Abstract
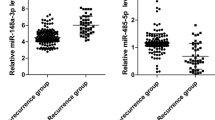
Prostate cancer (PCa) has become a prevalent malignant disease in males globally. Accumulating data suggested that hsa-microRNAs (miRNAs) could be potential biomarkers for tumor diagnosis due to their important roles in the cell cycle. This study investigated the diagnostic and prognostic values of hsa-miR-203 and hsa-miR-30c in PCa tissues. There were 44 pathologically confirmed PCa patients who were enrolled in this study. Tissue samples were collected from both tumor tissues and adjacent normal tissues. RNA was extracted and the expression levels of hsa-miR-203 and hsa-miR-30c in tumor and normal tissues were compared. The receiver operating characteristic (ROC) curves were plotted to evaluate the reliability of hsa-miR-203 and hsa-miR-30c in detecting PCa. All subjects in this study were followed up by 36 months, and the Kaplan-Meier method was conducted to investigate the survival status of PCa patients. The average relative expressions of hsa-miR-203 and hsa-miR-30c in tumor tissues were significantly different from those in adjacent normal tissues (P < 0.001), and the predictive power of the two hsa-miRNAs for PCa prognosis was reliable. Besides that, the average survival times of low-hsa-miR-30c and high-hsa-miR-203 groups were significantly lower than those of the corresponding groups with the log-rank P of 0.015 and 0.023, respectively. In summary, our study suggested that both hsa-miR-203 and hsa-miR-30c are potential biomarkers for detection and prognosis of PCa.




Similar content being viewed by others
References
Huang Z, Huang D, Ni S, Peng Z, Sheng W, Du X. Plasma microRNAs are promising novel biomarkers for early detection of colorectal cancer. Int J Cancer J Int du cancer. 2010;127:118–26.
Casanova-Salas I, Rubio-Briones J, Calatrava A, Mancarella C, Masia E, Casanova J, Fernandez-Serra A, Rubio L, Ramirez-Backhaus M, Arminan A, Dominguez-Escrig J, Martinez F, Garcia-Casado Z, Scotlandi K, Vicent MJ, Lopez-Guerrero JA. Identification of miR-187 and miR-182 as biomarkers of early diagnosis and prognosis in patients with prostate cancer treated with radical prostatectomy. J Urol 2014
Peyromaure EM, Mao K, Sun Y, Xia S, Jiang N, Zhang S, et al. A comparative study of prostate cancer detection and management in China and in France. Can J Urol. 2009;16:4472–7.
Roberts WW, Bergstralh EJ, Blute ML, Slezak JM, Carducci M, Han M, et al. Contemporary identification of patients at high risk of early prostate cancer recurrence after radical retropubic prostatectomy. Urology. 2001;57:1033–7.
Xiong SW, Lin TX, Xu KW, Dong W, Ling XH, Jiang FN, et al. MicroRNA-335 acts as a candidate tumor suppressor in prostate cancer. Pathol Oncol Res. 2013;19:529–37.
Sboner A, Demichelis F, Calza S, Pawitan Y, Setlur SR, Hoshida Y, et al. Molecular sampling of prostate cancer: a dilemma for predicting disease progression. BMC Med Genomics. 2010;3:8.
Catalona WJ, Partin AW, Finlay JA, Chan DW, Rittenhouse HG, Wolfert RL, et al. Use of percentage of free prostate-specific antigen to identify men at high risk of prostate cancer when PSA levels are 2.51 to 4 ng/ml and digital rectal examination is not suspicious for prostate cancer: an alternative model. Urology. 1999;54:220–4.
Emiliozzi P, Longhi S, Scarpone P, Pansadoro A, DePaula F, Pansadoro V. The value of a single biopsy with 12 transperineal cores for detecting prostate cancer in patients with elevated prostate specific antigen. J Urol. 2001;166:845–50.
Roehl KA, Antenor JA, Catalona WJ. Robustness of free prostate specific antigen measurements to reduce unnecessary biopsies in the 2.6 to 4.0 ng./ml. range. J Urol. 2002;168:922–5.
