Abstract
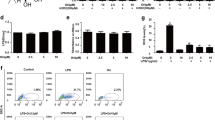
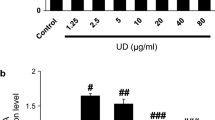
This study was undertaken to investigate the effect of natural bioactive compound thearubigin on neonatal acute lung injury (ALI) using LPS-induced ALI as a model. We also attempted to understand the possible underlying mechanism. The effect of thearubigin on lung wet-to-dry weight ratio, the activity of LDH, lung histopathology, BALF protein levels, the activity of MPO, production and extravasation of cytokines and oxidative stress were studied. The results showed that thearubigin caused a significant reduction in lung inflammation as evident from lung wet-to-dry weight ratio, BALF protein levels and MPO activity and histopathological analysis. It was further observed that the attenuation in inflammation happened due to a significant reduction in cytokine levels in alveolar cavities. Thearubigin also showed strong antioxidant properties as evidenced by reduced levels of oxygen species such as H2O2, MDA and OH ion. Additionally, the antioxidant response element nuclear factor erythroid-2-related factor 2 (Nrf2) pathway was found to be activated in thearubigin-treated group. These results provided a possible mechanism of antioxidant activity of thearubigin in neonatal ALI. Overall, this study showed that thearubigin can be a natural alternative for the treatment of neonatal ALI. However, further studies are required to understand its mechanism antioxidant and anti-inflammatory action.






Similar content being viewed by others
References
Ago T, Yeh I, Yamamoto M, Schinke-Braun M, Brown JA, Tian B, Sadoshima J (2006) Thioredoxin1 upregulates mitochondrial proteins related to oxidative phosphorylation and TCA cycle in the heart. Antioxid Redox Signal 8(9–10):1635–1650. https://doi.org/10.1089/ars.2006.8.1635
Cachofeiro V, Goicochea M, de Vinuesa SG, Oubina P, Lahera V, Luno J (2008) Oxidative stress and inflammation, a link between chronic kidney disease and cardiovascular disease. Kidney Int Suppl 111:S4–S9. https://doi.org/10.1038/ki.2008.516
Chagnon F, Bourgouin A, Lebel R, Bonin MA, Marsault E, Lepage M, Lesur O (2015) Smart imaging of acute lung injury: exploration of myeloperoxidase activity using in vivo endoscopic confocal fluorescence microscopy. Am J Physiol Lung Cell Mol Physiol 309(6):L543–L551. https://doi.org/10.1152/ajplung.00289.2014
Chakraborty M, McGreal EP, Kotecha S (2010) Acute lung injury in preterm newborn infants: mechanisms and management. Paediatr Respir Rev 11(3):162–170. https://doi.org/10.1016/j.prrv.2010.03.002 (quiz 170)
Cheifetz IM (2016) Year in review 2015: pediatric ARDS. Respir Care 61(7):980–985. https://doi.org/10.4187/respcare.05017
Deramaudt TB, Dill C, Bonay M (2013) Regulation of oxidative stress by Nrf2 in the pathophysiology of infectious diseases. Medecine et maladies infectieuses 43(3):100–107. https://doi.org/10.1016/j.medmal.2013.02.004
Ding Q, Liu GQ, Zeng YY, Zhu JJ, Liu ZY, Zhang X, Huang JA (2017) Role of IL-17 in LPS-induced acute lung injury: an in vivo study. Oncotarget 8(55):93704–93711. https://doi.org/10.18632/oncotarget.21474
Furukawa M, Tanaka R, Chuang VT, Ishima Y, Taguchi K, Watanabe H, Maruyama T, Otagiri M (2011) Human serum albumin-thioredoxin fusion protein with long blood retention property is effective in suppressing lung injury. J Control Release 154(2):189–195. https://doi.org/10.1016/j.jconrel.2011.05.013
