Abstract
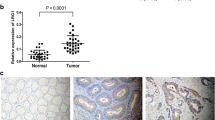
In order to find the correlation between transgelin gene (TAGLN) and colorectal carcinoma occurrence, we investigated the expression of TAGLN in colorectal carcinoma tissue samples and colorectal carcinoma LoVo cells. Meanwhile, the effects of TAGLN on the characteristics of LoVo cells were also examined. The expressions of TAGLN in colorectal carcinoma tissues, adjacent normal tissues, and LoVo cells were detected by the Western blot method. The recombinant plasmid pcDNA3.1-TAGLN was established and transfected into LoVo cells with the help of Lipofectamine™ 2000. At the same time, the TAGLN siRNA was transfected into LoVo cells in another group. Forty-eight hours later, the expressions of TAGLN in all groups were assayed by Western blot, and the cell viability was analyzed by MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide) assay. The cell cycle and cell apoptosis were examined by flow cytometry, and the cell invasive ability was analyzed by Transwell invasion experiment. The effect of TALGN on the expression of matrix metalloproteinase 9 (MMP9) was detected by Western blot. Western blot analysis showed that the expressions of TALGN in colorectal carcinoma tissues and LoVo cells were significantly decreased compared with colorectal carcinoma adjacent normal tissues (p < 0.01). In the overexpression or RNAi experiments, the plasmid pcDNA3.1-TAGLN significantly enhanced TALGN expression (p < 0.01), and TAGLN siRNA significantly decreased TAGLN expression (p < 0.01) in LoVo cells 48 h after transfection. In addition, MTT assay indicated that the cell viability of LoVo cells in the pcDNA3.1-TAGLN transfection group was significantly lower than that in the untransfected control group (p < 0.05). Furthermore, the overexpression of TAGLN significantly lowered the cell proliferation index (p < 0.05) and improved cell apoptosis (p < 0.01) in LoVo cells. In Transwell invasive experiments, the cell number, which had migrated through the chamber membrane, significantly decreased in the pcDNA3.1-TAGLN transfection group (p < 0.05) and significantly increased in the TAGLN knockdown group (p < 0.05) compared to the untransfected control group. At the same time, the expression of MMP9 was notably inhibited in the pcDNA3.1-TAGLN transfection group (p < 0.01). The expressions of TAGLN were inhibited in colorectal carcinoma tissues and colorectal carcinoma LoVo cells. The study also demonstrated that TAGLN could attenuate the proliferation and invasive ability of LoVo cells and enhance LoVo cell apoptosis. Furthermore, the expression of MMP9 was also inhibited by TAGLN. All these results could bring us a new perspective for biological therapy in colorectal carcinoma.







Similar content being viewed by others
References
Huang C, Huang RX, Xiang P, Qiu ZJ. Current research status of endoscopic submucosal dissection for colorectal neoplasms. Clin Invest Med. 2012;35(4):E158.
Wan DS. Epidemiological trend and control strategy of colorectal cancer in China. Zhonghua Zhong Liu Za Zhi. 2011;33(7):481–3.
Mitrovic B, Schaeffer DF, Riddell RH, Kirsch R. Tumor budding in colorectal carcinoma: time to take notice. Mod Pathol. 2012;25(10):1315–25.
Wang CC, Li J. An update on chemotherapy of colorectal liver metastases. World J Gastroenterol. 2012;18(1):25–33.
Moszkowicz D, Cauchy F, Dokmak S, Belghiti J. Routine pedicular lymphadenectomy for colorectal liver metastases. J Am Coll Surg. 2012;214(6):e39–45.
Fleming M, Ravula S, Tatishchev SF, Wang HL. Colorectal carcinoma: pathologic aspects. J Gastrointest Oncol. 2012;3(3):153–73.
Caruso R, Parisi A, Bonanno A, Paparo D, Quattrocchi E, Branca G, et al. Histologic coagulative tumour necrosis as a prognostic indicator of aggressiveness in renal, lung, thyroid and colorectal carcinomas: a brief review. Oncol Lett. 2012;3(1):16–8.
Aslam MI, Patel M, Singh B, Jameson JS, Pringle JH. MicroRNA manipulation in colorectal cancer cells: from laboratory to clinical application. J Transl Med. 2012;10(1):128.
Al-Sohaily S, Biankin A, Leong R, Kohonen-Corish M, Warusavitarne J. Molecular pathways in colorectal cancer. J Gastroenterol Hepatol. 2012;27(9):1423–31.
Assinder SJ, Stanton JA, Prasad PD. Transgelin: an actin-binding protein and tumour suppressor. Int J Biochem Cell Biol. 2009;41(3):482–6.
Mericskay M, Li Z, Paulin D. Transcriptional regulation of the desmin and SM22 genes in vascular smooth muscle cells. Curr Top Pathol. 1999;93:7–17.
Shi JH, Wen JK, Han M. The role of SM22 alpha in cytoskeleton organization and vascular remodeling. Sheng Li Ke Xue Jin Zhan. 2006;37(3):211–5.
Morgan KG, Gangopadhyay SS. Invited review: cross-bridge regulation by thin filament-associated proteins. J Appl Physiol. 2001;91(2):953–62.
Pankajakshan D, Makinde TO, Gaurav R, Del Core M, Hatzoudis G, Pipinos I, et al. Successful transfection of genes using AAV-2/9 vector in swine coronary and peripheral arteries. J Surg Res. 2012;175(1):169–75.
Shen J, Yang M, Jiang H, Ju D, Zheng JP, Xu Z, et al. Arterial injury promotes medial chondrogenesis in Sm22 knockout mice. Cardiovasc Res. 2011;90(1):28–37.
