Abstract
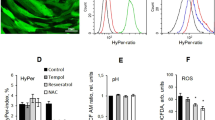
Cellular senescence of mesenchymal stem cells (MSCs) is often induced during in vitro expansion, by multiple experimental stimuli including oxidative stress. In this study, we investigated expression of senescence-associated proteins including SIRT1after inducing premature senescence of MSCs with hydrogen peroxide (H2O2). We also analyzed the effect of resveratrol (RSV) on premature senescence. We found that H2O2 triggered the recruitment of RCK (p54) to P-bodies in MSCs. Premature senescence of MSCs in response to H2O2 induced a decrease in SIRT1expression and activity (indirectly identified by measuring H3-K9ac). Cellular expression of p21 and phosphorylation of ERK1/2 and p38 kinases were increased in response to H2O2, whereas phosphorylation of pRb was decreased. In contrast, RSV pretreatment resulted in a decrease in the premature senescence of MSCs. In addition, RSV pretreatment before exposing cells to H2O2 not only alleviated changes in the levels of proteins that were sensitive to the H2O2 treatment (SIRT1, p21,ERK1/2 and p38) but also inhibited the decrease of SIRT1 induced by nicotinamide (NAM). Our results suggest that MSCs may exhibit an increased tolerance for H2O2-induced oxidative stress via the senescence-associated proteins that are regulated by RSV.
Similar content being viewed by others
References
Choi, M. R. et al. Selection of optimal passage of bone marrow-derived mesenchymal stem cells for stem cell therapy in patients with amyotrophic lateral sclerosis. Neurosci Lett 472:94–98 (2010).
Bruder, S. P., Jaiswal, N. & Haynesworth, S. E. Growth kinetics, self-renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation. J Cell Biochem 64:278–294 (1997).
Rombouts, W. J. & Ploemacher, R. E. Primary murine MSC show highly efficient homing to the bone marrow but lose homing ability following culture. Leukemia 17:160–170 (2003).
Choi, M. R. et al. Genome-scale DNA methylation pattern profiling of human bone marrow mesenchymal stem cells in long-term culture. Exp Mol Med 44:503–512 (2012).
Emara, M. M. et al. Hydrogen peroxide induces stress granule formation independent of eIF2alpha phosphorylation. Biochem Biophys Res Commun 423:763–769 (2012).
Ohn, T. & Anderson, P. The role of posttranslational modifications in the assembly of stress granules. Wiley Interdiscip Rev RNA 1:486–493 (2010).
Anderson, P. & Kedersha, N. RNA granules. J Cell Biol 172:803–808 (2006).
Kamata, H. & Hirata, H. Redox regulation of cellular signalling. Cell Signal 11:1–14 (1999).
Hong, E. H. et al. Ionizing radiation induces cellular senescence of articular chondrocytes via negative regulation of SIRT1 by p38 kinase. J Biol Chem 285:1283–1295 (2010).
Brandl, A., Meyer, M., Bechmann, V., Nerlich, M. & Angele, P. Oxidative stress induces senescence in human mesenchymal stem cells. Exp Cell Res 317:1541–1547 (2011).
Kim, J. S. et al. Proteomic and metabolomic analysis of H2O2-induced premature senescent human mesenchymal stem cells. Exp Gerontol 46:500–510 (2011).
Lee, J. S. et al. Senescent growth arrest in mesenchymal stem cells is bypassed by Wip1-mediated downregulation of intrinsic stress signaling pathways. Stem Cells 27:1963–1975 (2009).
Zhang, W. et al. Comparison of global DNA methylation profiles in replicative versus premature senescence. Life Sci 83:475–480 (2008).
Chen, W., Rezaizadehnajafi, L. & Wink, M. Influence of resveratrol on oxidative stress resistance and life span in Caenorhabditis elegans. J Pharm Pharmacol 65:682–688 (2013).
Pandey, K. B. & Rizvi, S. I. Resveratrol up-regulates the erythrocyte plasma membrane redox system and mitigates oxidation-induced alterations in erythrocytes during aging in humans. Rejuvenation Res 16:232–240 (2013).
Quincozes-Santos, A. et al. Resveratrol Protects C6 Astrocyte Cell Line against Hydrogen Peroxide-Induced Oxidative Stress through Heme Oxygenase 1. PloS one 8:e64372 (2013).
Brunet, A. et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303:2011–2015 (2004).
Huang, J. et al. SIRT1 overexpression antagonizes cellular senescence with activated ERK/S6k1 signaling in human diploid fibroblasts. PloS one 3:e1710 (2008).