Goodman RB, Pugin J, Lee JS, Matthay MA (2003) Cytokine-mediated inflammation in acute lung injury. Cytokine Growth Factor Rev 14(6):523–535
Grommes J, Soehnlein O (2011) Contribution of neutrophils to acute lung injury. Mol Med 17(3–4):293–307. https://doi.org/10.2119/molmed.2010.00138
Jacob S, Deyo DJ, Cox RA, Traber DL, Hawkins HK (2008) Assessment of lung inflammation in a mouse model of smoke inhalation and burn injury: strain-specific differences. Toxicol Mech Methods 18(7):551–559. https://doi.org/10.1080/15376510802251993
Jerala R (2007) Structural biology of the LPS recognition. Int J Med Microbiol 297(5):353–363. https://doi.org/10.1016/j.ijmm.2007.04.001
Karmpaliotis D, Kosmidou I, Ingenito EP, Hong K, Malhotra A, Sunday ME, Haley KJ (2002) Angiogenic growth factors in the pathophysiology of a murine model of acute lung injury. Am J Physiol Lung Cell Mol Physiol 283(3):585–595
Kim K, Li Y, Jin G, Chong W, Liu B, Lu J, Lee K, Demoya M, Velmahos GC, Alam HB (2012) Effect of valproic acid on acute lung injury in a rodent model of intestinal ischemia reperfusion. Resuscitation 83(2):243–248. https://doi.org/10.1016/j.resuscitation.2011.07.029
Lee JP, Li YC, Chen HY, Lin RH, Huang SS, Chen HL, Kuan PC, Liao MF, Chen CJ, Kuan YH (2010) Protective effects of luteolin against lipopolysaccharide-induced acute lung injury involves inhibition of MEK/ERK and PI3K/Akt pathways in neutrophils. Acta Pharmacol Sin 31(7):831–838. https://doi.org/10.1038/aps.2010.62
Lee HS, Kang P, Kim KY, Seol GH (2015) Foeniculum vulgare mill. Protects against lipopolysaccharide-induced acute lung injury in mice through ERK-dependent NF-kappaB activation. Korean J Physiol Pharmacol 19(2):183–189. https://doi.org/10.4196/kjpp.2015.19.2.183
Li Q, Verma IM (2002) NF-kappaB regulation in the immune system. Nat Rev Immunol 2(10):725–734. https://doi.org/10.1038/nri910
Li G, Zhou CL, Zhou QS, Zou HD (2016) Galantamine protects against lipopolysaccharide-induced acute lung injury in rats. Braz J Med Biol Res 49(2):e5008. https://doi.org/10.1590/1414-431x20155008
Liu J, Lin A (2007) Wiring the cell signaling circuitry by the NF-kappa B and JNK1 crosstalk and its applications in human diseases. Oncogene 26(22):3267–3278. https://doi.org/10.1038/sj.onc.1210417
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔ CT method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Lu YC, Yeh WC, Ohashi PS (2008) LPS/TLR4 signal transduction pathway. Cytokine 42(2):145–151. https://doi.org/10.1016/j.cyto.2008.01.006
Luh SP, Chiang CH (2007) Acute lung injury/acute respiratory distress syndrome (ALI/ARDS): the mechanism, present strategies and future perspectives of therapies. J Zhejiang Univ Sci B 8(1):60–69. https://doi.org/10.1631/jzus.2007.B0060
Maity S, Ukil A, Karmakar S, Datta N, Chaudhuri T, Vedasiromoni JR, Ganguly DK, Das PK (2003) Thearubigin, the major polyphenol of black tea, ameliorates mucosal injury in trinitrobenzene sulfonic acid-induced colitis. Eur J Pharmacol 470(1–2):103–112
Manicone AM (2009) Role of the pulmonary epithelium and inflammatory signals in acute lung injury. Expert Rev Clin Immunol 5(1):63–75. https://doi.org/10.1586/177666X.5.1.63
Matthay MA, Zemans RL (2011) The acute respiratory distress syndrome: pathogenesis and treatment. Annu Rev Pathol 6:147
Muller-Redetzky HC, Suttorp N, Witzenrath M (2014) Dynamics of pulmonary endothelial barrier function in acute inflammation: mechanisms and therapeutic perspectives. Cell Tissue Res 355(3):657–673. https://doi.org/10.1007/s00441-014-1821-0
