Abstract
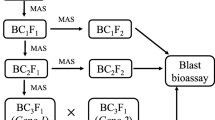
Blast disease is considered as a major limiting factor in the global rice production because of its wide distribution and destructiveness. Near Isogenic Lines (NILs) harboring different blast resistant Pi genes were surveyed for blast resistance along with resistant and susceptible varieties. These genotypes were randomly crossed to transfer disease resistance to agronomically superior varieties ADT 43, Improved White Ponni and BPT5204. Disease reaction was recorded in both artificial as well as natural epiphytotic conditions and differential response to blast disease incidence was observed. The minimum blast incidence was observed in F1s of ADT 43/CT13432-3R, ADT 43/C101A51 and ADT 43/C101LAC across the environments. Advanced back cross inbred lines developed from the cross combination ADT 43/CT13432-3R were also screened against blast disease. Gene pyramided back cross lines exhibited higher resistance than susceptible genotypes. Among the genotypes tested under epiphytotic conditions at different environments, lines with gene combinations Pi1 + Pi2 + Pi33 + Pi54 and Pi1 + Pi2 + Pi33 were highly resistant to blast disease than those with single genes indicating that these non-allelic genes have a complementary effect. Variation in resistance reaction was observed for the same gene when it acts alone or in combinations and also when the same genes were present in different genetic backgrounds. The gene pyramided lines developed in this study can serve as excellent donors for blast resistance in rice improvement and also for basic studies on gene-race interactions.












Similar content being viewed by others
References
Castano J, Amril B, Syahril D, Zaini Z (1990) Upland rice genotypes resistant to blast disease in west Sumatra. Int Rice Res Newslett 15:11–12
Chandrashekhra MV, Sunkad G, Naik MK, Nagaraju P (2008) Screening of Rice genotypes against blast caused by Pyricularia oryzae. Karnataka J Agric Sci 21(2):305–305
Chen DH, Vina MD, Inukai J, Mackill DJ, Ronald PC, Nelson RJ (1999) Molecular mapping of the blast resistance gene, Pi44(t), in a line derived from a durably resistant rice cultivar. Theor Appl Genet 98:1046–1053
Correa-Victoria FJ, Tharreau D, Martinez C, Vales M, Escobar F, Prado G, Aricada G (2002) Gene Combinations for durable rice blast (Pyricularia grisea) resistance in Colombia. Fitopatologia Colombiana 26:47–54
Dean R, Kan V, Pretorius ZA, Hammond-Kosack KE, Pietro AD, Pietro D, Rudd JJ, Dickman M, Kahmann R, Ellis J, Foster GD (2012) The Top 10 fungal pathogens in molecular plant pathology Issue. Mol Plant Pathol 13(4):414–430
FAOSTAT (2011) Statistical database of the food and agriculture of the united nations. FAO, Rome, Italy
Fujita D, Yoshimura A, Yasui H (2010) Development of near-isogenic lines and pyramided lines carrying resistance genes to green rice leafhopper (Nephotettix cincticeps Uhler) with the Taichung 65 genetic background in rice (Oryza sativa L.). Breed Sci 60:18–27
Fukuta Y, Tamura K, Hirae M, Oya S (1998) Genetic analysis of resistance to green rice leafhopper (Nephotettix cincticeps Uhler) in rice parental line, Norin-PL6, using RFLP markers. Breed Sci 48:243–249
Ghazanfar UM, Habib A, Sahi ST (2009) Pyricularia oryzae screening of rice germplasm against the cause of rice Blast disease. Pak J Phytopathol 21(1):41–44
Gowda M, Barman SR, Chattoo BB (2006) Molecular mapping of a novel blast resistance gene Pi38 in rice using SSR and AFLP markers. Plant Breed 125:596–599
Hittalmani S, Parco A, Mew TV, Zeigler RS, Huang N (2000) Fine mapping and DNA marker assisted pyramiding of the three major genes for blast resistance in rice. Theor Appl Genet 100:1121–1128
Jeon JS, Chen GHY, Wang GL, Ronald PC (2003) Genetic and physical mapping of Pi5(t), a locus associated with broad-spectrum resistance to rice blast. Theor Appl Genet 269:280–289
Khush GS, Jena KK (2009) Current status and future prospects for research on blast resistance in rice (Oryza sativa L.). In: Wang GL, Valent B (eds) Advances in genetics: genomics and control of rice blast disease. Springer, New York, pp 1–10
Li YB, Wu CJ, Jiang GH, Wang LQ, He YQ (2007) Dynamic analysis of rice blast resistance for the assessment of genetic and environmental effects. Plant Breed 126:541–547
Liu Y, Zhu XY, Zhang S, Bernardo M, Edwards J, Galbraith DW, Leach J, Zhang G, Liu B, Leung H (2011) Dissecting quantitative resistance against blast disease using heterogeneous inbred family lines in rice. Theor Appl Genet 122(2):341–353
Mackill DJ, Bonman JM (1992) Inheritance of blast resistance in near-isogenic lines of rice. Phytopathol 82:746–749
Maruyama K, Kikuchi F, Yokoo M (1983) Gene analysis of field resistance to rice blast (Pyricularia oryzae) in Rikuto Norin Mochi 4 and its use for breeding. Bull of Nat Agric Sci 35:1–31
Mohanta BK, Aslam MR, Kabir ME, Anam MK, Alam K, Habib MA (2003) Performance of different genotypes/cultivars to blast disease of rice in Boro and T. Aman crop in Bangladesh. Asian J Plant Sci 2(7):575–7
