Abstract
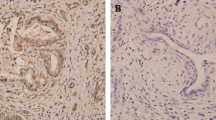
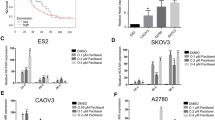
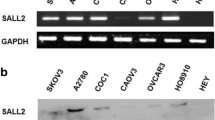
The objective of this study was to examine the expression levels of RE1-silencing transcription factor (REST) and class III β-tubulin (TUBB3) in ovarian cancer and to determine if there is a correlation between their expression and resistance to chemotherapy in ovarian cancer. The protein expression of REST and TUBB3 in ovarian cancer was detected by Western blot analysis. REST expression was inhibited by small interfering ribonucleic acid (siRNA) in human ovarian cancer cell lines. The levels of REST and TUBB3 protein expression were detected by immunohistochemistry. The relationship between REST and TUBB3 expression and chemotherapy resistance, clinicopathological parameters, and prognosis of ovarian cancer was then determined. The present study found that REST was more highly expressed in the ovarian SKOV3 carcinoma cell line compared to the paclitaxel-resistant ovarian cancer cell line, SKOV3/TAX (P = 0.01). In contrast, TUBB3 was more highly expressed in SKOV3/TAX cells compared to SKOV3 cells (P = 0.01). After REST siRNA interference, TUBB3 expression increased in SKOV3 cells. REST expression was significantly higher in the paclitaxel-sensitive group compared to the paclitaxel-resistant group (70.7 % vs. 37.0 %, P < 0.05). However, TUBB3 expression was significantly lower in the paclitaxel-sensitive group compared to the paclitaxel-resistant group (47.6 % vs. 77.8 %, P < 0.05). Notably, REST was more highly expressed in TUBB3-negative cases than TUBB3-positive cases (P < 0.05). Univariate analyses indicated that both REST and TUBB3 expression were unrelated to tumor differentiation, histological type, and clinical stage (all P > 0.05). According to the Cox regression model, negative REST and positive TUBB3 protein expression was detected as independent prognostic factors (P = 0.003 and P = 0.005, respectively). REST and TUBB3 protein may be potential biomarkers for chemoresistance and prognosis in ovarian cancer.





Similar content being viewed by others
References
Pylvs M, Puistola U, Laatio L, Kauppila S, Karihtala P. Elevated serum 8-OHdG is associated with poor prognosis in epithelial ovarian cancer. Anticancer Res. 2011;31(4):1411–5.
Morgan Jr RJ, Alvarez RD, Armstrong DK, et al. NCCN clinical practice guidelines in oncology: epithelial ovarian cancer. J Natl Compr Canc Netw. 2011;9(1):82–113. 374:1371–82.
Bagnato A, Rosanò L. Understanding and overcoming chemoresistance in ovarian cancer: emerging role of the endothelin axis. Curr Oncol. 2012;19(1):36–8.
Mori N, Stein R, Sigmund O, et al. A cell type-preferred silencer element that controls the neural-specific expression of the SCG10 gene. Neuron. 1990;4(4):583–59421.
Jones FS, Meech R. Knockout of REST/NRSF shows that the protein is a potent repressor of neuronally expressed genes in non-neural tissues. Bioessays. 1999;21(5):372–6.
Zhao JJ, Gjoerup OV, Subramanian RR, et al. Human mammary epithelial cell transformation through the activation of phosphatidylinositol 3-kinase. Cancer Cell. 2003;3(5):483–95.
Westbrook TF, Martin ES, Schlabach MR, et al. A genetic screen for candidate tumor suppressors identifies REST. Cell. 2005;121(6):837–48.
Coulson JM, Fiskerstrand CE, Woll PJ, et al. Arginine vasopressin promoter regulation is mediated by a neuron-restrictive silencer element in small cell lung cancer. Cancer Res. 1999;59(20):5123–7.
Xiao H, Verdier P, Fernandez N, et al. Insights into the mechanism of microtubule stabilization by taxol. Proc Natl Acad Sci. 2006;103(27):10166–73.
Ohishi Y, Oda Y, Basaki Y, et al. Expression of beta-tubulin isotypes in human primary ovarian carcinoma. Gynecol Oncol. 2007;105(3):586–92.
Gan PP, Pasquier E, Kavallaris M. Class III beta-tubulin mediates sensitivity to chemotherapeutic drugs in non small cell lung cancer. Cancer Res. 2007;67(19):9356–63.
Izutsu N, Maesawa C, Shibazaki M, et al. Epigenetic modification is involved in aberrant expression of class III beta-tubulin, TUBB3, in ovarian cancer cells. Int J Oncol. 2008;32(6):1227–35.
Akasaka K, Maesawa C, Shibazaki M, et al. Loss of class III beta-tubulin induced by histone deacetylation is associated with chemosensitivity to paclitaxel in malignant melanoma cells. J Invest Dermatol. 2009;129(6):1516–26.
Reddy BY, Greco SJ, Patel PS, et al. RE-1-silencing transcription factor shows tumor-suppressor functions and negatively regulates the oncogenic TAC1 in breast cancer cells. Proc Natl Acad Sci USA. 2009;106(11):4408–13.
Mozzetti S, Ferlini C, Concolino P, et al. Class III beta-tubulin overexpression is a prominent mechanism of paclitaxel resistance in ovarian cancer patients. Clin Cancer Res. 2005;11(1):298–305.
Gao Z, Ure K, Ding P, et al. The master negative regulator REST/NRSF controls adult neurogenesis by restraining the neurogenic program in quiescent stem cells. J Neurosci. 2011;31(26):9772–86.
Moss AC, Jacobson GM, Walker LE, et al. SCG3 transcript in peripheral blood is a prognostic biomarker for REST-deficient small cell lung cancer. Clin Cancer Res. 2009;15(1):274–83.
Guardavaccaro D, Frescas D, Dorrello NV, et al. Control of chromosome stability by the beta-TrCP-REST-Mad2 axis. Nature. 2008;452(7185):365–9.
Su X, Gopalakrishnan V, Stearns D, et al. Abnormal expression of REST/NRSF and Myc in neural stem/progenitor cells causes cerebellar tumors by blocking neuronal differentiation. Mol Cell Biol. 2006;26(5):1666–78.
Weissman AM. How much REST is enough? Cancer Cell. 2008;13(5):381–3.
Westbrook TF, et al. A genetic screen for candidate tumor suppressors identifies REST. Cell. 2005;121(6):837–48.
Dumontet C, Isaac S, Souquet PJ, et al. Expression of class III beta tubulin in non-small cell lung cancer is correlated with resistance to taxane chemotherapy. Bull Cancer. 2005;92(2):E25–30.
Kamath K, Wilson L, Cabral F, et al. Beta III-tubulin induces paclitaxel resistance in association with reduced effects on microtubule dynamic instability[J]. J Biol Chem. 2005;280(13):12902–7.
Acknowledgments
This study was supported by funding from the Scientific Research Fund of Liaoning Provincial Education (No. L2011129).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, S., Zhao, X., Lin, B. et al. Clinical implications of REST and TUBB3 in ovarian cancer and its relationship to paclitaxel resistance. Tumor Biol. 33, 1759–1765 (2012). https://doi.org/10.1007/s13277-012-0435-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0435-y