Pedruzzi LM, Stockler-Pinto MB, Leite M Jr, Mafra D (2012) Nrf2-keap1 system versus NF-kappaB: the good and the evil in chronic kidney disease? Biochimie 94(12):2461–2466. https://doi.org/10.1016/j.biochi.2012.07.015
Rahman I, MacNee W (2012) Antioxidant pharmacological therapies for COPD. Curr Opin Pharmacol 12(3):256–265. https://doi.org/10.1016/j.coph.2012.01.015
Reumaux D, de Boer M, Meijer AB, Duthilleul P, Roos D (2003) Expression of myeloperoxidase (MPO) by neutrophils is necessary for their activation by anti-neutrophil cytoplasm autoantibodies (ANCA) against MPO. J Leukoc Biol 73(6):841–849
Rittirsch D, Flierl MA, Day DE, Nadeau BA, McGuire SR, Hoesel LM, Ipaktchi K, Zetoune FS, Sarma JV, Leng L, Huber-Lang MS, Neff TA, Bucala R, Ward PA (2008) Acute lung injury induced by lipopolysaccharide is independent of complement activation. J Immunol 180(11):7664–7672
Stodt UW, Stark J, Engelhardt UH (2015) Comparison of three strategies for the isolation of black tea thearubigins with a focus on countercurrent chromatography. J Food Compos Anal 43:160–168
Suliman HB, Carraway MS, Piantadosi CA (2003) Postlipopolysaccharide oxidative damage of mitochondrial DNA. Am J Respir Crit Care Med 167(4):570–579
Tasoulis MK, Livaditi O, Stamatakos M, Stefanaki C, Paneris P, Prigouris P, Flevari A, Goutas N, Vlachodimitropoulos D, Villiotou V, Douzinas EE (2009) High concentrations of reactive oxygen species in the BAL fluid are correlated with lung injury in rabbits after hemorrhagic shock and resuscitation. Tohoku J Exp Med 219(3):193–199
Trocha M, Merwid-Lad A, Chlebda E, Sozanski T, Piesniewska M, Gliniak H, Szelag A (2014) Influence of ezetimibe on selected parameters of oxidative stress in rat liver subjected to ischemia/reperfusion. Arch Med Sci AMS 10(4):817–824. https://doi.org/10.5114/aoms.2013.38087
Tsai CL, Lin YC, Wang HM, Chou TC (2014) Baicalein, an active component of Scutellaria baicalensis, protects against lipopolysaccharide-induced acute lung injury in rats. J Ethnopharmacol 153(1):197–206. https://doi.org/10.1016/j.jep.2014.02.010
Vomund S, Schafer A, Parnham MJ, Brune B, von Knethen A (2017) Nrf2, the master regulator of anti-oxidative responses. Int J Mol Sci. https://doi.org/10.3390/ijms18122772
Yang J, Li W, Duan M, Zhou Z, Lin N, Wang Z, Sun J, Xu J (2005) Large dose ketamine inhibits lipopolysaccharide-induced acute lung injury in rats. Inflamm Res 54(3):133–137. https://doi.org/10.1007/s00011-004-1334-5
Acknowledgements
The project is funded by the National Natural Science Foundation of China (No: 81660013).
Author information
Authors and Affiliations
Contributions
XW designed the study and carried out experiments. PH, SY, CW carried out experiments and interpreted the data. XW wrote the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All the animals’ upkeeping and experimental procedures were carried out in accordance with the ethical standards given in the Declaration of Helsinki. The animal experiments were conducted with prior approval from the institutional review board and institutional animal care and use committee (IACUC). All efforts were made to minimize the number of animals and their suffering.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, X., He, P., Yi, S. et al. Thearubigin regulates the production of Nrf2 and alleviates LPS-induced acute lung injury in neonatal rats. 3 Biotech 9, 451 (2019). https://doi.org/10.1007/s13205-019-1986-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-019-1986-z