Abstract
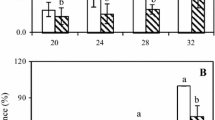
Unripe mangoes contain a network of minute latex canals in its exocarp, outer mesocarp and the pedicel. Latex, when retrieved, separates into an upper oily layer containing antifungal resorcinols and a lower aqueous layer with chitinase activity. Latex disappears in coincidence with ripening and decline of fruit resistance to fungal pathogens. The present study investigated if retention of latex at harvest enhances fruit resistance and reduces anthracnose and the stem-end rot (SER) development during ripening. Latex was retained by harvesting fruit with a portion (approximately 1 cm) of pedicel while in the controls, latex was drained off by removing the pedicel. Anthracnose and SER development from natural infections or following artificial inoculation was assessed at ripe stage. The results showed a significant reduction in the incidence and severity of anthracnose in the cultivar ‘Willard’ susceptible to anthracnose when latex was retained at harvest. There was delayed SER development when latex was retained in the susceptible cultivar ‘Karutha Colomban’. A negative trend was observed between the pedicel length and anthracnose or SER level in cultivars susceptible to the two respective diseases. The fruit peel in which latex was retained had greater chitinase activity. The reduction of anthracnose and SER could be due to the greater resorcinols and chitinase activity respectively in latex-retained fruit. The results indicate a direct involvement of latex in fruit resistance and the possibility of its manipulation to protect ripe fruit from fungal rotting.






Similar content being viewed by others
References
Adikaram NKB, Karunanayake LC, Abayasekara CL (2010) The role of pre-formed antifungal substances in the resistance of fruits to postharvest pathogens. In: Gullino L, Prusky D (eds) Postharvest Pathology. Springer, New York, pp 1–13
Bandyopadhyay C, Gholap AS, Mamdapur VR (1985) Characterization of alkenylresorcinols in mango (Mangifera indica L.) latex. J Agr Food Chem 33:377–379
Cojocaru M, Droby S, Glotter E, Goldman A, Glottlieb H, Jacoby B, Prusky D (1986) 5-(12-heptadecenyl)-resorcinol, the major component of the antifungal agent from the peel of mango fruit. Phytochemistry 25:1093–1095
Droby S, Prusky D, Jacoby B, Goldman A (1986) The presence of an antifungal compound and its relation to the latency of Alternaria alternata in unripe peels of mango fruits. Physiol Mol Plant Pathol 29:173–183
Hassan MK 2006 Constitutive alk(en)ylresorcinols and resistance to postharvest disease in mango (Mangifera indica L.). PhD Thesis, University of Queensland, Australia
Hassan MK, Dann EK, Irving DE, Coates LM (2007) Concentrations of constitutive alk(en)ylresorcinols in peel of commercial mango varieties and resistance to postharvest anthracnose. Physiol Mol Plant Pathol 71:158–165
Hassan MK, Irving DE, Dann EK, Coates LM, Hofman PJ (2009) Sap properties and alk(en)ylresorcinol concentrations in Australian-grown mangoes. Ann Appl Biol 154:419–427
Holmes RJ, O’Hare TJ, Underhill SJR, Ledger SN, Bally ISE, Macloed WNB, Ferguson J, Bowden BF Landrigan M (1999) FR440: mango skin browning. Horticultural Research and Development Corporation, Gordon, pp 50–57
Joel DM (1980) Resin ducts in the mango fruit: a defence system. J Exp Bot 31:1707–1718
Joel DM (1981) The duct system of the base and stalk of the mango fruit. Bot Gaz 142(3):329–333
John SK, Bhat SG, Prasad Rao UJ (2003) Biochemical characterization of sap (latex) of a few Indian mango varieties. Phytochemistry 62:13–19
Johnson G, Cooke T, Mead AJ (1992) Infection and quiescence of mango stem-end rot pathogens. Acta Horticult 341:329–336
Karunanayake CL, Adikaram NKB, Kumarihamy BMM, Ratnayake Bandara BM, Abayasekara C (2011) Role of antifungal gallotannins, resorcinols and chitinases in the constitutive defence of immature mango (Mangifera indica L.) against Colletotrichum gloeosporioides. J Phytopathol 159:657–664
Karunanayake LC, Sinniah GD, Adikaram NKB, Abayasekara CL (2014) Cultivar differences in antifungal activity and the resistance to postharvest anthracnose and stem-end rot in mango (Mangifera indica L.). Australas Plant Pathol 43:151–159
Meah MB (1993) Mode of infection of mango stem-end rot pathogen Botryodiplodia theobromae Pat. (Bangladesh). Bangladesh J Botany 22:21–27
Negi PS, John KS, Rao U (2002) Antimicrobial activity of mango sap. Eur Food Res Technol 214:327–330
O’Hare TJ, Prasad A (1992) The alleviation of sap-induced mango skin injury by calcium hydroxide. Acta Horticult 321:372–381
Oka K, Saito F, Yasuhara T, Sugimito A (2004) A study of cross-reactions between mango contact allergens and urishiol. Contact Dermatitis 51:292–296
Robinson SP, Loveys BR, Chacko EK (1993) Polyphenol oxidase enzymes in the sap and skin of mango fruit. Aust J Plant Physiol 20:99–107
Sinniah GD, Adikaram NKB, Abayasekara CL (2013) Differential defence responses expressed in mango (Mangifera indica L.) cultivars resistant and susceptible to Colletotrichum gloeosporioides. Indian Phytopathol 66(1):34–40
Zou X, Nonogaki H, Welbaum GE (2002) A gel diffusion assay for visualization and quantification of chitinase activity. Mol Biotechnol 22:19–23
Acknowledgments
The authors gratefully acknowledge the Australian Centre for International Agricultural Research (ACIAR) for funding a collaborative research programme involving Queensland government (Department of Agriculture, Fisheries and Forestry).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karunanayake, K.O.L.C., Sinniah, G.D., Adikaram, N.K.B. et al. Retention of latex at harvest, enhanced mango (Mangifera indica L.) fruit resistance and reduced anthracnose and stem-end rot. Australasian Plant Pathol. 44, 113–119 (2015). https://doi.org/10.1007/s13313-014-0330-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-014-0330-7