ahmed MU, Alam MM, Chowdhury NS, Haque MM, Shahid N, Kobayashi N, Taniguchi K, Urasawa T, Urasawa S. Analysis of human rotavirus G serotype in Bangladesh by enzyme-linked immunosorbent assay and polymerase chain reaction. J Diaprhoeal Dis Res. 1999;17:22–7.
Sravanan P, Ananthan S, Ananthasubramanian M. Rotavirus infection among infants and young children in Chennai, South India. Indian J Med Microbiol. 2004;22:212–21.
Van Damme P, Giaquinto C, Maxwell M, Todd P, Van der Wielen M. Distribution of rotavirus genotypes in Europe, 2004–2005: the REVEAL study. J Infect Dis. 2007;195:S17–25.
Santos N, Lima RCC, Pereira CFA, Gouvea V. Detection of rotavirus types G8 and G10 among Brazilian children with diarrhaea. J Clin Microbiol. 1998;36:2727–9.
Reidy N, ÓHalloran F, Fanning S, Cryan B, ÓShea H. Emergence of G3 and G9 rotavirus and increased incidence mixed infections in the Southern region of Ireland 2001–2004. J Med Virol. 2005;27:571–8. doi:10.1002/jmv.20492.
Ahed MI, Wade A, Taniguchi K. Molecular epidemiology of rotavirus in Nigeria: detection of unusual strains with G2P[6] and G8P[1] specificities. J Clin Microbiol. 2001;39:3969–75. doi:10.1128/JCM.39.11.3969-3975.2001.
Antunes H, Afonso A, Iturriza M, Martinho I, Ribeiro C, Magalhães C, Carvalho L, Branca F, Gray J. G2P[4] the most prevalent rotavirus genotype in 2007 winter season in European non-vaccinated population. J Clin Virol. 2009;45:76–8. doi:10.1016/j.jcv.2009.03.010.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kargar, M., Akbarizadeh, A.R. Prevalence and Molecular Genotyping of Group A Rotaviruses in Iranian Children. Indian J. Virol. 23, 24–28 (2012). https://doi.org/10.1007/s13337-012-0070-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-012-0070-7