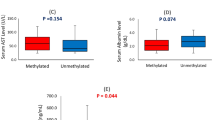
Abstract
Both hepatitis B virus (HBV) and gene methylation play important roles in hepatocarcinogenesis. However, their association between HBV infection and gene methylation is not fully understood. Cell cycle control involving RB1 gene-related cell inhibitors is one of the main regulatory pathways were reported to be altered in hepatocellular carcinoma (HCC). The purpose of this research is to assess the methylation status of p14 ARF and INK4 gene family (p14 ARF, p15 INK4B, p16 INK4A, and p18 INK4C) in HCC with HBV infection and HCC without it, and discuss possible role of HBV-induced hypermethylation in the mechanism of hepatocarcinogenesis. Methylation status of RB, p14 ARF, and INK4 gene family in 64 case of HCC with HBV infection and 24 cases without it were detected by methylation-specific polymerase chain reaction, and HBV-DNA of the plasma were detected by quantitative real-time polymerase chain reaction. p14 ARF, p15 INK4B, p16 INK4A, and RB hypermethylation were observed in 30 (34.1 %), 50 (56.8 %), 62 (70.5 %), and 24(27.3 %) of 88 hepatocellular carcinomas, respectively. Methylation frequencies of them between HCC with HBV infection and HCC without it were 43.8 % versus 8.3 % (p14 ARF), 68.9 % versus 25 % (p15 INK4B), 90.6 % versus 16.7 % ( p16 INK4A), and 28.1 % versus 25 % (RB), respectively. In HBV-associated HCC, the numbers of methylated genes were also more than HCC without virus infection, more than two methylated genes were seen in 48 of 64 (75 %) cases; more than three methylated genes were found in 32 of 64 (50 %); correspondently, no one case has more than two genes methylated. p18 INK4C methylation product was not found in cancerous or non-cancerous tissues of 88 HCC. HBV infection is associated with p14 ARF, p15 INK4B, p16 INK4A, and RB gene methylation (P = 0.048, 0.035, 0.02); HBV-DNA replication is associated with p14 ARF, p15 INK4B, p16 INK4A, and RB gene methylation (P = 0.048, 0.035, 0.02); high rate of p14 ARF, p15 INK4B, and p16 INK4A in HCC with HBV infection suggests that HBV-induced hypermethylation may be one of the mechanisms of HBV involved in hepatocellular carcinogenesis.




Similar content being viewed by others
References
Hanto DW, Curry MP, Sun M, et al. Hepatocellular carcinoma (HCC) in a patient with hepatitis B virus (HBV) infection. Am J Transplant. 2012;12:786–7.
Lee JH, Han KH, Lee JM, et al. Impact of hepatitis B virus (HBV) x gene mutations on hepatocellular carcinoma development in chronic HBV infection. Clin Vaccine Immunol. 2011;18:914–21.
Kaur P, Paliwal A, Durantel D, et al. DNA methylation of hepatitis B virus (HBV) genome associated with the development of hepatocellular carcinoma and occult HBV infection. J Infect Dis. 2010;202:700–4.
Christman JK. 5-Azacytidine and 5-aza-2'-deoxycytidine as inhibitors of DNA methylation: mechanistic studies and their implications for cancer therapy. Oncogene. 2002;21:5483–95.
Lee JY, Locarnini S. Hepatitis B virus: pathogenesis, viral intermediates, and viral replication. Clin Liver Dis. 2004;8(2):301–20.
Han HJ, Jung EY, Lee WJ, et al. Cooperative repression of cyclin-dependent kinase inhibitor p21 gene expression by hepatitis B virus X protein and hepatitis C virus core protein. FEBS Lett. 2002;518:169–72.
Yoshida I, Oka K, Hidajat R, et al. Inhibition of p21/Waf1/Cip1/Sdi1 expression by hepatitis C virus core protein. Microbiol Immunol. 2001;45:689–97.
Fang D, Guo Y, Zhu Z, et al. Silence of p15 expression by RNAi enhances cisplatin resistance in hepatocellular carcinoma cells. Bosn J Basic Med Sci. 2012;12:4–9.
Kanellou P, Zaravinos A, Zioga M, et al. Deregulation of the tumour suppressor genes p14(ARF), p15(INK4b), p16(INK4a) and p53 in basal cell carcinoma. Br J Dermatol. 2009;160:1215–21.
Moad AI, Lan TM, Kaur G, et al. Immunohistochemical determination of the P15 protein expression in cutaneous squamous cell carcinoma. J Cutan Pathol. 2009;36:183–9.
Zhang Y, Xiong Y, Yarbrough WG. ARF promotes MDM2 degradation and stabilizes p53: ARF-INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways. Cell. 1998;92:725–34.
Xiaofang L, Kun T, Shaoping Y, et al. Correlation between promoter methylation of p14(ARF), TMS1/ASC, and DAPK, and p53 mutation with prognosis in cholangiocarcinoma. World J Surg Oncol. 2012;10:5.
Verdoodt B, Sommerer F, Palisaar RJ, et al. Inverse association of p16 INK4a and p14 ARF methylation of the CDKN2a locus in different Gleason scores of prostate cancer. Prostate Cancer Prostatic Dis. 2011;14:295–301.
