Abstract
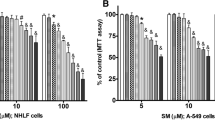
N-Nitrosodialkylamines are considered a potential health hazard for humans. Among various N-nitrosodialkylamines compound, N-nitrosodimethylamine (NDMA), was applied to lung cells, MLG. In order to examine the effect of NDMA in lung cells, cell death was first examined. NDMA induced cell death in a dose-dependent manner. Mitogen activated protein kinase (MAPK) involvement was tested using SP600125 for JNK, SB203580 for P38 MAPK, or PD98059 for ERK. SP compound induced a significant reduction of cell death, compared with other inhibitors. NDMA induced an increase in expression of ER stress response proteins, including GRP78, IRE-1α, CHOP, GRP94, p-eIF2α, p-JNK, or sXBP-1. Pretreatment with SP compound regulated expression of CHOP, the transcription factor associated with proapoptotic gene induction. This study showed that NDMA-induced cell death was resulted due to JNK activation. ER stress was also induced in the toxic situations. Study of the meaning of ER stress will be necessary in the near future.
Similar content being viewed by others
References
Witschi, H., Wilson, D. W. & Plopper, C. G. Modulation of N-nitrosodiethylamine-induced hamster lung tumors by ozone. Toxicology 77:193–202 (1993).
Giri, R. K., Baral, R. N. & Das, B. R. Induction of lung carcinogenesis in AKR-mice by N-nitrosodiethylamine/phenobarbitone, associated with high expression of c-myc and c-jun oncoproteins. Cancer Lett 112:57–63 (1997).
Ramakrishna, G. et al. Ki-ras and the characteristics of mouse lung tumors. Mol Carcinog 28:156–167 (2000).
Souliotis, V. L. et al. DNA adducts, mutant frequencies and mutation spectra in lambda lacZ transgenic mice treated with N-nitrosodimethylamine. Carcinogenesis 19:731–739 (1998).
Oury, B., Limasset, J. C. & Protois, J. C. Assessment of exposure to carcinogenic N-nitrosamines in the rubber industry. Int Arch Occup Environ Health 70:261–271 (1997).
Westin, J. B., Castegnaro, M. J. & Friesen, M. D. Nnitrosamines and nitrosatable amines, potential precursors of N-nitramines, in children’s pacifiers and babybottle nipples. Environ Res 43:126–134 (1987).
Erkekoglu, P. & Baydar, T. Evaluation of the protective effect of ascorbic acid on nitrite- and nitrosamineinduced cytotoxicity and genotoxicity in human hepatoma line. Toxicol Mech Methods 20:45–52 (2010).
Kamendulis, L. M. & Corcoran, G. B. Dimethylnitrosamine-induced DNA damage and toxic cell death in cultured mouse hepatocytes. J Toxicol Environ Health 46:31–46 (1995).
Paz-Elizur, T. et al. DNA repair activity for oxidative damage and risk of lung cancer. J Natl Cancer Inst 95:1312–1319 (2003).
Robbiano, L., Baroni, D., Novello, L. & Brambilla, G. Correlation between induction of DNA fragmentation in lung cells from rats and humans and carcinogenic activity. Mutat Res 605:94–102 (2006).
Chen, L., Ovesen, J. L., Puga, A. & Xia, Y. Distinct contributions of JNK and p38 to chromium cytotoxicity and inhibition of murine embryonic stem cell differentiation. Environ Health Perspect 117:1124–1130 (2009).
Schaeffer, H. J. & Weber, M. J. Mitogen-activated protein kinases: specific messages from ubiquitous messengers. Mol Cell Biol 19:2435–2444 (1999).
Liu, H. et al. Enantioselective cytotoxicity of isocarbophos is mediated by oxidative stress-induced JNK activation in human hepatocytes. Toxicology 276:115–121 (2010).
Kaufman, R. J. Orchestrating the unfolded protein response in health and disease. J Clin Invest 110:1389–1398 (2002).
Rutkowski, D. T. & Kaufman, R. J. A trip to the ER: coping with stress. Trends Cell Biol 14:20–28 (2004).
Oyadomari, S., Araki, E. & Mori, M. Endoplasmic reticulum stress-mediated apoptosis in pancreatic betacells. Apoptosis 7:335–345 (2002).
Zhang, K. & Kaufman, R. J. Signaling the unfolded protein response from the endoplasmic reticulum. J Biol Chem 279:25935–25938 (2004).
Su, Q. et al. Modulation of the eukaryotic initiation factor 2 alpha-subunit kinase PERK by tyrosine phosphorylation. J Biol Chem 283:469–475 (2008).
Huang, J. et al. Progressive endoplasmic reticulum stress contributes to hepatocarcinogenesis in fatty acyl-CoA oxidase 1-deficient mice. Am J Pathol 179:703–713 (2011).