Mukherjee AK, Nayak P (1995) Identification of slow blasting resistance in early rice. Oryza 32:101–104
Nagaraju, Vasanthakumar HC, Devaiah BM, Sehadri VS, Naidu BS (1991) Evaluation of rice genotypes for blast and sheath rot resistance in hilly region of Mysore. J Agric Sci Karnataka 25:139–141
Ou SH (1985) Rice Diseases, 2nd edn. Commonwealth Mycological Institute Kew, UK, pp 380–392
Priya V (2008) Studies on the variability of Pyricularia grisea (Cooke) Sacc. M.Sc. Thesis, TNAU, Coimbatore
Ram T, Majumder ND, Mishra B, Ansari MM, Padmavathi G (2007) Introgression of broad-spectrum blast resistance gene(s) into cultivated rice (Oryza sativa ssp indica) from wild rice O. rufipogon. Curr Sci 92(2):225–230
Saifulla M, Nagaraju V, Kumar ML, Devaiah BM (1991) Evaluation of promising rice genotypes for blast. Oryza 28:281–282
Kumar S, Singh SS, Singh AK, Elanchezhian R, Sangale UR, Sundaram PK (2012) Evaluation of rice genotypes for resistance to blast disease under rainfed lowland ecosystem. J Pl Dis Sci 7(2):175–178
Selvaraj CI, Nagarajan P, Thiyagarajan K, Bharathi M, Rabindran R (2011) Genetic parameters of variability, correlation and path coefficient studies for grain yield and other yield Attributes among rice blast disease resistant genotypes of rice (Oryza sativa L.). Afr. J. of. Biotechnol 10(17):3322–3334
SES, IRRI (2002) Standard Evaluation System. International Rice Research Institute, Manila, Philippines, pp 11–30
Sharma TR, Gupta SK, Vijayan J, Devanna BN, Ray S (2012) Review: Rice Blast Management Through Host-Plant Resistance: Retrospect and Prospects. Agric Res 1(1):37–52
Sivaraj R, Gnanamanickam SS, Levy M (1996) Studies on genetic diversity of Pyricularia grisea: a molecular approach for management of rice blast. In: Khush GS (ed) Rice Genetics III. International Rice Research Institute, Manila, The Philippines, pp 958–962
Steele KA, Price AH, Shashidhar HE, Witcombe JR (2006) Marker-assisted selection to introgress rice QTLs controlling root traits into an Indian upland rice variety. Theor Appl Genet 112:208–221
Tacconi G, Baldassarre VC, Lanzanova O, Faivre-Rampant S, Cavigiolo S, Urso E, Lupotto GV (2010) Polymorphism analysis of genomic regions associated with broad-spectrum effective blast resistance genes for marker development in rice. Mol Breed 26:595–617
Tharreau D, Fudal I, Andriantsimialona D, Santoso D, Utami D, Fournier E, LeBrun MH, Notteghem JL (2009) World population structure and migration of the rice blast fungus Magnaporthe oryzae. Theor Appl Genet 76:815–829
TNAU. 2012. Crop Production Guide 2012, Tamil Nadu agricultural University. agritech.tnau.ac.in/pdf/2013/CPG%202012.pdf
Tuberosa R, Salvi S, Sanguineti MC, Landi P, Maccaferri M, Conti S (2002) Mapping QTLs regulating morpho-physiological traits and yield: case studies, shortcomings and perspectives in drought-stressed maize. Ann Bot (Lond) 89:941–963
Wang GL, Mackill DJ, Bonman JM, McCouch SR, Champoux MC, Nelson RJ (1994) RFLP mapping of genes conferring complete and partial resistance to blast in a durably resistant rice cultivar. Genetics 136:1421–1434
Wu JL, Fan YY, Li DB, Zheng KL, Leung H, Zhuang JY (2005) Genetic control of rice blast resistance in the durably resistant cultivar Gumei 2 multiple isolates. Theor Appl Genet 111:50–56
Yanoria TMJ, Koide Y, Fukuta Y, Imbe T, Tsunematsu H, Kato H, Ebron L, Nguyen TN, Kobayashi N (2011) A set of near-isogenic lines of Indica-type rice variety CO 39 as differential varieties for blast resistance. Mol Breed 27:357–373
Yoshimura S, Yoshimura A, Iwata N, McCouch SR, Abenes ML, Baraoidan MR, Mew TW, Nelson RJ (1995) Tagging and combining bacterial blight resistance genes in rice using RAPD and RFLP markers. Mol Breed 1:375–387
Zeigler RS, Tohme J, Nelson R, Levy M, Correa-Victoria FJ (1994) Lineage exclusion a proposal for linking blast population analysis to resistance breeding. In: Rice Blast Disease. CAB International. IRRI, Wallingford, U.K, pp 267–292
Zhou T, Wang Y, Chen JQ, Araki H, Jing Z, Jiang K, Shen J, Tian D (2004) Genome-wide identification of NBS genes in japonica rice reveals significant expansion of divergent non-TIR NBS-LRR genes. Mol Genet Genomics 271:402–415
Acknowledgments
We thank Indian Council of Agriculture Research (ICAR) for Senior Research fellowship awarded to the first author. We thank Centre for Plant Molecular Biology and Biotechnology (CPMBB), Centre for Plant Protection Studies (CPPS) and Hybrid Rice Evaluation Centre, Gudalur (HREC), TNAU for providing facilities to support this work. We are grateful to Dr. N. Sarla, ICAR National Professor, Directorate of Rice Research (DRR) for a critical review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(DOCX 15 kb)
Supplementary Table 2
(DOCX 26 kb)
Rights and permissions
About this article
Cite this article
Divya, B., Robin, S., Rabindran, R. et al. Resistance reaction of gene introgressed lines against rice blast (Pyricularia oryzae) disease. Australasian Plant Pathol. 43, 177–191 (2014). https://doi.org/10.1007/s13313-013-0262-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-013-0262-7