Danila DC, Heller G, Gignac GA, Gonzalez-Espinoza R, Anand A, Tanaka E, et al. Circulating tumor cell number and prognosis in progressive castration-resistant prostate cancer. Clin Cancer Res. 2007;13:7053–8.
de Bono JS, Scher HI, Montgomery RB, Parker C, Miller MC, Tissing H, et al. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin Cancer Res. 2008;14:6302–9.
Gonzales JC, Fink LM, Goodman Jr OB, Symanowski JT, Vogelzang NJ, Ward DC. Comparison of circulating microRNA 141 to circulating tumor cells, lactate dehydrogenase, and prostate-specific antigen for determining treatment response in patients with metastatic prostate cancer. Clin Genitourin Cancer. 2011;9:39–45.
Mavridis K, Stravodimos K, Scorilas A. Downregulation and prognostic performance of microRNA 224 expression in prostate cancer. Clin Chem. 2013;59:261–9.
Loeb S, Partin AW. Review of the literature: PCA3 for prostate cancer risk assessment and prognostication. Rev Urol. 2011;13:e191–195.
Yaman Agaoglu F, Kovancilar M, Dizdar Y, Darendeliler E, Holdenrieder S, Dalay N, et al. Investigation of miR-21, miR-141, and miR-221 in blood circulation of patients with prostate cancer. Tumour Biol. 2011;32:583–8.
Haj-Ahmad TA, Abdalla MA, Haj-Ahmad Y. Potential urinary miRNA biomarker candidates for the accurate detection of prostate cancer among benign prostatic hyperplasia patients. J Cancer Educ. 2014;5:182–91.
Chen X, Ba Y, Ma L, Cai X, Yin Y, Wang K, et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008;18:997–1006.
Casanova-Salas I, Rubio-Briones J, Fernandez-Serra A, Lopez-Guerrero JA. MiRNAs as biomarkers in prostate cancer. Clin Transl Oncol. 2012;14:803–11.
Deng Y, Huang Z, Xu Y, Jin J, Zhuo W, Zhang C, et al. MiR-215 modulates gastric cancer cell proliferation by targeting RB1. Cancer Lett. 2014;342:27–35.
Yuxia M, Zhennan T, Wei Z. Circulating miR-125b is a novel biomarker for screening non-small-cell lung cancer and predicts poor prognosis. J Cancer Res Clin Oncol. 2012;138:2045–50.
Mar-Aguilar F, Luna-Aguirre CM, Moreno-Rocha JC, Araiza-Chavez J, Trevino V, Rodriguez-Padilla C, et al. Differential expression of miR-21, miR-125b and miR-191 in breast cancer tissue. Asia Pac J Clin Oncol. 2013;9:53–9.
Guo F, Tian J, Lin Y, Jin Y, Wang L, Cui M. Serum microrna-92 expression in patients with ovarian epithelial carcinoma. J Int Med Res. 2013;41:1456–61.
Gottardo F, Liu CG, Ferracin M, Calin GA, Fassan M, Bassi P, et al. Micro-rna profiling in kidney and bladder cancers. Urol Oncol. 2007;25:387–92.
Lodes MJ, Caraballo M, Suciu D, Munro S, Kumar A, Anderson B. Detection of cancer with serum miRNAs on an oligonucleotide microarray. PLoS One. 2009;4, e6229.
Saini S, Majid S, Dahiya R. Diet, microRNAs and prostate cancer. Pharm Res. 2010;27:1014–26.
He HC, Zhu JG, Chen XB, Chen SM, Han ZD, Dai QS, et al. MicroRNA-23b downregulates peroxiredoxin III in human prostate cancer. FEBS Lett. 2012;586:2451–8.
Viticchie G, Lena AM, Latina A, Formosa A, Gregersen LH, Lund AH, et al. MiR-203 controls proliferation, migration and invasive potential of prostate cancer cell lines. Cell Cycle. 2011;10:1121–31.
Moltzahn F, Olshen AB, Baehner L, Peek A, Fong L, Stoppler H, et al. Microfluidic-based multiplex qRT-PCR identifies diagnostic and prognostic microRNA signatures in the sera of prostate cancer patients. Cancer Res. 2011;71:550–60.