Fu, W. et al. Upregulation of GRP78 in renal cell carcinoma and its significance. Urology 75:603–607 (2010).
Jamora, C., Dennert, G. & Lee, A. S. Inhibition of tumor progression by suppression of stress protein GRP78/BiP induction in fibrosarcoma B/C10ME. Proc Natl Acad Sci USA 93:7690–7694 (1996).
Uramoto, H. et al. Expression of endoplasmic reticulum molecular chaperone Grp78 in human lung cancer and its clinical significance. Lung Cancer 49:55–62 (2005).
Ratajczak-Wrona, W. et al. Effect of N-nitrosodimethylamine on inducible nitric oxide synthase expression and production of nitric oxide by neutrophils and mononuclear cells: the role of JNK signalling pathway. APMIS 119:431–441 (2011).
Bellec, G. et al. Cytochrome P450 metabolic dealkylation of nine N-nitrosodialkylamines by human liver microsomes. Carcinogenesis 17:2029–2034 (1996).
Lin, H. L., Roberts, E. S. & Hollenberg, P. F. Heterologous expression of rat P450 2E1 in a mammalian cell line: in situ metabolism and cytotoxicity of Nnitrosodimethylamine. Carcinogenesis 19:321–329 (1998).
Boyd, J. M. et al. A cell-microelectronic sensing technique for profiling cytotoxicity of chemicals. Anal Chim Acta 615:80–87 (2008).
Lin, H. L., Parsels, L. A., Maybaum, J. & Hollenberg, P. F. N-Nitrosodimethylamine-mediated cytotoxicity in a cell line expressing P450 2E1: evidence for apoptotic cell death. Toxicol Appl Pharmacol 157:117–124 (1999).
Wu, J. & Kaufman, R. J. From acute ER stress to physiological roles of the unfolded protein response. Cell Death Differ 13:374–384 (2006).
Schroder, M. & Kaufman, R. J. The mammalian unfolded protein response. Annu Rev Biochem 74:739–789 (2005).
Kitamura, M. Endoplasmic reticulum stress and unfolded protein response in renal pathophysiology: Janus faces. Am J Physiol Renal Physiol 295:F323–334 (2008).
Kim, R., Emi, M., Tanabe, K. & Murakami, S. Role of the unfolded protein response in cell death. Apoptosis 11:5–13 (2006).
Szegezdi, E., Logue, S. E., Gorman, A. M. & Samali, A. Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep 7:880–885 (2006).
Wek, R. C., Jiang, H. Y. & Anthony, T. G. Coping with stress: eIF2 kinases and translational control. Biochem Soc Trans 34:7–11 (2006).
Yoshida, H. ER stress and diseases. FEBS J 274:630–658 (2007).
Matsukawa, J., Matsuzawa, A., Takeda, K. & Ichijo, H. The ASK1-MAP kinase cascades in mammalian stress response. J Biochem 136:261–265 (2004).
Urano, F. et al. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1. Science 287:664–666 (2000).
Choi, J. H. et al. Baicalein protects HT22 murine hippocampal neuronal cells against endoplasmic reticulum stress-induced apoptosis through inhibition of reactive oxygen species production and CHOP induction. Exp Mol Med 42:811–822 (2010).
Verma, G. & Datta, M. IL-1beta induces ER stress in a JNK dependent manner that determines cell death in human pancreatic epithelial MIA PaCa-2 cells. Apoptosis 15:864–876 (2010).
Yang, W., Tiffany-Castiglioni, E., Koh, H. C. & Son, I. H. Paraquat activates the IRE1/ASK1/JNK cascade associated with apoptosis in human neuroblastoma SH-SY5Y cells. Toxicol Lett 191:203–210 (2009).
Oh, S. H. & Lim, S. C. Endoplasmic reticulum stressmediated autophagy/apoptosis induced by capsaicin (8-methyl-N-vanillyl-6-nonenamide) and dihydrocapsaicin is regulated by the extent of c-Jun NH2-terminal kinase/extracellular signal-regulated kinase activation in WI38 lung epithelial fibroblast cells. J Pharmacol Exp Ther 329:112–122 (2009).
Ahmad, A., Kikuchi, H., Takami, Y. & Nakayama, T. Different roles of N-terminal and C-terminal halves of HIRA in transcription regulation of cell cycle-related genes that contribute to control of vertebrate cell growth. J Biol Chem 280:32090–32100 (2005).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, B., Kim, DS., Oh, HW. et al. N-Nitrosodimethylamine induced lung fibroblast cell death is associated with JNK activation. Mol. Cell. Toxicol. 8, 155–161 (2012). https://doi.org/10.1007/s13273-012-0019-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-012-0019-3