Langley, E. et al. Human SIR2 deacetylates p53 and antagonizes PML/p53-induced cellular senescence. EMBO J 21:2383–2396 (2002).
Vaquero, A. et al. Human SirT1 interacts with histone H1 and promotes formation of facultative heterochromatin. Mol Cell 16:93–105 (2004).
Klimova, T. A. et al. Hyperoxia-induced premature senescence requires p53 and pRb, but not mitochondrial matrix ROS. FASEB J 23:783–794 (2009).
Han, M. K. et al. SIRT1 regulates apoptosis and Nanog expression in mouse embryonic stem cells by controlling p53 subcellular localization. Cell Stem Cell 2:241–251 (2008).
Ju, Z., Choudhury, A. R. & Rudolph, K. L. A dual role of p21 in stem cell aging. Ann N Y Acad Sci 1100:333–344 (2007).
Anderson, P. & Kedersha, N. Stressful initiations. J Cell Sci 115:3227–3234 (2002).
Guarente, L. Diverse and dynamic functions of the Sir silencing complex. Nat Genet 23:281–285 (1999).
Vaziri, H. et al. hSIR2 (SIRT1) functions as an NAD-dependent p53 deacetylase. Cell 107:149–159 (2001).
Kao, C. L. et al. Resveratrol protects human endothelium from H(2)O(2)-induced oxidative stress and senescence via SirT1 activation. J Atheroscler Thromb 17: 970–979 (2010).
Yuan, H. F. et al. SIRT1 is required for long-term growth of human mesenchymal stem cells. J Mol Med (Berl) 90:389–400 (2012).
Chaudhary, N. & Pfluger, P. T. Metabolic benefits from Sirt1 and Sirt1 activators. Curr Opin Clin Nutr Metab Care 12:431–437 (2009).
Penumathsa, S. V. & Maulik, N. Resveratrol: a promising agent in promoting cardioprotection against coronary heart disease. Can J Physiol Pharmacol 87:275–286 (2009).
Baur, J. A. & Sinclair, D. A. Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov 5:493–506 (2006).
Lin, H. Y. et al. Resveratrol causes COX-2- and p53-dependent apoptosis in head and neck squamous cell cancer cells. J Cell Biochem 104:2131–2142 (2008).
Cheng, T. et al. Hematopoietic stem cell quiescence maintained by p21cip1/waf1. Science 287:1804–1808 (2000).
Kippin, T. E., Martens, D. J. & van der Kooy, D. p21 loss compromises the relative quiescence of forebrain stem cell proliferation leading to exhaustion of their proliferation capacity. Genes Dev 19:756–767 (2005).
Yu, J. M. et al. Age-related changes in mesenchymal stem cells derived from rhesus macaque bone marrow. Aging Cell 10:66–79 (2011).
Herbig, U., Jobling, W. A., Chen, B. P., Chen, D. J. & Sedivy, J. M. Telomere shortening triggers senescence of human cells through a pathway involving ATM, p53, and p21 (CIP1), but not p16 (INK4a). Mol Cell 14:501–513 (2004).
Martindale, J. L. & Holbrook, N. J. Cellular response to oxidative stress: signaling for suicide and survival. J Cell Physiol 192:1–15 (2002).
Nebreda, A. R. & Porras, A. p38 MAP kinases: beyond the stress response. Trends Biochem Sci 25:257–260 (2000).
Pearson, G. et al. Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev 22:153–183 (2001).
Conde de la Rosa, L. et al. Superoxide anions and hydrogen peroxide induce hepatocyte death by different mechanisms: involvement of JNK and ERK MAP kinases. J Hepatol 44:918–929 (2006).
Czaja, M. J., Liu, H. & Wang, Y. Oxidant-induced hepatocyte injury from menadione is regulated by ERK and AP-1 signaling. Hepatology 37:1405–1413 (2003).
Guyton, K. Z. et al. Age-related changes in activation of mitogen-activated protein kinase cascades by oxidative stress. J Investig Dermatol Symp Proc 3:23–27 (1998).
Yu, J. et al. Involvement of oxidative stress and mitogen-activated protein kinase signaling pathways in heat stress-induced injury in the rat small intestine. Stress 16:99–113 (2013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, M.R., Han, D.M.R., Kim, S.H. et al. Resveratrol relieves hydrogen peroxide-induced premature senescence associated with SIRT1 in human mesenchymal stem cells. Mol. Cell. Toxicol. 10, 29–39 (2014). https://doi.org/10.1007/s13273-014-0004-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-014-0004-0