Zhong S, Tang MW, Yeo W, et al. Silencing of GSTP1 gene by CpG island DNA hypermethylation in HBV-associated hepatocellular carcinomas. Clin Cancer Res. 2002;8:1087–92.
Sherr CJ. Cancer cell cycles. Science. 1996;274:1672–7.
Barzily-Rokni M, Friedman N, Ron-Bigger S, et al. Synergism between DNA methylation and macroH2A1 occupancy in epigenetic silencing of the tumor suppressor gene p16(CDKN2A). Nucleic Acids Res. 2011;39:1326–35.
Nakata S, Sugio K, Uramoto H, et al. The methylation status and protein expression of CDH1, p16(INK4A), and fragile histidine triad in nonsmall cell lung carcinoma: epigenetic silencing, clinical features, and prognostic significance. Cancer. 2006;106:2190–9.
Csepregi A, Ebert MP, Röcken C, et al. Promoter methylation of CDKN2A and lack of p16 expression characterize patients with hepatocellular carcinoma. BMC Cancer. 2010;10:317.
Zhang C, Guo X, Jiang G, et al. CpG island methylator phenotype association with upregulated telomerase activity in hepatocellular carcinoma. Int J Cancer. 2008;123:998–1004.
Formeister EJ, Tsuchiya M, Fujii H, et al. Comparative analysis of promoter methylation and gene expression endpoints between tumorous and non-tumorous tissues from HCV-positive patients with hepatocellular carcinoma. Mutat Res. 2010;692:26–33.
Jin M, Piao Z, Kim NG, et al. p16 is a major inactivation target in hepatocellular carcinoma. Cancer. 2000;89:60–8.
Lin YW, Chen CH, Huang GT, et al. Infrequent mutations and no methylation of CDKN2A (P16/MTS1) and CDKN2B (p15/MTS2) in hepatocellular carcinoma in Taiwan. Eur J Cancer. 1998;34:1789–95.
Jicai Z, Zongtao Y, Jun L, et al. Persistent infection of hepatitis B virus is involved in high rate of p16 methylation in hepatocellular carcinoma. Mol Carcinog. 2006;45:530–6.
Narimatsu T, Tamori A, Koh N, et al. p16 promoter hypermethylation in human hepatocellular carcinoma with or without hepatitis virus infection. Intervirology. 2004;47:26–31.
Xing C, Wang QF, Li B, et al. Methylation and expression analysis of tumor suppressor genes p15 and p16 in benzene poisoning. Chem Biol Interact. 2010;184:306–9.
Jha AK, Nikbakht M, Jain V, et al. p16(INK4a) and p15(INK4b) gene promoter methylation in cervical cancer patients. Oncol Lett. 2012;3:1331–5.
Wong IH, Lo YM, Yeo W, et al. Frequent p15 promoter methylation in tumor and peripheral blood from hepatocellular carcinoma patients. Clin Cancer Res. 2000;6:3516–21.
Chen MN, Mao Q, Liu YH, et al. Methylation and expression analysis of p16(INK4a) and RB genes in meningiomas. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 2004;21:277–9.
Radpour R, Barekati Z, Haghighi MM, et al. Correlation of telomere length shortening with promoter methylation profile of p16/Rb and p53/p21 pathways in breast cancer. Mod Pathol. 2010;23:763–72.
Geddert H, zur Hausen A, Gabbert HE, et al. EBV-infection in cardiac and non-cardiac gastric adenocarcinomas is associated with promoter methylation of p16, p14 and APC, but not hMLH1. Cell Oncol (Dordr). 2011;34:209–14.
Ferrasi AC, Pinheiro NA, Rabenhorst SH, et al. Helicobacter pylori and EBV in gastric carcinomas: methylation status and microsatellite instability. World J Gastroenterol. 2010;16:312–9.
Koyuncu OO, Dobner T. Arginine methylation of human adenovirus type 5 L4 100-kilodalton protein is required for efficient virus production. J Virol. 2009;83:4778–90.
Mikovits JA, Young HA, Vertino P, et al. Infection with human immunodeficiency virus type 1 upregulates DNA methyltransferase, resulting in de novo methylation of the gamma interferon (IFN-gamma) promoter and subsequent downregulation of IFN-gamma production. Mol Cell Biol. 1998;18:5166–77.
Kang GH, Lee S, Kim WH, et al. Epstein-barr virus-positive gastric carcinoma demonstrates frequent aberrant methylation of multiple genes and constitutes CpG island methylator phenotype-positive gastric carcinoma. Am J Pathol. 2002;160:787–94.
Yang B, Guo M, Herman JG, et al. Aberrant promoter methylation profiles of tumor suppressor genes in hepatocellular carcinoma. Am J Pathol. 2003;163:1101–7.
Acknowledgments
This work funded by Research Project of Department of Education of Hubei Province (Z20082401).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, JC., Gao, B., Yu, ZT. et al. Promoter hypermethylation of p14 ARF, RB, and INK4 gene family in hepatocellular carcinoma with hepatitis B virus infection. Tumor Biol. 35, 2795–2802 (2014). https://doi.org/10.1007/s13277-013-1372-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-1372-0