Abstract
Background
Under hypoxia, mouse embryonic stem cells (mESCs) lose the ability to self-renew and begin to differentiate through down-regulation of LIFR-STAT3 pathway via hypoxia-inducible factor-1α (HIF-1α). However, it remains largely unknown what kinds of factors are involved in hypoxia-induced differentiation of mESCs.
Purpose
This study aims to identify the differentially expressed genes (DEGs) in early differentiation of mESCs under hypoxia.
Methods
Here we utilized a Genefishing techniqueTM to discover the new DEGs during hypoxia-induced early differentiation in CCE mESCs. Next, we investigated the role of DEGs using morphological observation, alkaline phosphatase (ALP) assay, STAT3 activation analysis, and biomarkers analysis for stemness.
Results
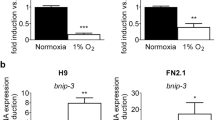
We detected 19 DEGs under hypoxia and performed cloning with sequencing in six genes. We confirmed the expression patterns of five DEGs including H2afz and GOT1 by realtime PCR assay. Among them, H2afz was significantly decreased under hypoxia, depending on HIF-1α. H2afz-overexpressing CCE mESCs maintained their ALP activity and stem cell markers (Nanog and Rex1), even in hypoxic condition. On the other hand, the early differentiation markers such as FGF5 and STAT5a, which had been increased in hypoxic conditions, were reduced by H2afz overexpression.
Conclusion
We discovered that H2afz could be a new target gene that functions in hypoxia-induced differentiation in mESCs and have revealed that it is involved in maintaining the pluripotency of mESCs in the early stages of differentiation. These findings will provide insights into mechanisms of hypoxia-mediated differentiation of mESCs during early development.





Similar content being viewed by others
References
Bost F, Caron L, Marchetti I, Dani C, Le Marchand-Brustel Y, Binetruy B (2002) Retinoic acid activation of the ERK pathway is required for embryonic stem cell commitment into the adipocyte lineage. Biochem J 361:621–627
Coleman-Derr D, Zilberman D (2012) Deposition of histone variant H2A.Z within gene bodies regulates responsive genes. PLoS Genet 8:e1002988
Du Z, Cong H, Yao Z (2001) Identification of putative downstream genes of Oct-4 by suppression-subtractive hybridization. Biochem Biophys Res Commun 282:701–706
Du KL, Chen M, Li J, Lepore JJ, Mericko P, Parmacek MS (2004) Megakaryoblastic leukemia factor-1 transduces cytoskeletal signals and induces smooth muscle cell differentiation from undifferentiated embryonic stem cells. J Biol Chem 279:17578–17586
Ernst M, Novak U, Nicholson SE, Layton JE, Dunn AR (1999) The carboxyl-terminal domains of gp130-related cytokine receptors are necessary for suppressing embryonic stem cell differentiation. Involvement of STAT3. J Biol Chem 274:9729–9737
Evans MJ, Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292:154–156
Faast R, Thonglairoam V, Schulz TC, Beall J, Wells JR, Taylor H, Matthaei K, Rathjen PD, Tremethick DJ, Lyons I (2001) Histone variant H2A.Z is required for early mammalian development. Curr Biol 11:1183–1187
Gassmann M, Fandrey J, Bichet S, Wartenberg M, Marti HH, Bauer C, Wenger RH, Acker H (1996) Oxygen supply and oxygen-dependent gene expression in differentiating embryonic stem cells. Proc Natl Acad Sci U S A 93:2867–2872
Hwang IT, Kim YJ, Kim SH, Kwak CI, Gu YY, Chun JY (2003) Annealing control primer system for improving specificity of PCR amplification. Biotechniques 35:1180–1184
Jeong JW, Bae MK, Ahn MY, Kim SH, Sohn TK, Bae MH, Yoo MA, Song EJ, Lee KJ, Kim KW (2002) Regulation and destabilization of HIF-1alpha by ARD1-mediated acetylation. Cell 111:709–720
Jeong CH, Lee HJ, Cha JH, Kim JH, Kim KR, Kim JH, Yoon DK, Kim KW (2007) Hypoxia-inducible factor-1 alpha inhibits self-renewal of mouse embryonic stem cells in Vitro via negative regulation of the leukemia inhibitory factor-STAT3 pathway. J Biol Chem 282:13672–13679
Kim KJ, Choi JS, Kang I, Kim KW, Jeong CH, Jeong JW (2013) Melatonin suppresses tumor progression by reducing angiogenesis stimulated by HIF-1 in a mouse tumor model. J Pineal Res 54:264–270
