Abstract
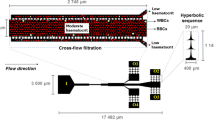
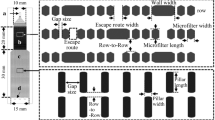
Red blood cell (RBC) deformability has become one of the important factors to assess blood and cardiovascular diseases. The interest on blood studies have promoted a development of various microfluidic devices that treat and analyse blood cells. Recent years, besides the RBC deformability assessment, these devices are often applied to cancer cell detection and isolation from the whole blood. The devices for cancer cell isolation rely mainly on size and deformability of the cells. However, the examination of deformability of the RBCs mixed with cancer cells is lacking. This study aims at determining the deformation index (DI) of the RBCs in contact with cancer cells using a hyperbolic microchannel which generates a strong extensional flow. The DIs of human healthy RBCs and human RBCs in contact with a tumor cell line (HCT-15, colon carcinoma) were compared by analyzing the flowing RBCs images captured by a high speed camera. The results reveal that the RBCs that were in contact with HCT-15 cells have lower deformability than the normal RBCs.
Similar content being viewed by others
References
Caro, C., Pedley, T., Schroter, R. & Seed, W. The Mechanics of the Circulation. Oxford University Press (1978).
Skalak, R. & Branemark, P-I. Deformation of red blood cells in capillaries. Science 164, 717–719 (1969).
Abkarian, M. et al. Cellular-scale hydrodynamics. Biomed. Mater. 3, 034011 (2008).
Hardeman, M.R. & Ince, C. Clinical potential of in vitro measured red cell deformability, a myth? Clin. Hemorheol. Microcirc. 21, 277–284 (1999).
Cho, Y.I., Mooney, M.P. & Cho, D.J. Hemorheological disorders in diabetes mellitus. J. Diabetes Sci. Technol. 2, 1130–1138 (2008).
Gueguen, M. et al. Filtration pressure and red blood cell deformability: evaluation of a new device: erythrometre. Biorheology Suppl 1, 261–265 (1984).
Shin, S., Ku, Y., Park, M.S. & Suh, J.S. Measurement of red cell deformability and whole blood viscosity using laser-diffraction slit rheometer. Korea-Australia Rheol. J. 16, 85–90 (2004).
Dobbe, J.G.G. et al. Analyzing red blood cell-deformability distributions. Blood Cells Mol. Dis. 28, 373–384 (2002).
Mokken, F.C., Kedaria, M., Henny, C.P., Hardeman, M.R. & Gelb, A.W. The clinical importance of erythtrocyte deformability, a hemorrheological parameter. Ann. Hematol. 64, 113–122 (1992).
Bow, H. et al. A microfabricated deformability-based flow cytometer with application to malaria. Lab Chip 11, 1065–1073 (2011).
Lima, R. et al. In vitro blood flow in a rectangular PDMS microchannel: experimental observations using a confocal micro-PIV system. Biomed. Microdevices 10, 153–167 (2008).
Fujiwara, H. et al. Red blood cell motions in a high hematocrit blood flowing through a stenosed microchannel. J. Biomech. 42, 838–843 (2009).
Lima, R. et al. Axisymmetric PDMS microchannels for in vitro haemodynamics studies. Biofabrication 1, 035005 (2009).
Lee, S.S., Yim, Y., Ahn, K.H. & Lee, S.J. Extensional flow-based assessment of red blood cell deformability using hyperbolic converging microchannel. Biomed. Microdevices 11, 1021–1027 (2009).
Zhao, R. et al. Microscopic investigation of erythrocyte deformation dynamics. Biorheology 43, 747–765 (2006).
Yaginuma, T., Oliveira, M.S.N., Lima, R., Ishikawa, T. & Yamaguchi, T. Human red blood cell behavior under homogeneous extensional flow in a hyperbolicshaped microchannel. Biomicrofluidics 7, 054110 (2013).
Pinho, D., Yaginuma, T. & Lima, R. A microfluidic device for partial cell separation and deformability assessment. BioChip J. 7, 367–374 (2013).
Hou, H.W. et al. Microfluidics for applications in cell mechanics and mechanobiology. Cel. Mol. Bioeng. 4, 591–602, (2011).
Tan, S.J., Yobas, L., Lee, G.Y., Ong, C.N. & Lim, C.T. Microdevice for the isolation and enumeration of cancer cells from blood. Biomed. Microdevices 11, 883–892 (2009).
Mohamed, H., Murray, M., Turner, J.N. & Caggana, M. Isolation of tumor cells using size and deformation. J. Chromatogr. A 1216, 8289–8295 (2009).
Hur, S.C., Henderson-MacLennan, N.K., McCabe, E.R. & Di Carlo, D. Deformability-based cell classification and enrichment using inertial microfluidics. Lab Chip 11, 912–920 (2011).
Tanaka, T. et al. Separation of cancer cells from a red blood cell suspension using inertial force. Lab Chip 12, 4336–4343 (2012).
Lee, M.G., Shin, J.H., Bae, C.Y., Choi, S. & Park, J.K. Label-free cancer cell separation from human whole blood using inertial microfluidics at low shear stress. Anal. Chem. 85, 6213–6218 (2013).
Jain, R.K. Determinants of tumor blood flow: a review. Cancer Res. 48, 2641–2658 (1988).
Sevick, E.M. & Jain, R.K. Effect of red blood cell rigidity on tumor blood flow: increase in viscous resistance during hyperglycemia. Cancer Res. 51, 2727–2730 (1991).
Kuzman, D. et al. Effect of pH on red blood cell deformability. Pflugers Arch. 440, R193–194 (2000).
Griffiths, J.R. Are cancer cells acidic? Br. J. Cancer 64, 425–427 (1991).
Estrella, V. et al. Acidity generated by the tumor microenvironment drives local invasion. Cancer Res. 73, 1524–1535 (2013).
Otsu, N. A threshold selection method from gray-level histograms. IEEE Trans. Syst. Man Cybern. 9, 62–66 (1979).
Abramoff, M.D., Magalhaes, P.J. & Ram, S.J. Image Processing with ImageJ. Biophotonics Int. 11, 36–42 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Faustino, V., Pinho, D., Yaginuma, T. et al. Extensional flow-based microfluidic device: deformability assessment of red blood cells in contact with tumor cells. BioChip J 8, 42–47 (2014). https://doi.org/10.1007/s13206-014-8107-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-014-8107-1