Dos Santos Hidalgo G, Meola J, Rosa ESJC, de Paz CC P, Ferriani RA. TAGLN expression is deregulated in endometriosis and may be involved in cell invasion, migration, and differentiation. Fertil Steril. 2011;96(3):700–3.
Li Q, Zhu F, Chen P. MiR-7 and miR-218 epigenetically control tumor suppressor genes RASSF1A and Claudin-6 by targeting HoxB3 in breast cancer. Biochem Biophys Res Commun. 2012;424(1):28–33.
Xiao L, Ma ZL, Li X, Lin QX, Que HP, Liu SJ. cDNA microarray analysis of spinal cord injury and regeneration related genes in rat. Sheng Li Xue Bao. 2005;57(6):705–13.
Worthington J, Bertani M, Chan HL, Gerrits B, Timms JF. Transcriptional profiling of ErbB signalling in mammary luminal epithelial cells—interplay of ErbB and IGF1 signalling through IGFBP3 regulation. BMC Cancer. 2010;10:490.
Knapinska A, Fields GB. Chemical biology for understanding matrix metalloproteinase function. Chembiochem. 2012;13(14):2002–20.
Liu D, Duan W, Guo H, Xu X, Bai Y. Meta-analysis of associations between polymorphisms in the promoter regions of matrix metalloproteinases and the risk of colorectal cancer. Int J Colorectal Dis. 2011;26(9):1099–105.
Peng WJ, Zhang JQ, Wang BX, Pan HF, Lu MM, Wang J. Prognostic value of matrix metalloproteinase 9 expression in patients with non-small cell lung cancer. Clin Chim Acta. 2012;413(13–14):1121–6.
Bauvois B. New facets of matrix metalloproteinases MMP-2 and MMP-9 as cell surface transducers: outside-in signaling and relationship to tumor progression. Biochim Biophys Acta. 2012;1825(1):29–36.
Nair RR, Solway J, Boyd DD. Expression cloning identifies transgelin (SM22) as a novel repressor of 92-kDa type IV collagenase (MMP-9) expression. J Biol Chem. 2006;281(36):26424–36.
Daly ME, Kapp DS, Maxim PG, Welton ML, Tran PT, Koong AC, et al. Orthovoltage intraoperative radiotherapy for locally advanced and recurrent colorectal cancer. Dis Colon Rectum. 2012;55(6):695–702.
Guo Z, Jia X, Liu JP, Liao J, Yang Y. Herbal medicines for advanced colorectal cancer. Cochrane Database Syst Rev. 2012;5:CD004653.
Gespach C. Guidance for life, cell death, and colorectal neoplasia by netrin dependence receptors. Adv Cancer Res. 2012;114:87–186.
He K, Jin K, Wang H, Teng L. Anti-angiogenic therapy for colorectal cancer: on the way to getting better! Hepatogastroenterology. 2012;59(116):1113–7.
Weber GF, Rosenberg R, Murphy JE, Meyer zum Buschenfelde C, Friess H. Multimodal treatment strategies for locally advanced rectal cancer. Expert Rev Anticancer Ther. 2012;12(4):481–94.
Dewdney A, Cunningham D. Toward the non-surgical management of locally advanced rectal cancer. Curr Oncol Rep. 2012;14(3):267–76.
Ibrahim SA, Yip GW, Stock C, Pan JW, Neubauer C, Poeter M, et al. Targeting of syndecan-1 by microRNA miR-10b promotes breast cancer cell motility and invasiveness via a Rho-GTPase- and E-cadherin-dependent mechanism. Int J Cancer. 2012;131(6):E884–96.
Herencia C, Martinez-Moreno JM, Herrera C, Corrales F, Santiago-Mora R, Espejo I, et al. Nuclear translocation of beta-catenin during mesenchymal stem cells differentiation into hepatocytes is associated with a tumoral phenotype. PLoS One. 2012;7(4):e34656.
Thompson O, Moghraby JS, Ayscough KR, Winder SJ. Depletion of the actin bundling protein SM22/transgelin increases actin dynamics and enhances the tumourigenic phenotypes of cells. BMC Cell Biol. 2012;13:1.
Daniel C, Ludke A, Wagner A, Todorov VT, Hohenstein B, Hugo C. Transgelin is a marker of repopulating mesangial cells after injury and promotes their proliferation and migration. Lab Invest. 2012;92(6):812–26.
Kim TR, Moon JH, Lee HM, Cho EW, Paik SG, Kim IG. SM22alpha inhibits cell proliferation and protects against anticancer drugs and gamma-radiation in HepG2 cells: involvement of metallothioneins. FEBS Lett. 2009;583(20):3356–62.
Zhang ZW, Yang ZM, Zheng YC, Chen ZD. Transgelin induces apoptosis of human prostate LNCaP cells through its interaction with p53. Asian J Androl. 2010;12(2):186–95.
Kawakami K, Enokida H, Chiyomaru T, Tatarano S, Yoshino H, Kagara I, et al. The functional significance of miR-1 and miR-133a in renal cell carcinoma. Eur J Cancer. 2012;48(6):827–36.
Mansouri D, McMillan DC, Crighton EM, Horgan PG. Screening for colorectal cancer: what is the impact on the determinants of outcome? Crit Rev Oncol Hematol. 2012. doi:10.1016/j.critrevonc.2012.08.006.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Q., Shi, R., Wang, Y. et al. TAGLN suppresses proliferation and invasion, and induces apoptosis of colorectal carcinoma cells. Tumor Biol. 34, 505–513 (2013). https://doi.org/10.1007/s13277-012-0575-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0575-0