Land SC (2004) Hochachka’s “Hypoxia Defense Strategies” and the development of the pathway for oxygen. Comp Biochem Physiol B Biochem Mol Biol 139:415–433
Lee HJ, Kim KW (2012) Suppression of HIF-1alpha by valproic acid sustains self-renewal of mouse embryonic stem cells under hypoxia in vitro. Biomol Ther (Seoul) 20:280–285
Lee JW, Bae SH, Jeong JW, Kim SH, Kim KW (2004) Hypoxia-inducible factor (HIF-1)alpha: its protein stability and biological functions. Exp Mol Med 36:1–12
Lee HJ, Jeong CH, Cha JH, Kim KW (2010) PKC-delta inhibitors sustain self-renewal of mouse embryonic stem cells under hypoxia in vitro. Exp Mol Med 42:294–301
Li B, Pattenden SG, Lee D, Gutierrez J, Chen J, Seidel C, Gerton J, Workman JL (2005) Preferential occupancy of histone variant H2AZ at inactive promoters influences local histone modifications and chromatin remodeling. Proc Natl Acad Sci USA 102:18385–18390
Li Z, Wu JC, Sheikh AY, Kraft D, Cao F, Xie X, Patel M, Gambhir SS, Robbins RC, Cooke JP et al (2007) Differentiation, survival, and function of embryonic stem cell derived endothelial cells for ischemic heart disease. Circulation 116:I46-54
Masuda S, Kobayashi T, Chikuma M, Nagao M, Sasaki R (2000) The oviduct produces erythropoietin in an estrogen- and oxygen-dependent manner. Am J Physiol Endocrinol Metab 278:E1038-1044
Mie Lee Y, Kim SH, Kim HS, Jin Son M, Nakajima H, Jeong Kwon H, Kim KW (2003) Inhibition of hypoxia-induced angiogenesis by FK228, a specific histone deacetylase inhibitor, via suppression of HIF-1alpha activity. Biochem Biophys Res Commun 300:241–246
Mizuguchi G, Shen X, Landry J, Wu WH, Sen S, Wu C (2004) ATP-driven exchange of histone H2AZ variant catalyzed by SWR1 chromatin remodeling complex. Science 303:343–348
Niwa H, Burdon T, Chambers I, Smith A (1998) Self-renewal of pluripotent embryonic stem cells is mediated via activation of STAT3. Genes Dev 12:2048–2060
Peyssonnaux C, Zinkernagel AS, Schuepbach RA, Rankin E, Vaulont S, Haase VH, Nizet V, Johnson RS (2007) Regulation of iron homeostasis by the hypoxia-inducible transcription factors (HIFs). J Clin Invest 117:1926–1932
Powers DE, Millman JR, Huang RB, Colton CK (2008) Effects of oxygen on mouse embryonic stem cell growth, phenotype retention, and cellular energetics. Biotechnol Bioeng 101:241–254
Schrenk-Siemens K, Perez-Alcala S, Richter J, Lacroix E, Rahuel J, Korte M, Muller U, Barde YA, Bibel M (2008) Embryonic stem cell-derived neurons as a cellular system to study gene function: lack of amyloid precursor proteins APP and APLP2 leads to defective synaptic transmission. Stem Cells 26:2153–2163
Semenza GL (2001) HIF-1, O(2), and the 3 PHDs: how animal cells signal hypoxia to the nucleus. Cell 107:1–3
Shui YB, Fu JJ, Garcia C, Dattilo LK, Rajagopal R, McMillan S, Mak G, Holekamp NM, Lewis A, Beebe DC (2006) Oxygen distribution in the rabbit eye and oxygen consumption by the lens. Invest Ophthalmol Vis Sci 47:1571–1580
Simon MC, Keith B (2008) The role of oxygen availability in embryonic development and stem cell function. Nat Rev Mol Cell Biol 9:285–296
Soboleva TA, Nekrasov M, Pahwa A, Williams R, Huttley GA, Tremethick DJ (2011) A unique H2A histone variant occupies the transcriptional start site of active genes. Nat Struct Mol Biol 19:25–30
Tighe AP, Gudas LJ (2004) Retinoic acid inhibits leukemia inhibitory factor signaling pathways in mouse embryonic stem cells. J Cell Physiol 198:223–229
Yu J, Thomson JA (2008) Pluripotent stem cell lines. Genes Dev 22:1987–1997
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2019R1C1C1003558) and the Ministry of Education, Science and Technology (2019R1A6A3A01091754).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hwang, S., Lee, HJ. Identification of differentially expressed genes in mouse embryonic stem cell under hypoxia. Genes Genom 43, 313–321 (2021). https://doi.org/10.1007/s13258-020-01009-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-020-01009-4