Ling XH, Han ZD, Xia D, He HC, Jiang FN, Lin ZY, et al. MicroRNA-30c serves as an independent biochemical recurrence predictor and potential tumor suppressor for prostate cancer. Mol Biol Rep. 2014;41:2779–88.
Qu KZ, Zhang K, Li H, Afdhal NH, Albitar M. Circulating microRNAs as biomarkers for hepatocellular carcinoma. J Clin Gastroenterol. 2011;45:355–60.
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci U S A. 2008;105:10513–8.
Benaich N, Woodhouse S, Goldie SJ, Mishra A, Quist SR, Watt FM. Rewiring of an epithelial differentiation factor, miR-203, to inhibit human squamous cell carcinoma metastasis. Cell Rep. 2014;9:104–17.
Noguchi S, Kumazaki M, Mori T, Baba K, Okuda M, Mizuno T, Akao Y. Analysis of microrna-203 function in CREB/MITF/RAB27a pathway: comparison between canine and human melanoma cells. Vet Comp Oncol 2014
Boll K, Reiche K, Kasack K, Morbt N, Kretzschmar AK, Tomm JM, et al. MiR-130a, miR-203 and miR-205 jointly repress key oncogenic pathways and are downregulated in prostate carcinoma. Oncogene. 2013;32:277–85.
Wellner U, Schubert J, Burk UC, Schmalhofer O, Zhu F, Sonntag A, et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat Cell Biol. 2009;11:1487–95.
Fang Y, Shen H, Cao Y, Li H, Qin R, Chen Q, et al. Involvement of miR-30c in resistance to doxorubicin by regulating YWHAZ in breast cancer cells. Braz J Med Biol Res. 2014;47:60–9.
Kong X, Xu X, Yan Y, Guo F, Li J, Hu Y, et al. Estrogen regulates the tumour suppressor miRNA-30c and its target gene, MTA-1, in endometrial cancer. PLoS One. 2014;9, e90810.
Ren Q, Liang J, Wei J, Basturk O, Wang J, Daniels G, et al. Epithelial and stromal expression of miRNAs during prostate cancer progression. Am J Transl Res. 2014;6:329–39.
Kim JH, Lee JM, Nam HJ, Choi HJ, Yang JW, Lee JS, et al. SUMOylation of pontin chromatin-remodeling complex reveals a signal integration code in prostate cancer cells. Proc Natl Acad Sci U S A. 2007;104:20793–8.
Wang XS, Shankar S, Dhanasekaran SM, Ateeq B, Sasaki AT, Jing X, et al. Characterization of KRAS rearrangements in metastatic prostate cancer. Cancer Discovery. 2011;1:35–43.
Wang H, Fan L, Wei J, Weng Y, Zhou L, Shi Y, et al. Akt mediates metastasis-associated gene 1 (MTA1) regulating the expression of e-cadherin and promoting the invasiveness of prostate cancer cells. PLoS One. 2012;7, e46888.
Kao CJ, Martiniez A, Shi XB, Yang J, Evans CP, Dobi A, et al. MiR-30 as a tumor suppressor connects EGF/Src signal to ERG and EMT. Oncogene. 2014;33:2495–503.
Zhang K, Dai L, Zhang B, Xu X, Shi J, Fu L, Chen X, Li J, Bai Y. MiR-203 is a direct transcriptional target of E2F1 and causes G1 arrest in esophageal cancer cells. J Cell Physiol 2014
Zhong K, Chen K, Han L, Li B. MicroRNA-30b/c inhibits non-small cell lung cancer cell proliferation by targeting rab18. BMC Cancer. 2014;14:703.
Acknowledgments
This study is supported by the National Science Foundation of China (NSFC 81570053), Shanghai Science and Technology Commission Foundation of Key Medical Research (034119868 and 09411951600), and Research Foundation of Shanghai Municipal Health Bureau (20134034).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
None
Additional information
Ziling Huang and Long Zhang are first co-authors.
Rights and permissions
About this article
Cite this article
Huang, Z., Zhang, L., Yi, X. et al. Diagnostic and prognostic values of tissue hsa-miR-30c and hsa-miR-203 in prostate carcinoma. Tumor Biol. 37, 4359–4365 (2016). https://doi.org/10.1007/s13277-015-4262-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4262-9